Explain why structure 4 is a more potent inhibitor (lower IC50 value) than inhibitor 3 and why structure 5 is a much weaker inhibitor (higher IC50 value) than 3 and 4.
Explain why structure 4 is a more potent inhibitor (lower IC50 value) than inhibitor 3 and why structure 5 is a much weaker inhibitor (higher IC50 value) than 3 and 4.
Chapter9: Parenteral Medication Labels And Dosage Calculation
Section: Chapter Questions
Problem 6.4P
Related questions
Question
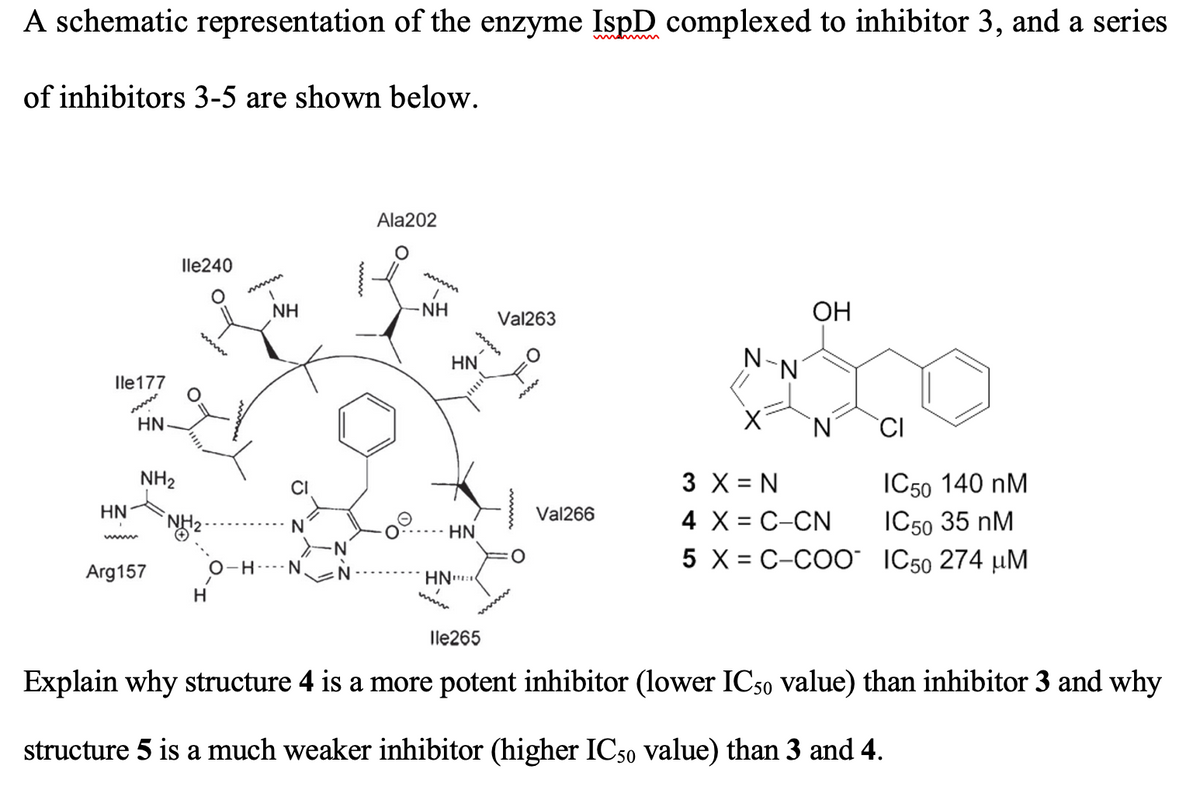
Transcribed Image Text:A schematic representation of the enzyme IspD complexed to inhibitor 3, and a series
of inhibitors 3-5 are shown below.
Ala202
lle240
mwww
NH
NH
Val263
ОН
www
HN
N-
lle177
HN
'N'
CI
3 X = N
4 X = C-CN
5 X = C-COO IC50 274 µM
IC50 140 nM
IC50 35 nM
NH2
HN
Val266
N
-N
O-H---- N
HN
%3D
Arg157
HN
wwww
lle265
Explain why structure 4 is a more potent inhibitor (lower IC50 value) than inhibitor 3 and why
structure 5 is a much weaker inhibitor (higher IC50 value) than 3 and 4.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

