Q: What is the solubility of SrF2 in g/L if its Ksp is 1.5 x 10-9 ? (MM = 125.6 g/mol) 0.019…
A:
Q: How many silicon atoms are present in the following compound? (Please answer using whole numbers.)…
A:
Q: 2 NH3 + 3 CuO → 3 Cu + N₂ + 3 H₂O In the above equation, how many grams of N₂ can be made when 10.9…
A:
Q: What will happen the the Lf if the calorimeter used was twice as large as the one used in the…
A: Answer: Lf mentioned over here is the enthalpy of fusion, it is equal to the amount of heat required…
Q: a pair of chemical compounds A and B can react in two different ways: A+B C Reaction 1 gives product…
A:
Q: A 250 mL glass of orange juice contains 22 grams of sugar. How much sugar is in a two-liter (2,000…
A:
Q: A metallic object to be plated with copper is placed in a solution of CuSO.. What mass of copper…
A: Given, Current, I = 0.22 amp Time, t = 1.5 hours = 5400 sec Molar mass of Cu =…
Q: MF C₂H₂NO Relative Intensity 100- 80 40 20 wwww.ppt=upppluut.yupw0 25 50 75 m/z 100 125 15.0 18.0…
A:
Q: Precipitation of Salts - == Net Ionic Equations Determine net ionic equations, if any, occuring when…
A: Here we have to write the net ionic equation and predict the true or false statements about the…
Q: Given the reaction: Fe +2+ Cr₂O7-2 Fe+3+ Cr+3 → How many mole/s of H₂O molecules is/are uproduced in…
A: The unbalanced redox reaction is: Fe2++Cr2O72-→Fe3++Cr3+
Q: How many total atoms are present in the following compound? (Please answer using whole numbers.)…
A:
Q: The epimerization of D-glucose to D-mannose forms an enolate ion with a double bond between which of…
A:
Q: Which one of the following is NOT a possible completion of the following reaction? CH₂OH HO- HO- 1.…
A:
Q: 13. The pka of an acid is 2.35. What is the acid-dissociation constant of the acid? O 4.5 x 10- O…
A: We can solve the Given problem by recalling the following equation Pka = -logka Where ka =…
Q: S + 6 HNO3 --> H2SO4 + 6 NO2 + 2 H2O In the above equation how many moles of water…
A:
Q: Indicate which atoms in the following compound are chiral centers. CHO H-C-OH H-C-OH d CH₂OH…
A:
Q: 39. A monodentate ligand can occupy how many sites in a coordination sphere of a metal ion? O 4 3 02…
A: Answer the following questions. Questions Nos. 39-40 For the correct option--
Q: On the product frame on the left, the blue molecules adopt the shape of the container, and the red…
A: The shape of the liquid phase is indefinite but the volume of the liquid phase is definite. For the…
Q: For the reaction C + 2H2 → CH4, how many moles of hydrogen are needed to make 103.9 grams of…
A: In chemistry, one mole can be defined as quantity containing particles equivalent to 6.023 × 1023…
Q: 49. The KCIO3 (122.55 g/mol) in a 0.1862-g sample of an explosive was determined by reaction with…
A:
Q: Consider the reaction below. Which is the oxidizing agent? * Fe + O₂ + H+ Fe+2+ H₂O. A. Fe B. 02 C.…
A:
Q: II. Identify the oxidizing agent and reducing agent in each reaction. 1. 2H₂ + O2 → 2H₂O 2. Zn+S →…
A: Here we are required to identify the oxidizing and reducing agent.
Q: What is the mechanism for reaction 1? a electrophilic addition b nucleophilic addition c…
A: #Q.1: In an addition reaction, an electrophile or nucleophile is added. In a substitution reaction…
Q: A particular reaction is found to be first order. The reactant has an initial concentration of 1.71…
A: For first-order reaction, the equation ln[A] = -kt + ln[A]0 depicts a straight line (y = mx + c)…
Q: Interaction / Compound kJ/mol kcal/mol HH eclipsing 4.0 1.0 H: CH3 eclipsing 5.8 1.4 CH3 CH3…
A: When angle between CH3 and CH3 is 60° then interaction is called butane gauche interaction
Q: Write the correct answer with 1 place after the decimal point. What is the molecular mass of…
A: What is the molecular mass of C10(N1O1]3?
Q: 3 Cu + 8HNO3 --> 3 Cu(NO3)2 + 2 NO + 4 H2O In the above equation how many moles of…
A:
Q: concentration
A: The ion-exchange membrane is a semi-permeable membrane which transports certain dissolved ions,…
Q: Question 14 Indicate all the types of intermolecular forces of attraction in CF4(1). Answer 1 of the…
A: The correct answer is given below
Q: 93. What are the electron-pair geometry and the molecular structure of each of the following…
A: Electron pair geometry and the molecular struture of each of the following molecule.
Q: Give the IUPAC name of the following molecule: OH 6 Give the IUPAC name of the following molecule:…
A:
Q: Question Four a) The concentration of As(III) in water can be determined by differential pulse…
A: Few questions based on general chemistry that is to be accomplished.
Q: Which of the following can form a carbinolamine intermediate with ammonia? a Bromoethane b…
A:
Q: Consider a 0.050 M succinate buffer at pH5.0. Draw the ionization reactions for succinic acid,…
A: Given the pH of the buffer solution = 5.0 The concentration of both the constituents of the buffer…
Q: 2 NH3 + 3 CuO → 3 Cu + N₂ + 3 H₂O In the above equation, how many grams of N₂ can be made when 6.1…
A:
Q: Without using a pK₂ table, predict the position of equilibrium for the given reaction, and explain…
A: In the given question aniline and ammonium ion given . The aniline is aromatic in nature and behave…
Q: Write the correct answer with 1 place after the decimal point. What is the molecular mass of…
A: The molecular mass of a compound can be obtained by addition of atomic mass of each element present…
Q: 3. This produces the observable physical change upon reaching the enc point of a titration indicator…
A: A point marking the completion of a reaction called end point
Q: 2. Calculate K for the following reaction given the following equilibrium concentrations of PCI5,…
A: Equilibrium constant (K) is the ratio of the equilibrium concentrations of product over equilibrium…
Q: 5- Ethanol C₂H,OH is burned with dry air for one mole of fuel. Find the air-fuel ratio, The…
A:
Q: 27. When mixing Rb₂CO3 with CaCl₂ aqueous solutions, what is the identity of the precipitate? RbCl…
A: To write the reaction and to check which is soluble or insoluble.
Q: pH=2.45 Your answer pH 8.28 Your answer pH=7.36 Your answer pH= 1.25
A:
Q: A. Give the IUPAC name of the following unsaturated hydrocarbons. 1 2 t 3 H₂C. had 4 CH₂ CH₂
A: We have find out the IUPAC nomenclature of unsaturated compounds. Note -as per our company…
Q: The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an…
A:
Q: 3 Cu + 8HNO3 g 3 Cu(NO3)2 + 2 NO + 4 H2O In the above equation how many moles of NO…
A:
Q: Suppose 220. mmol of electrons must be transported from one side of an electrochemical cell to…
A: Given mmol of electrons = 220 Time = 214 s We have to calculate current
Q: A 244.5-g sample of ground water is analyzed for calcium. The Ca2+ in the sample is first…
A:
Q: 48. How much 0.0500 M EDTA is needed to titrate the Mg in a 0.1135-g of hydromagnesite, 3MgCO,…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: An industrial chemist introduces 2.0 atm of H₂ and 2.0 atm of CO₂ into a 4.00-L container at 25.0 °C…
A: The equilibrium constant of a reaction is the ratio between the product's concentration and the…
Q: P What is the name of the carbohydrate compound C? HO CH₂OH *** C A. D-xylose B. D-lyxose C.…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

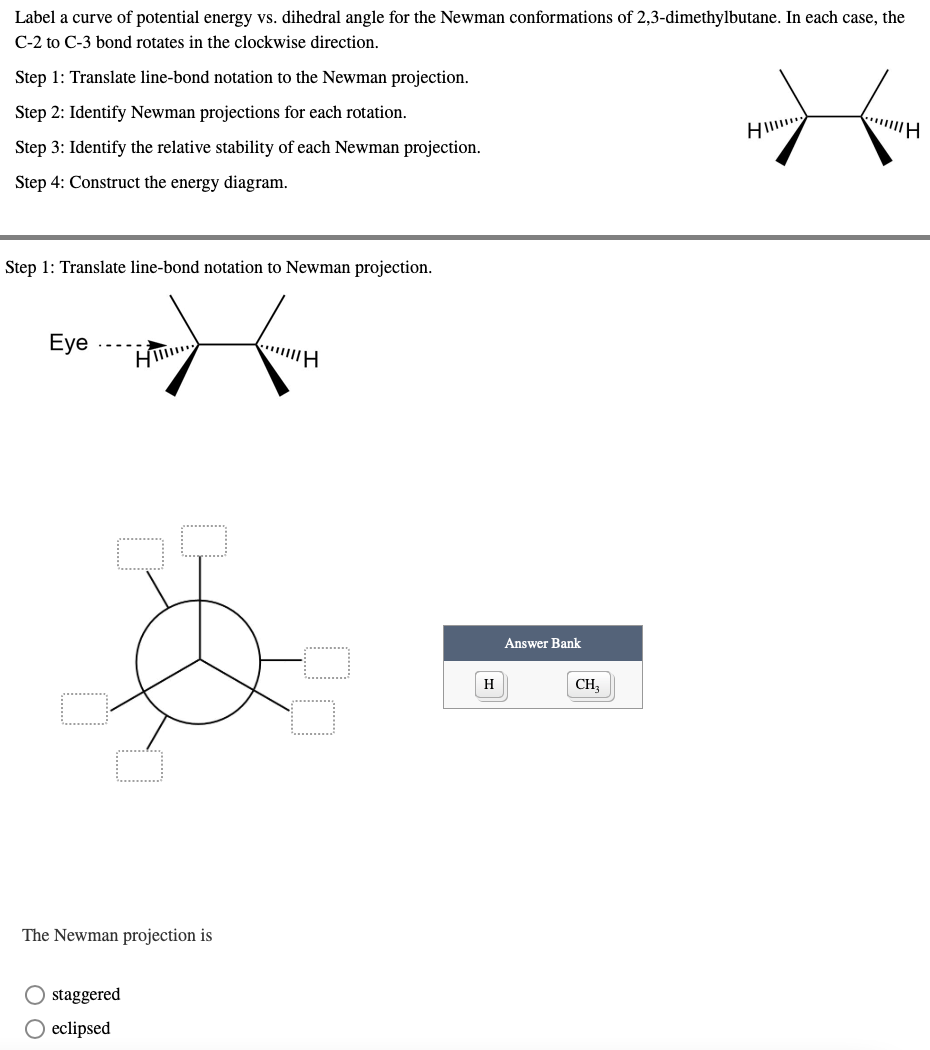
- a) Sighting down the C3-C4 bond, draw the gauche (60 degrees) and anti (180 degrees) Newman projections of 2,4-dimethylhexane. b) Circle the conformation that you drew that is lower energy.1. Estimate the relative stabilities of the 4 conformationseclipsed and eclipsed n-pentane by drawing the corresponding Newman projections in descending order according to their stability. Place the most stable or lowest energy on the left. 2. Explain what the difference in energy between the most stable and the least stable is due to. 3. What is the difference in stability between the two staggered conformations?For pentane draw Newman projections for the Syn-periplanar, conformation. the Anti- periplanar conformation and a Gauche conformation. Use C2 as the front carbon and C3 as the back carbon. Label each conformation, circle the highest energy conformation andunderline the lowest energy conformation.
- Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of methylcyclohexane in the chair conformation: a. When the methyl group is in an axial or equatorial (circle one) position, the molecule is inits lowest potential energy conformation. b. Label one Newman projection above anti and the other gauche to describe the relationshipbetween the methyl group and C3 of the ring. c. In general, which is a lower PE conformation, anti or gauche? d. Explain how your answer to b and c provide an explanation for why it is more favorable fora large group to be in an equatorial than an axial position.For rotation about the C3-C4 bond of 2-chlorohexane: a) Draw a Newman projection showing the most stable conformation. b) Draw a Newman projection showing the least stable conformation.Pls do fast within 5 minutes and i will give like for sure Solution must be in typed form 1. The alcohol group is axial or equatorial. 2. The methyl group is axial or equatorial. 3. The substituents are trans or cis. 4. The given chair conformation is higher/equal/lower in energy than the flipped conformation (not provided). 5. When drawing a planar form of the cyclohexane given, the methyl group would be wedged or dashed.
- Consider 1-bromopropane, CH3CH2CH2Br. (a) Draw a Newman projection for the conformation in which CH3 and -Br are anti (dihedral angle 180°). (b) Draw Newman projections for the conformations in which - CH3 and -Br are gauche (dihedral angles 60° and 300°). (c) Which of these is the lowest energy conformation? (d) Which of these conformations, if any, are related by reflection?Using Newman projections, draw each of the following molecules it most stable staggered, least stable staggered, least stable eclipse and most stable eclipse conformations with respect to the bond indicated: (b) 4-isoproply-2,4,5-trimethylheptane, C4-C5(c) 6-isopropyl-2,5-dimethylnonane, C5-C6 please answer B and CSight along the C2-C1 bond of 2-methylpropane (isobutane).(a) Draw a Newman projection of the most stable conformation.(b) Draw a Newman projection of the least stable conformation.(c) Make a graph of energy versus angle of rotation around the C2-C1 bond.(d) Assign relative values to the maxima and minima in your graph, given that an H↔H eclipsing interaction costs 4.0 kJ/mol and an H↔CH3 eclipsing interaction costs 6.0 kJ/mol.
- Draw a qualatative potential energy diagram for rotation about the C3-C4 bond in 2-methylpentane. Show newman projections for all conformations located at the maximum and minimum points on the diagram.Draw a Newman projection, similar to Figure 3-25, down the C1¬C6 bond in the equatorial conformation of methylcyclohexane. Show that the equatorial methyl group is also anti to C5.Rank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Cl

