Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
Transcribed Image Text:ngli
G Gran
Balar
Para
4 Build
A www
E Phet
Isoto
O Racia
Edwa
Tn Cour
C X
G tradu
If the
+
ege.com/course.html?courseld=17244391&OpenVellumHMAC=42c8679334b9bb195e40e403c3f01f9e#10001
E Reading list
GC1 sp22
Hi, Joelvi
Help
Sign Out
Course Home
<Post-Lecture Homework Chapter 03 Adaptive Follow-Up
Problem 3.8
< Item 5
II Review|
Constants | Periodic Table
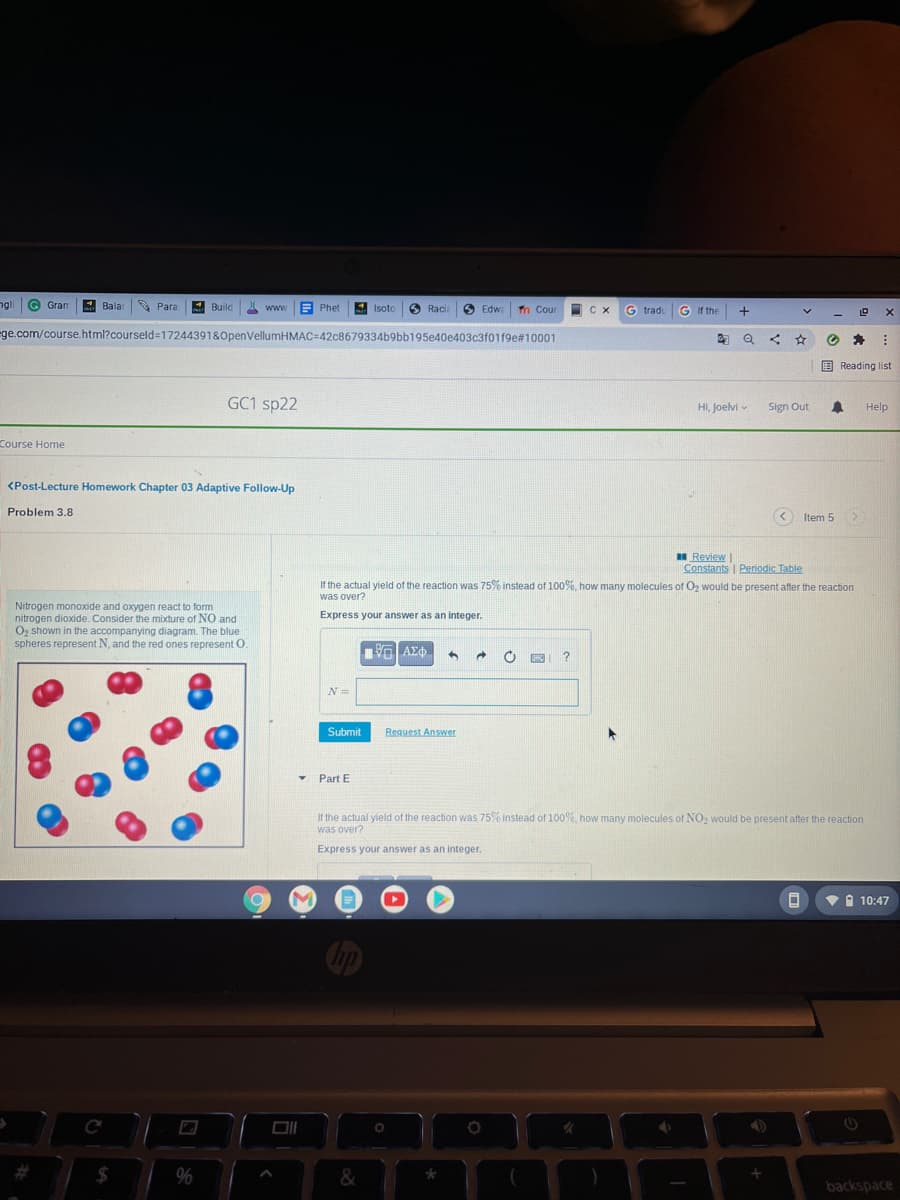
If the actual yield of the reaction was 75% instead of 100%, how many molecules of Oz would be present after the reaction
was over?
Nitrogen monoxide and oxygen react to form
nitrogen dioxide. Consider the mixture of NO and
Oz shown in the accompanying diagram. The blue
spheres represent N, and the red ones represent O.
Express your answer as an integer.
N =
Submit
Reguest Answer
Part E
If the actual yield of the reaction was 75% instead of 100%, how many molecules of NO2 would be present after the reaction
was over?
Express your answer as an integer.
VA 10:47
%23
%24
*
backspace
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY