Find the A and T% of a 6.5X10-4 mol L-1 solution of a drug (Molecular weight 200) whose absorptivity is 0.026 L cm1 g'at 520 nm when measured in a cell of 2.00 cm pathlength?
Q: 1. What is the molar concentration of a sulfuric acid (H,SO.) solution with 7.3g/dm' concentration?
A: Given, Concentration of the solution = 7.3 g/dm3 Molar concentration of sulphuric acid solution = ?…
Q: The absorbance of a solution of copper nitrate is measured using UV?Vis spectrophotometry? What will…
A:
Q: he absorption spectrum of [Ni(NH3)6]2+ in water shows broad absorption with max 370, 625, and 1000…
A:
Q: 0.3872g of an unknown weak acid with general formula HY, was dissolved in water and made up to a…
A: Since HY has 1 replaceable H, it is a monoprotic acid. Equation for the association of HY is: HY(aq)…
Q: How does the pKe of the conjugate acid of benzylamine (CgH5CH,NH2) compare to the pk,'s of the…
A: pKa is inversely proportional to acidity strength. A more acidic molecule will have lower pKa value.…
Q: Given that Kb For ammonia is 1.8 x 105 and that For hydroxylamine is 1.1 x1D-8 A.What is the ha…
A: Ka * Kb is equivalent to adding the acid and base reactions together, which results in a net…
Q: Protein A binds to molecule Z with a Ka of 15.0 x 109. If the concentration of molecule Z is 30.0…
A: For a protein that binds to a ligand (Z), the fraction of sites occupied (θ) on the protein is…
Q: Why is it easier to see all three pKa values in a tripolic acid but not all 3 equivalence points?
A:
Q: A solution of Tryptophan has an absorbance at 280 nm of 0.25 in a 0.5 cm length cuvette. Given the…
A:
Q: Propranolol binds to beta-adrenergic receptors with a Kj of 8.9 x 10-9M. What concentration of…
A:
Q: calculate the dilution required to analyse the concentration of paracetamol in a commercially…
A: Molar extinction coefficient (ε) of Acetaminophen at 250 nm = 14 mM-1cm-1…
Q: Find the weight factor of iodide I in, Hg(IO2)
A: The amount of quantity of matter present in the substance is explained by the term mass. It is…
Q: An unknown chemical has an absorbance of 0.265 in a 1.0cm cuvette at a concentration of 0.703 M.…
A: From given data A = 0.265 l = 1 cm c = 0.703 M Formula used A= ԑcl Where, ԑ is absorptivity…
Q: A stock solution of alcohol dehydrogenase (ADH) has been diluted 1 in 10 and the absorbance reading…
A: Absorption coefficient = 1.6 ml/mg/cm Absorbance (A) = 0.04 Dilution = 10 times Complete solution…
Q: Q1: 10.00 gram of Ca3(PO4)2.7H2O was dissolved in 2.0L calculate the following: 1- рCа 2- wt/vol% P…
A: 1. Given that - Mass of Ca3(PO4)2.7H2O = 10.00 g Volume of Solution = 2.0 L = 2000 mL (1) Now,…
Q: A sterile ophthalmic suspension contains 0.5% timolol maleate (m.w. 433). How many milligrams of the…
A: We have 0.5% w/v opthalmic solution. Solute here is timolol maleate. Mol mass of timolol maleate =…
Q: Calcula HNO2 ( A typic (acetyls Ka = 3. %3D 300ML the per
A: Concept pH of the solution can be calculated as: pH= - log [H+]
Q: 1. At the end of a reaction, I ended up with a 200 mL aqueous solution containing 20 g of…
A: To solve these problems, we will be using the following equations: KD=XorgVorgXaqVaq .. (1) Here, KD…
Q: 49. Which one of the following is the required amount of Benzalkonium Chloride Solution BP to…
A: According to the company guidelines, we have to answer only the first question. So, please kindly…
Q: what is the CFU per
A: Y=mx+c where Y is absorbance and X is the concentration and C is intercept made by line on the…
Q: - How many grams of sodium hydroxide (M.wt=58.5) can be added to 2.5 liter of a solution mixture of…
A: Given a buffer solution consisting of mixture of 0.3M ammonia and 0.15M ammonium chloride.
Q: 3. Why did a mixture of 2.00 mL of pH 7.00 phosphate buffer, 0.900 mL of deionized water and 0.100…
A: 3. p-nitrophenyl acetate? in solution yields a light yellow to yellow solution?. This has a…
Q: 1. C3HO2 a Proton-decoupled CDCI3 (solvent) 200 150 100 50
A:
Q: 1)What is the concentration of the final diluted solution in mcg/mL?(1 decimal) 2) What volume of…
A: Mass of paracetamol = 140.1 mg Volume of solution = 200 mL Concentration = 140.1…
Q: what is the purpose of TLC Separation & Analysis of Analgesics?
A: A mixture is a type of matter which is composed of two or more substances. These are called…
Q: In 250 ml ethanol solution, a tablet of saccharin was dissolved, and it was measured by UV…
A: Beer's law states that the absorbance is directly proportional to the concentration of the absorbing…
Q: 5. (1) (ii) Ibuprofen (206.3 g/mol) absorbs at 255 nm and shows a molar absorptivity of 3010…
A:
Q: SPECTROPHOTOME TRY The DPD colorimetric method is used to determine the amount of residual chlorine…
A: Note - Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: 9. The following molecular tweezers bind 2,4,5,7-tetranitrofluorenone (TENF). The association…
A: A tweezer is a chemical structure that have two or more binding sites and which converge on a single…
Q: Explain (2 max :375 nm) absorbs at a longer wavelength than phitrobenzene (^-max : 260 nm). why…
A: Wavelength maximum value depends on the resonance. Greater is the resonance, higher is the…
Q: A triamcinolone acetonide topical aerosol spray contains 0.147 mg of triamcinolone acetonide in each…
A: We have to predict the percentage strength of triamcinolone acetonide.
Q: MW (mg/mmol) Density (mg/uL) Amount used vanillin 152.15 XXXXX 400.0 mg acetic anhydride 102.09 1.08…
A: Number of millimoles=Amount/MW Amount of acetic anhydride=500 mg (since, 1ul=1mg) MW of acetic…
Q: Acetanilide 14,3°C compound identity literature m.p. of compound experimental m.p. of your impure…
A: Solution- 1a) Let us consider the given data. The compound identity = Acetanilide The literature…
Q: HBr HCI NaOH a Br2 you OH /mol) of reections 1 anhn ata, ask ma and send ond b HO. H2SO4 Br O2 H2SO4…
A:
Q: Cuivan Coreeak base C₂H₂0 Nuo ₂. a 2. kb = 3.98 x 10-11 trund the PM of a Solution prefered by…
A: Here we are required to find the pH of weak base solution.
Q: 8) Rank the following compounds in order from lowest to highest retention time in GC (using a OV-101…
A: Gas chromatography (GC) is an analytical instrumental method that is used for the…
Q: A 5.00-mL aliquot of a solution that contains 3.05 ppm Ni2+ is treated with an appropriate excess of…
A:
Q: 0.122 mg of Crystal Violet you synthesized was dissolved in 100 ml of solvent. The absorbance was…
A: The expression of the absorbance is shown below: A = logI0I where; A = absorbance I0 = initial…
Q: Effect of analyte polarity on the Rf of an analyte on silica TLC. a) Which is more polar, benzoic…
A:
Q: Zn, heat 7- Succinimide
A: Succinimide is an organic compound that possesses the chemical formula of C4H5NO2. It is white in…
Q: After the above reaction was completed, you obtain the following GC results. You observe 3 peaks,…
A: To identify the substance corresponding to the peak observed at a retention time of 3.25 min.
Q: a. Pb2* ion Probable Observation Treatment After heating After adding K2CrO4 b. Ag* ion Treatment…
A:
Q: 2. Compound A has a molar absorptivity of 1.025 x 103 L cm-1 mol-1 at 280nm and a molar absorptivity…
A: We are given molar absorptivities of compound-A and B at two different wavelengths, 280 nm and 360…
Q: A sample of Al2(SO4); weighing 7.52-g was dissolved in enough water and diluted to 250.0-mL.…
A: Given: Mass of sample = 7.52 g. The sample is diluted to 250 mL. Volume of diluted sample used =…
Q: Identify the parent and propose a structure for the base peak in the massspectrum of butan-1-amine.
A: Mass spectrometry is used to detect the mass-to-charge ratio of the molecular ion. A plot of…
Q: An unknown chemical has an absorbance of 0.884 in a 1.0cm cuvette at a concentration of 1.668 M.…
A: The given data contains, Absorbance = 0.884 Length of cuvette = 1 cm Concentration = 1.668 M.
Q: 1) Oz then DMS 2) LIAIH, (excess) then H20 3) NaH (1 equiv.) 4) TSCI (1 equiv.) 5) NaOH (dilute)…
A:
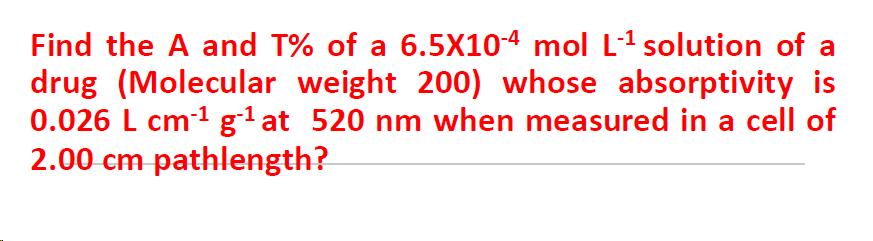
Step by step
Solved in 2 steps with 2 images

- EI-MS of ortho nitrotoluene shows a large fragment ion at m/z 120 .The EI-MS of Tri deuteron ortho nitrotoluene does not show any peak at m/z 120 but does have a peak at m/z 122 .Show the fragmentation pattern that accounts for the above observation.Explain the order of chemical shifts of the carbon atoms in the 13C spectra of cyclohexanone (fig 22.6 ) and adipic acid (fig .22.9) attached below .Treatment of butan-2-one (CH3COCH2CH3) with strong base followed byCH3I forms a compound Q, which gives a molecular ion in its massspectrum at 86. The IR (> 1500 cm−1 only) and 1H NMR spectra of Q aregiven below. What is the structure of Q?
- All methyl esters of long-chain aliphatic acids (e.g., methyl tetradecanoate, C13H27COOCH3) show significant fragment ions at m/z 74, 59, and 31. What are the structures of these ions? How are they formed?A 4.0 x 10-5 M solution of a compound in hexane shows an absorbance of 0.40 at 252 nm in a cell with a 1 cm light path. What is the molar absorptivity of the compound in hexane at 252 nm?The leaves of the Brazilian Tree Senna multijunga contain a number of pryidine alkaloids that inhibit acetylcholinterinase. Two recentyl isolated isomeric compounds have the strcture have the strcture shown below. (NOTE: M=293) Use the mass spectral data provided to determine the precise location of the hydroxyl group in each isomer. Isomer A: EI-MS, m/z(rel. int): 222(20), 150(10), 136(25), 123(100) Isomer B:EI-MS, m/z(re;. int): 236(20), 150(10), 136(25), 123(100)
- A student completed a similar lab experiment and got an εavg of 56 M-1cm-1. If their change in absorbance (∆A) for the first 1.5 minutes was 1.089 and the cuvette was 1.0 cm wide, what was the rate for the first 1.5 minutes? Give your answer to the nearest 0.001 M/min.Why is it better to acquire the 1H-NMR spectrum in DMSO-d6 and not in CDCl3?A solution was prepared using 1.0 mg of a compound of Molecular weight 160 g / mol isdissolved in 10 mL of Ethanol and part of the solution is placed in a 1 cm UV cell. TheUV spectrum is taken at a (I: 247 nm). The maximum absorbance is 0.5. Determine the value of molar absorptivity (c) in M ^ -1cm ^ -1
- Whta is the Difference of infrared spectrum for the starting material and product?which one can be use to determine successfully made product? The starting mat.=0.819g Mass of prd=1.06g melting point range=83-85The molar absorptivity of 2,5-dimethyl-2,4-hexadiene in methanol is 13,100 M-1cm-1. What concentration of this diene in methanol is required to give an absorbance of 1.6? Assume a light path of 1.00 cm. Calculate concentration in these units. Q.) Moles per liter(a) What is the molar absorptivity of compound Z at 295 nm and 348nm, given the absorption spectrum shown in Fig.22-14 (which was obtained using a UV-Vis spectrophotometer and a 1mM solution of compound Z in a sample cell with a path length of 1 cm)? (b) Now you have decided to make quantitative measurement of the level of compound Z in different solutions. Based on the above spectrum, which wavelength will you use for your measurements? Give two reasons why this is the optimum wavelength.



