EMPIRICAL GAS LAW T01/S01 Unit conversion 2740 torr and a temperature of 174 °C. If the piston is locked to maintain constant volume, calculate the pressure (atm) of the gas at 407 K. K = C + 273 1 atm = 760 torr the tolerance is +/-2% EMPIRICAL GAS LAW T01/SO2 An expandable weather balloon with a volume of 4.61 × 102 mL on the ground at 0.970 atm and 132 °C ascended to a height at which the temperature was 124 (°C) and the volume was 14.2 L. Calculate the atmospheric pressure (torr). Unit conversion K = C + 273 x 10 1 atm = 760 torr
EMPIRICAL GAS LAW T01/S01 Unit conversion 2740 torr and a temperature of 174 °C. If the piston is locked to maintain constant volume, calculate the pressure (atm) of the gas at 407 K. K = C + 273 1 atm = 760 torr the tolerance is +/-2% EMPIRICAL GAS LAW T01/SO2 An expandable weather balloon with a volume of 4.61 × 102 mL on the ground at 0.970 atm and 132 °C ascended to a height at which the temperature was 124 (°C) and the volume was 14.2 L. Calculate the atmospheric pressure (torr). Unit conversion K = C + 273 x 10 1 atm = 760 torr
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter5: Gases, Liquids, And Solids
Section: Chapter Questions
Problem 5.24P
Related questions
Question
Please help me with both parts, they are attached as 2 screenshots, make it clear what the answer to each part is, and double and triple check your answers previous tutors gave the wrong answers.
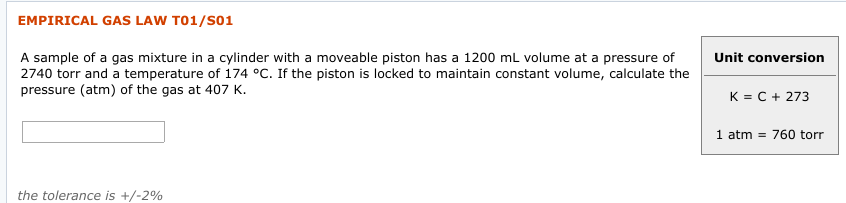
Transcribed Image Text:EMPIRICAL GAS LAW T01/S01
Unit conversion
2740 torr and a temperature of 174 °C. If the piston is locked to maintain constant volume, calculate the
pressure (atm) of the gas at 407 K.
K = C + 273
1 atm = 760 torr
the tolerance is +/-2%

Transcribed Image Text:EMPIRICAL GAS LAW T01/SO2
An expandable weather balloon with a volume of 4.61 × 102 mL on the ground at 0.970 atm and 132 °C
ascended to a height at which the temperature was 124 (°C) and the volume was 14.2 L. Calculate the
atmospheric pressure (torr).
Unit conversion
K = C + 273
x 10
1 atm = 760 torr
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 7 steps with 7 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning