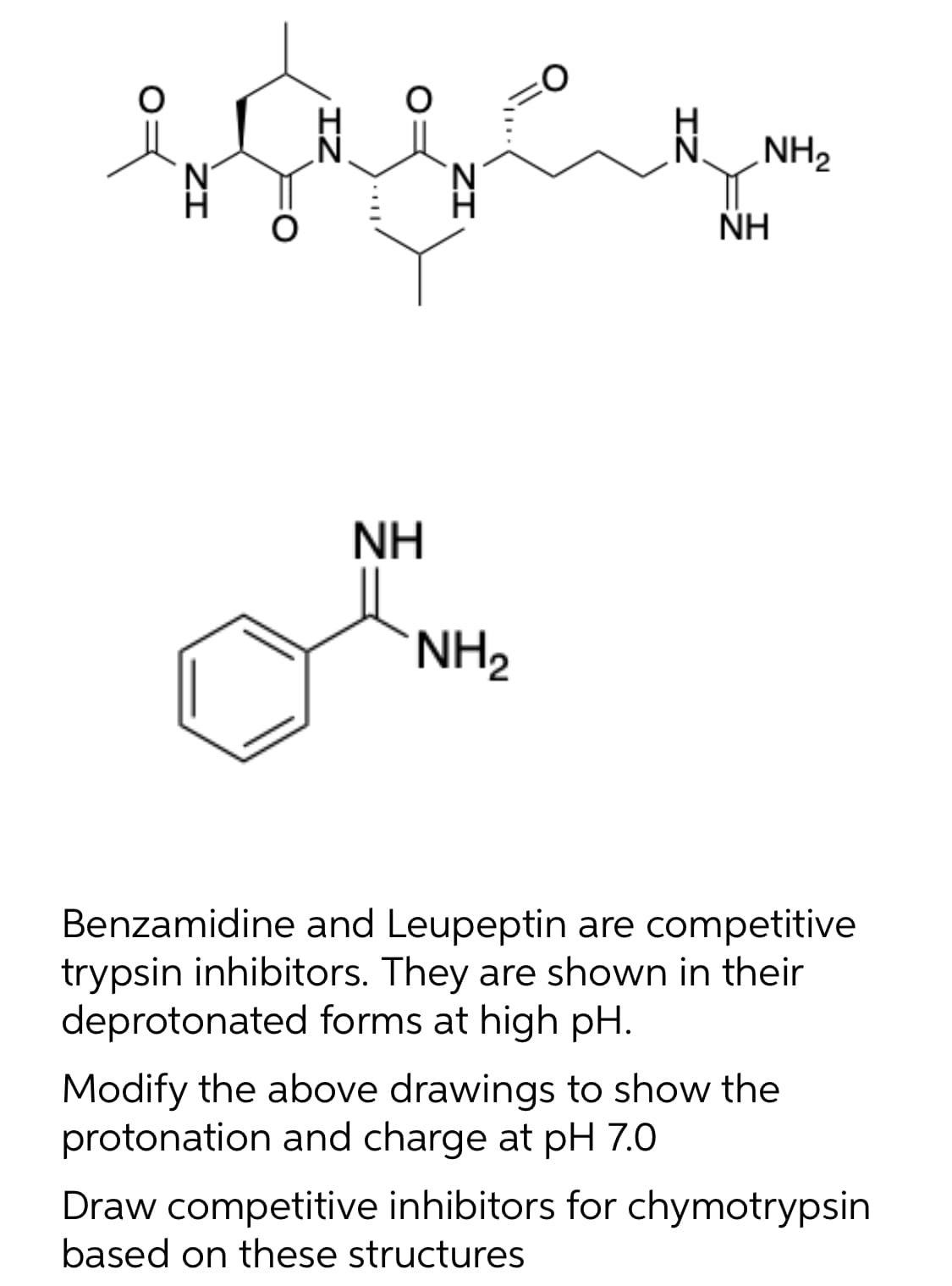
NH, N. NH NH NH2 Benzamidine and Leupeptin are competitive trypsin inhibitors. They are shown in their deprotonated forms at high pH. Modify the above drawings to show the protonation and charge at pH 7.0 Draw competitive inhibitors for chymotrypsin based on these structures ZI
Q: Phosphocreatine (G0ʹ = -43.1 kJ/mol) has a higher phosphoryl group transfer potential than ATP…
A: Hi! Since you have posted multiple questions and have not mentioned which to answer, we shall answer…
Q: Drug Metabolism Reactions Identify the enzyme family involved in the following metabolic reaction:…
A: Drug metabolism is the process in which a substance of therapeutic nature is converted to some other…
Q: rounds of beta-oxidation are needed to oxidize the fatty acid
A:
Q: The KMof the enzyme for the substrate adenosine is 3 × 10ꟷ5M. The product inosine acts as an…
A: Given values in the question: Km of the enzyme for the substrate adenosine = 3 × 10-5 MKi of the…
Q: Relationship between structure and biological activity 1. Draw the component of the opioid analgesic…
A: The term opioid analgesics refers to medications that act on opioid receptors and are used for pain…
Q: Gemcitabine is a deoxycytidine analog used in treating pancreatic and ovarian cancer. The drug…
A: Gemcitabine It is a chemotherapy medicine used in the treatment of various cancers. It is a…
Q: Think about what part of chymotrypsin, according to our coverage, most contributes to stabilization…
A: Chymotrypsin is an enzyme that is present in the digestive system of many organisms that is…
Q: 6) high abuse potential. Once in the body, amphetamine is metabolized to a series of metabolites.…
A: Drugs that are consumed are primarily metabolized from the liver. The study of metabolism, excretion…
Q: 1. Requirements of Active Sites in Enzymes The active site of an enzyme usually consists of a pocket…
A: Peptidases are enzymes that cleave peptide bonds and peptides. Carboxypeptidases are peptidases…
Q: 3. Trypsin selectively cleaves polypeptide chains on the carbonyl side of the amino acids arginine…
A: Enzymes are proteins that accelerate the rate of biochemical reactions. Proteins are polymers of…
Q: Calculate the number of ATPS produced from the complete oxidation of a TAG containing two caproic…
A:
Q: Phosphorylated compounds with a high hydrolysis rate have a high phosphoryl group transfer…
A: Because phosphorus is thermodynamically unstable and kinetically stable, it is an important molecule…
Q: Arachidonic acid, a 20-C saturated fatty acid. Use only 1 mole: 1. How many rounds of…
A: Beta oxidation is a catabolic process that occurs in both prokaryotes and eukaryotes in order to…
Q: The negative impact of PKU can be reduced by stimulation of alternative pathways of phenylalanine…
A: All the genes in the body are present in the DNA of the cell. these genes are a triplet codon of…
Q: xanthine oxidase and reduce the production of uric acid. You are a biochemist and just discovered a…
A: Human xanthine oxidase is an enzyme that catalyses the oxidation of hypoxanthine to xanthine and…
Q: Time Which is correct regarding the concept of the lock-and-key as a theory of enzyme action: O a.…
A: The formation of an enzyme-substrate complex is the first step in enzymatic catalysis. The enzyme…
Q: Riboflavin (ry-boh-FLAY-vin), also called vitamin Riboflavin (ry-boh-FLAY-vin), also called vitamin…
A: Riboflavin is also known as vitamin B2 is an important vitamin that is required for the overall…
Q: You are investigating inhibitors of chymotrypsin, and the two molecules shown on the right (A&B) are…
A: Lineweaver Burk Plot is generally helped to determine important terms in enzyme kinetics, such as Km…
Q: 7. kynureninase with an inhibitor, 3-hydroxyhippurate, bound in the active site. The structure is…
A: Inhibitors bind to the active site of Enzyme, reversibly or irreversibly. The active site forms…
Q: help fill in the word between brackets Serine proteases are an example of…
A: Serine protease are the enzyme which causes cleavage of peptide bonds in protein . They are called…
Q: competitive inhibitor of the enzyme sucrase slows the production of glucose and fructose in a…
A: Enzymes are the protein molecules which are of biological origins or sometimes may be produced…
Q: Trypsin contains a/an [aspartate/serine/histidine] in its specificity pocket to attract lysine and…
A: Trypsin is an enzyme involved in the cleavage of peptide bonds at specific sites. In the pancreas,…
Q: NH2 H. Download Image... One of the products released by the action of phospholipase A1 on the lipid…
A: Phospholipase A1(PLA1) hydrolyses phospholipids and produces 2-acyl-lysophospholipids and fatty…
Q: Fill in the blanks: is the type of enzyme specificity exhibited by hydrolysis of urea using urease…
A: Enzymes are molecules that help in the catalysis of reactions. Most of the reactions in our body are…
Q: Indicate the FALSE alternative: a) The formation of osazones is a characteristic reaction of…
A: Carbohydrates are carbon, hydrogen, and oxygen molecules with the formula (CH2O)n. Monomers are the…
Q: Phosphorylated molecules with high _____________ values of hydrolysis have large phosphoryl group…
A: Phosphorylated molecules can either donate their phosphate group to other molecules or can get…
Q: IX. Insulin, a hormone vital in blood sugar regulation and having a polypeptide chain with disulfide…
A: Insulin is a hormone responsible for the regulation of sugar levels in the blood. Insulin hormone is…
Q: Ethylene glycol (HO−CH2−CH2−OH) is a major component of antifreeze. In the body, it is first…
A: Catalyst is a chemical substance that increases the rate of a chemical reaction by lowering the…
Q: TRIGLYCERIDE: I. Formed through dehydration resulting in loss of water II. Formed through hydrolysis…
A: OXIDATION: I. Saturated fats are more susceptible II. Degree of oxidation can be measured by acid…
Q: peptide C=O bond. O The imidazole group acts as a general base and extracts a proton from serine…
A: Introduction Histidine is α -amino acid that is required for protein biosynthesis. It has an…
Q: 4. The reaction shown below is part of the glycolytic pathway. The standard free energy change for…
A: Cellular respiration is a catabolic pathway of the process of metabolism, where a series of chemical…
Q: Which statement is NOT completely true? O Cyanide, a poison, is an example of a reversible…
A: ans: Non-Competitive inhibitors bind to an allosteric site of the enzyme (A site on the enzyme…
Q: The hydrolysis of ATP has a large negative ΔG'°; nevertheless it is stable in solution due to: a.…
A: ∆G (free energy change) is the difference in free energy between reactants and products. The value…
Q: 11. This below notation represents a Bi-Bi reaction. Aspartate Oxaloacetate a-Ketoglutarate…
A: Introduction In ping pong reactions, the first substrate binds to the enzyme, transfers a piece of…
Q: How does enzyme inhibition occur? Give and illustrate the reaction between enzyme and substrate in…
A: Enzymes or Biocatalysts catalyze the chemical reaction in a biological system at a higher rate. They…
Q: The molecular structure of vitamin A is conjugated with N = 10. Calculatethe wavelength of light in…
A: Available data: N=10 c = 3×108m/sh = 6.626×10-34J/s2m = 9.11 × 10-31kgd = 3.05×10-10m
Q: Energy Energy 2. For both of the graphs below, (A) draw a new blue line showing what the reaction…
A: exergonic biochemical reactions are high energy reactions where the reactants have higher energy…
Q: catalytic function of C25
A: Protease is one of several enzymes that hydrolyze the proteins. They cut the peptide bond which…
Q: Saberate concentration dwys in escem Trypsin (digests profein in ntestine) Pepsm (digests profein in…
A: Trypsin and pepsin both are the enzymes of the digestive system which break protein chains in the…
Q: Why is the reaction rate low at pH7? Be specific and say something about the enzyme structure at the…
A: Reaction rate is often described as how fast the reaction takes place or proceeds. Enzymes are often…
Q: What's true about Purdue Pharma and OxyContin? (Select all that apply] O OxyContin is an opioid…
A: John Purdue Gray founded Purdue Pharma LP, a privately held pharmaceutical corporation. OxyContin's…
Q: Regarding the mechanism of action of chymotrypsin, the formation of a strong hydrogen bond with the…
A: Chymotrypsin performs a mechanism called covalent hydrolysis. Enzyme firstly form a covalent bond…
Q: 1. Requirements of Active Sites in Enzymes The active site of an enzyme usually consists of a pocket…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation energy…
Q: (a) Write down the reaction of phosphatidylcholine (PC) cleaved by PLA2. Suppose the two acyl groups…
A: Phospholipids are degraded by phospholipases that cleaves the phosphodiester bonds. Phosphatidyl…
Q: Describe the structure of the Insulin. Should include: The overall secondary structure of Insulin.…
A: The insulin is a protein hormone consisting of 2 polypeptide chain. Insulin is produced in the beta…
Q: Pheneticillin (below) is a B-lactamase sensitive penicillin. Draw the structure of the inactive form…
A: Penicillin is a group of antibiotics which kill the bacteria by inhibiting the synthesis of…
Q: tch the following descriptions to the given choices. Enzyme involved in the conversion of…
A: Prostaglandin are active substances, having hormone like functions. Found in human at 1935 in semen…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 4 images

- Salicylamides are inhibitors for an enzyme called scytalone dehydratase. SAR shows that there are three important hydrogen bonding interactions. Explain whether you think quinazolines could act as a bioisostere for salicylamides.Homopolysaccharide are found abundant in nature. Explain how this class ofpolysaccharide acts as primary component of plants and insects.The substitution of His 64 of carbonic anhydrase II with Ala results in a sharp decrease in the activity of the enzyme in HEPES buffer (molecular weight of HEPES = 238.3 g/mol). However, increasing concentrations of imidazole (molecular weight = 68.1 g/mol) restores the reaction rate close to that of the wild-type enzyme. Propose an explanation for these results.
- Chymotrypsin information about enzyme? Trivial/Common Name Systematic (EC) name and number Chemical reaction catalyzed Metabolic pathway Function and cellular localization Size (kDa) and structure (tertiary and quaternary) pH stability Kinetic properties (Km- and Kcat value/s for natural substrates) Stability and potential inhibitorsCarboxylic acids, such as the one shown below, can be difficult to reduce Provide the name of a reductant A. Explain why this reductant is able to react with the carboxylic acid whereas others cannot (Hint: Draw and discuss the potential reactive intermediates/transition states).MULTIPLE CHOICE In patients with Lesch Nyhan Syndrome, purine nucleotides are overproduced and over excreted. The hypoxanthine analogue Allopurinol, which effectively treats gout , has no effect on the severe neurological symptoms of Lesch- Nyhan patients because it does not A. decrease de novo pyrimidine synthesis B. decrease de novo purine synthesis C. decrease urate synthesis D. increase PRPP levels (Phosphoribosyl pyrophosphate)
- A competitive inhibitor interacts with the free enzyme to form an enzyme•inhibitor complex(E•I). This equilibrium reaction can be described as follows:E + I ⇌ EIModify the simplified kinetic scheme for the reaction E + S ⇌ E + P to include this equilibriumexpressionThere are parts A-C for this picture included. A) What type of enzyme is Malate Dehydrogenase? choices: Hydrolase, Isomerase, Ligase, Oxideoreductase, Transferase, or Translocase B) Which of the following statements are true in biochemical standard conditions? There can be more than 1. Choices: The reaction is spontaneous since ∆G°' is positive The reaction is spontaneous since ∆G°' is negative The reaction is not spontaneous since ∆G°' is positive The reaction is not spontaneous since ∆G°' is positive The equilibrium favors products since K is greater than 1 The equilibrium favors reactants since K is greater than 1 The equilibrium favors products since K is less than 1 The equilibrium favors reactants since K is less than 1 The reaction is always at equilibrium C) If the concentration of Oxaloacetate is 10^7 times lower than the concentration of Malate D.,Is the reaction Spontanuous? Choices: No, because RTInQ is very positive Yes, because RTlnQ is very…The KMof the enzyme for the substrate adenosine is 3 × 10ꟷ5M. The product inosine acts as an inhibitor of the reaction, with an inhibition constant (KI, the dissociation constant for enzyme-inhibitor binding) of 3 × 10ꟷ4M. However, a transition state analog,Inhibits the reaction with KIof 1.5 × 10ꟷ13M. Explain why 1,6-dihydroinosine serves as a better inhibitor of adenosine deaminase than inosine. Elaborate on your answe
- The following phenylboronate is a competitive inhibitor of a chymotrypsin-like enzyme. Unlike carbon, boron is quite happy to be bonded to three oxygens (and one carbon)—explain why this feature makes it a much better inhibitor than the corresponding carboxylic acid (which is quite ineffective).RuBP carboxylase is not an idel enzyme by any means. Describe some of the active site's and substrate specificity's issues. When the amino acid sequences of this enzyme from several species are compared, they are nearly similar. What importance does this homogeneity have?The pKa for histidine is pKa = 6.1 while that for cysteine is pKa = 8.0 2. Assume that both histidine and cysteine are catalytic groups for a particular enzyme. Assume also that the side chain of cysteine must be in the deprotonated form. Estimate the pH at which the catalytic activity of this enzyme is the maximum and sketch a pH-activity graph.

