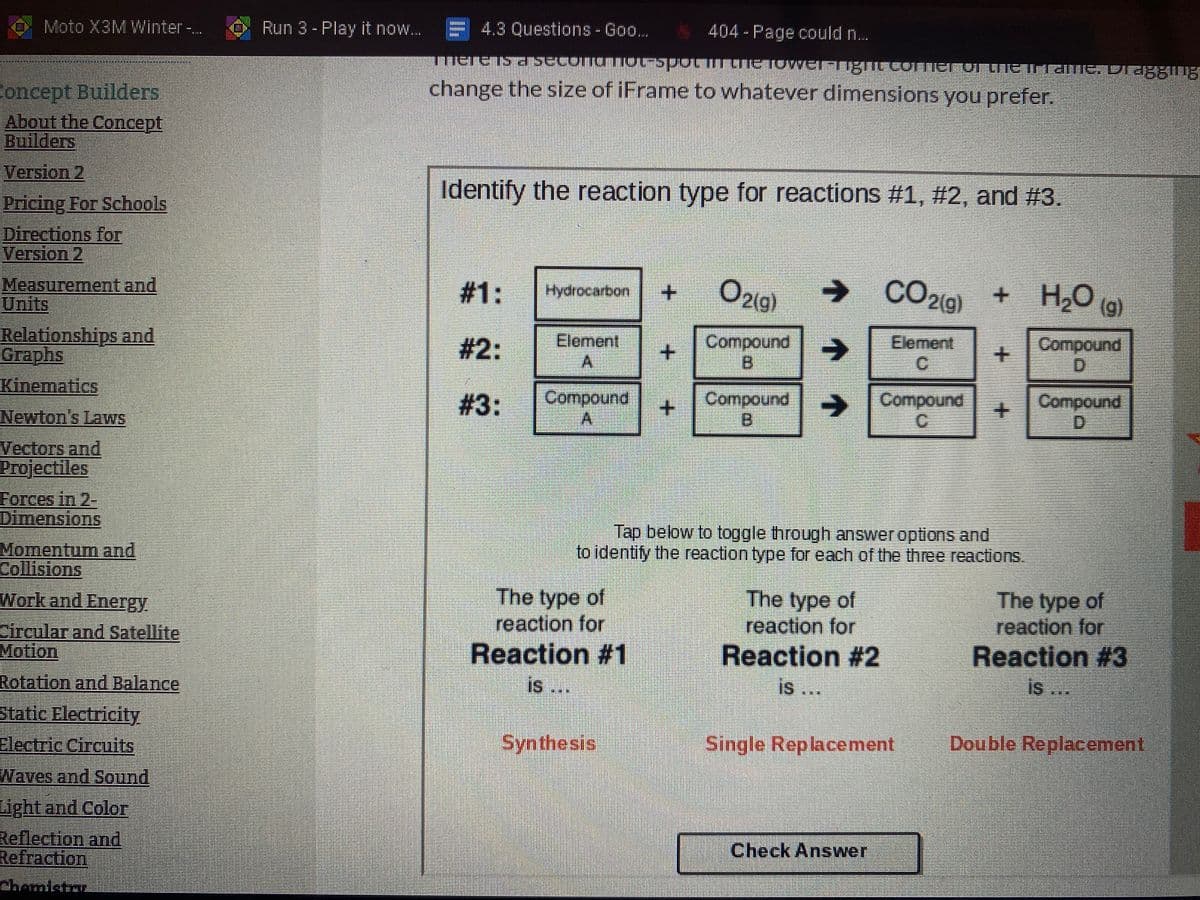
entify the reaction type for reactions #1, #2, and #3. 1: O219) -> CO2(9) + + H2O Hydrocarbon 2: Element Compound B Element C Compor D 3: Compound A Compound -> Compound + C Compo D Tap below to toggle through answer options and to identify the reaction type for each of the three reactions. The type of reaction for The type of reaction for The type c reaction fc Reaction #1 Reaction #2 Reaction is is is
entify the reaction type for reactions #1, #2, and #3. 1: O219) -> CO2(9) + + H2O Hydrocarbon 2: Element Compound B Element C Compor D 3: Compound A Compound -> Compound + C Compo D Tap below to toggle through answer options and to identify the reaction type for each of the three reactions. The type of reaction for The type of reaction for The type c reaction fc Reaction #1 Reaction #2 Reaction is is is
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.6PAE
Related questions
Question
In the red are my answers can u please help me
Transcribed Image Text:Moto X3M Winter-..
Run 3 - Play it now. E 4.3 Questions - Goo..
404-Page could n...
change the size of iFrame to whatever dimensions you prefer.
Concept Builders
About the Concept
Builders
Version 2
Identify the reaction type for reactions #1, #2, and #3.
Pricing For Schools
Directions for
Version 2
Measurement and
பாரட
#1:
O21g)
→ CO2(9)
H2O (g)
Hydrocarbon
Relationships and
Graphs
Kinematics
Element
Compound
B.
#2:
->
Element
Compound
Compound
Compound
B.
# 3:
->
SCompound
Compound
D.
+.
Newton's Lavws
Vectors and
Projectiles
Forces in 2-
DimensionS
Tap below to toggle through answer options and
to identity the reaction type for each of the three reactions.
Momentum and
Collisions
Work and Energy
The type of
reaction for
Reaction #1
The type of
reaction for
Reaction #2
is ..
The type of
reaction for
Reaction #3
is
Circular and Satellite
Motion
Rotation and Balance
is ..
s ..
Static Electricity
Electric Circuits
Synthesis
Single Replacement
Double Replacement
Waves and Sound
Light and Color
Reflection and
Refraction
Check Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning