ent Score: 50.1% Resources Lx Give Up? O Hint Check Ans 27 of 28 Attempt Consider the reaction. 2 Pb(s) + O,(8 2 PbO(s) An excess of oxygen reacts with 451.4 g of lead, forming 310.5 g of lead(II) oxide. Calculate the percent yield of the reaction. percent yield: + TOOLS x10 about us carcers privacy policy terms of use contact us neip
ent Score: 50.1% Resources Lx Give Up? O Hint Check Ans 27 of 28 Attempt Consider the reaction. 2 Pb(s) + O,(8 2 PbO(s) An excess of oxygen reacts with 451.4 g of lead, forming 310.5 g of lead(II) oxide. Calculate the percent yield of the reaction. percent yield: + TOOLS x10 about us carcers privacy policy terms of use contact us neip
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 60QAP: The text explains that one reason why the actual yield for a reaction may be less than the...
Related questions
Question
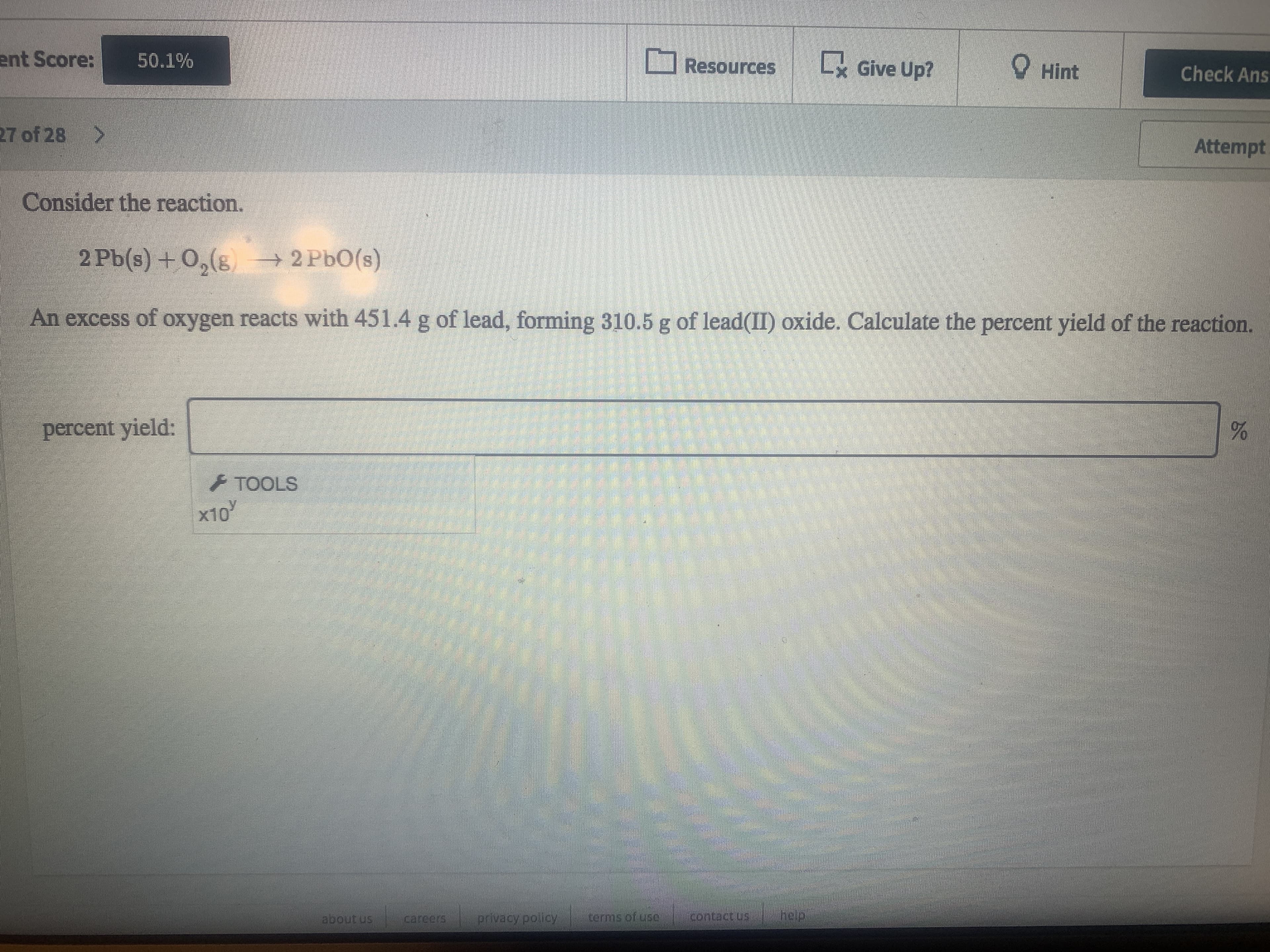
Transcribed Image Text:ent Score:
50.1%
Resources
Lx Give Up?
O Hint
Check Ans
27 of 28
Attempt
Consider the reaction.
2 Pb(s) + O,(8 2 PbO(s)
An excess of oxygen reacts with 451.4 g of lead, forming 310.5 g of lead(II) oxide. Calculate the percent yield of the reaction.
percent yield:
+ TOOLS
x10
about us
carcers
privacy policy
terms of use
contact us
neip
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning