Entropy, denoted by the symbol S, is the thermodynamic property describing the amount of molecular randomness or disorder in a system. It is a state thermodynamic function, meaning that its value does not depend on the path used to arrive at a specific set of conditions. Each system with a specified temperature, pressure, and composition has a unique entropy value. The third law of thermodynamics relates the entropy (randomness) of matter to its absolute temperature. Which of the following statements are in agreement with the third law of thermodynamics? Select all that apply. • View Available Hint(s) The entropy change, A,S, is the difference in entropy between two states of a system, such as between the reactants and products of a reaction or between a flask before and after adding a mixture of gas. O At zero kelvin, substances maximize entropy. O At absolute zero, the interatomic distance within a crystal is minimized. O In a crystalline state, there is a tendency to minimize entropy. O In a gaseous state, the entropy of the system is zero. O As the temperature of a perfect crystal increases, the random vibrations of the molecules decrease. O At the melting point, the entropy of a system increases abruptiy as the compound transforms into a liquid. Submit Part B Calculate the standard-state entropy for the following reaction: 2NIS(s) + 302(g)→2S02 (9) + 2N10(s) The standard entropy values are given in the table. Formula IK-i mol SO2 (9) NiO(s) 248 38.0 NiS(s) 53.0 O2(9) 205 Express your answer with the appropriate units. > View Available Hint(s) Tempjetes Symbols undo redo Tesat keyboard shortcuts Help A,s° = Value Units
Entropy, denoted by the symbol S, is the thermodynamic property describing the amount of molecular randomness or disorder in a system. It is a state thermodynamic function, meaning that its value does not depend on the path used to arrive at a specific set of conditions. Each system with a specified temperature, pressure, and composition has a unique entropy value. The third law of thermodynamics relates the entropy (randomness) of matter to its absolute temperature. Which of the following statements are in agreement with the third law of thermodynamics? Select all that apply. • View Available Hint(s) The entropy change, A,S, is the difference in entropy between two states of a system, such as between the reactants and products of a reaction or between a flask before and after adding a mixture of gas. O At zero kelvin, substances maximize entropy. O At absolute zero, the interatomic distance within a crystal is minimized. O In a crystalline state, there is a tendency to minimize entropy. O In a gaseous state, the entropy of the system is zero. O As the temperature of a perfect crystal increases, the random vibrations of the molecules decrease. O At the melting point, the entropy of a system increases abruptiy as the compound transforms into a liquid. Submit Part B Calculate the standard-state entropy for the following reaction: 2NIS(s) + 302(g)→2S02 (9) + 2N10(s) The standard entropy values are given in the table. Formula IK-i mol SO2 (9) NiO(s) 248 38.0 NiS(s) 53.0 O2(9) 205 Express your answer with the appropriate units. > View Available Hint(s) Tempjetes Symbols undo redo Tesat keyboard shortcuts Help A,s° = Value Units
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter16: Thermodynamics: Directionality Of Chemical Reactions
Section16.3: Measuring Dispersal Of Energy: Entropy
Problem 16.3CE
Related questions
Question
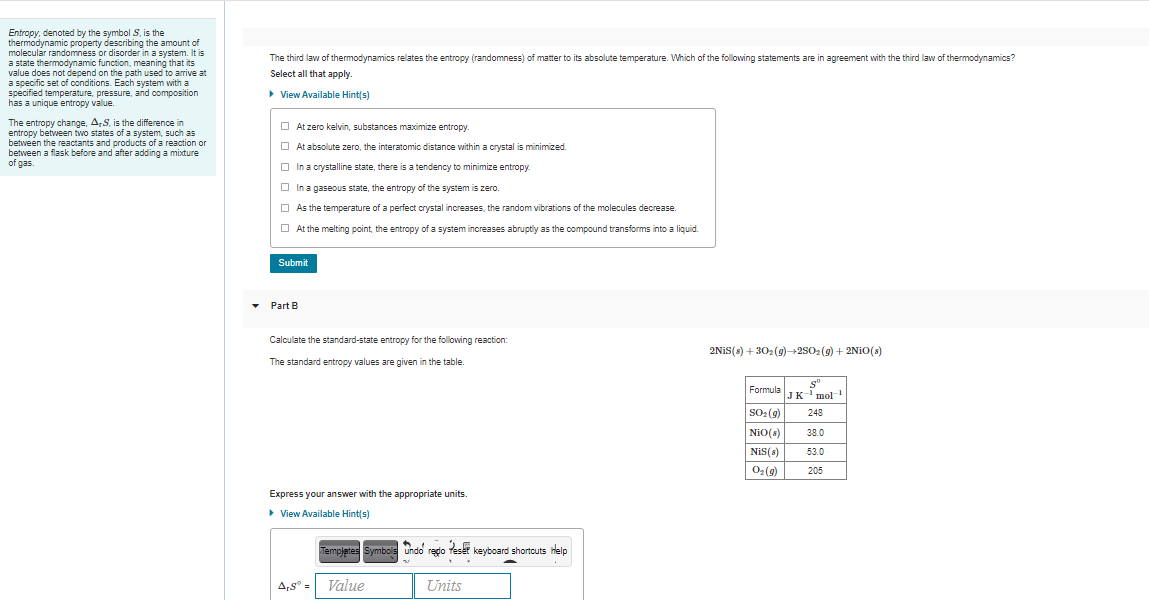
Transcribed Image Text:Entropy, denoted by the symbol S, is the
thermodynamic property describing the amount of
molecular randomness or disorder in a system. It is
a state thermodynamic function, meaning that its
value does not depend on the path used to arrive at
a specific set of conditions. Each system with a
specified temperature, pressure, and composition
has a unique entropy value.
The third law of thermodynamics relates the entropy (randomness) of matter to its absolute temperature. Which of the following statements are in agreement with the third law of thermodynamics?
Select all that apply.
• View Available Hint(s)
The entropy change, A,S, is the difference in
entropy between two states of a system, such as
between the reactants and products of a reaction or
between a flask before and after adding a mixture
of gas.
O At zero kelvin, substances maximize entropy.
O At absolute zero, the interatomic distance within a crystal is minimized.
O In a crystalline state, there is a tendency to minimize entropy.
O In a gaseous state, the entropy of the system is zero.
O As the temperature of a perfect crystal increases, the random vibrations of the molecules decrease.
O At the melting point, the entropy of a system increases abruptiy as the compound transforms into a liquid.
Submit
Part B
Calculate the standard-state entropy for the following reaction:
2NIS(s) + 302(g)→2S02 (9) + 2N10(s)
The standard entropy values are given in the table.
Formula IK-i mol
SO2 (9)
NiO(s)
248
38.0
NiS(s)
53.0
O2 (9)
205
Express your answer with the appropriate units.
> View Available Hint(s)
Templates Symbols Undo redo teset keyboard shortcuts lelp
A,s° =
Value
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax