Estimate (a) the maximum, and (b) the minimum thermal conductivity values (in W/m-K) for a cermet that contains 82 vol% carbide particles in a metal matrix. Assume thermal conductivities of 27 and 67 W/m-K for the carbide and metal, respectively. (a) W/m-K (b) W/m-K
Estimate (a) the maximum, and (b) the minimum thermal conductivity values (in W/m-K) for a cermet that contains 82 vol% carbide particles in a metal matrix. Assume thermal conductivities of 27 and 67 W/m-K for the carbide and metal, respectively. (a) W/m-K (b) W/m-K
Chapter23: Voltammetry
Section: Chapter Questions
Problem 23.14QAP
Related questions
Question
100%
V5
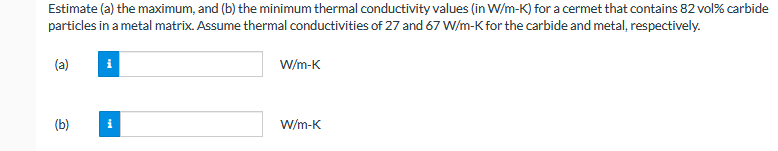
Transcribed Image Text:Estimate (a) the maximum, and (b) the minimum thermal conductivity values (in W/m-K) for a cermet that contains 82 vol% carbide
particles in a metal matrix. Assume thermal conductivities of 27 and 67 W/m-K for the carbide and metal, respectively.
(a)
W/m-K
(b)
W/m-K
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning