Q: What type of isomer are 1,1-dichloroethene, and Trans-1,2-dichloroethene?
A: Structure for 1,1-dichloroethene, and Trans-1,2-dichloroethene
Q: Bayer Extra Strength aspirin tablets each contain 500 milligrams of acetylsalicylic acid. How many t...
A: Given mass of 1 tablet of aspirin = 500 milligrams mass of 50 tablets of aspirin = 500 milligr...
Q: The vapor pressure of water is 23.76 mm Hg at 25 °C. A nonvolatile, nonelectrolyte that dissolves in...
A:
Q: A researcher studying the nutritional value of a new candy places a 5.00 g5.00 g sample of the candy...
A: Given: Temperature change = 2.95 ∘C = 2.95 K heat capacity of the calorimeter = 29.70 kJ⋅K−1 mass of...
Q: The specific heat of a certain type of metal is 0.128 J/(g⋅∘C).0.128 J/(g⋅∘C). What is the final tem...
A: q = mc∆T q is the amount of heat energy required m is the mass of substance c is the specific hea...
Q: Convert -46.2°C to Kelvin.
A:
Q: The hydrolysis of the sugar sucrose to the sugars glucose and fructose can be described as follows. ...
A:
Q: Analyze the spectra below and determine the structure of each compound from which the carbon NMR Is ...
A: Number of signals in 13C NMR = Number of sets of carbon in different electronic environment. Multip...
Q: A major component of gasoline is octane (CH18). When octane is burned in air, it chemically reacts w...
A: Balanced chemical equation: 2 C8H18 + 25 O2 -----> 18 H2O + 16 CO2 Form the above equation: Numbe...
Q: A 9.47 g sample of an aqueous solution of hydroiodic acid contains an unknown amount of the acid. If...
A:
Q: Br 1. KOt-Bu KBr HOt-Bu Br Aqueous acetone H20 +. 2. H3C HBr Br H OH optically active racemic a = Pr...
A:
Q: In the course of developing his model, Bohr arrived at the fol-lowing formula for the radius of the ...
A: The radius of orbit of electron could be calculated using Bohr's radius expression. The expression i...
Q: help please
A:
Q: Now a solution is prepared by dissolving solid FeCl3 ( molar mass = 162.2 g/mol)in water. If the mas...
A: Given that: Mass percentage = 26.0 % Density of solution = 1.25 g/mL...
Q: 11. Calculate the equilibrium concentration for Hae for the reaction (H2 + ace = HI) who se, Keg is ...
A:
Q: when the following molecular equation is balanced using the smallest possible integer coefficients t...
A: When chlorine reacts with water hydrogen chloride and chloric acid is formed. Chloric acid is very s...
Q: You wish to make a 0.395 M hydroiodic acid solution from a stock solution of 6.00 M hydroiodic acid....
A: Given stock solution of 6.00 M hydroiodic acid. 0.395 M hydroiodic acid solution and 50.0 mL of th...
Q: Using the table of standard formation enthalpies that you'll find under the ALEKS Data tab, calculat...
A: The given balanced chemical reaction is - 2NaOH (s) + CaCl2 (s) →Ca(OH)2 (s) + 2NaCl (s) The standar...
Q: A compound with a molar mass of 164.12 g/mole is 58.54 % carbon (12.01 g/mole), 2.46% hydrogen (1.01...
A: Given :- Molecular mass of compound = 164.12 g/mol composition of C, H, & O are 58.54%, 2.46%, ...
Q: How many mL of 0.547 M HNO3 are needed to dissolve 5.63 g of MgCO3?2HNO3(aq) + MgCO3(s) Mg(NO3)2(aq)...
A: Given :- mass of MgCO3 = 5.63 g Molar concentration of HNO3 solution = 0.547 mol/L To calculate :-...
Q: A researcher finds that a sample of an unknown solution, when placed in a 1.00 cm wide cuvette, abso...
A:
Q: for elements (Br, Ca, Fe, Na, S, Si, and Xe) which of the following term(s) apply? a. metal b. nonme...
A: To classify the elements we have see in which block of the periodic table the given element is pres...
Q: 3. Calculate the normality when 7.88g of Ca(NO3)2 was dissolved in 3 lite solution ANSWER: NORMALITY...
A: Normality of the solution is equal to the number of equivalents of solute present in one liter of th...
Q: Part A. Calculate the initial molarity of MCHMMCHM in the river, assuming that the first part of the...
A: MCHM is 4-methylcyclohexanmethanol. The molarity of a solution defines the number of moles of solute...
Q: help question 18
A:
Q: Complete the balanced dissociation equation for the compound below in aqueous solution. If the compo...
A:
Q: Describe (write sentences) a non-computationally-based experiment (i.e. is not solely based upon the...
A: Given: prove that 2-methylpropene is more stable than trans-2-butene
Q: Is hydrogen metal nonmetal metalloid
A: Answer= non-metal.
Q: What is the rate of consumption of C (M/s), if the rate of consumption of B is 0.228 M/s? Use the re...
A: Given data are as follows Rate of consumption of B= Rate of decomposition of B=0.228 M/s Reaction gi...
Q: Which reaction is an example of a precipitation reaction? Group of answer choices 6 HCl (aq) + 2 Al...
A:
Q: What is the vapor pressure of Ag at 2365.8 K if its heat of vaporization and boiling point are 285.0...
A: boiling point = T1 = 2485.2 K We know that liquid boil at 1 atm pressure So, intial vapor pressure =...
Q: Consider the reaction: NO2 + 03 → N03 + 02 [NO,], M [02], M Rate, M/s 6.30 × 10-3 Experiment 1 0.21 ...
A: Rate law gives the relation between rate and concentration of the reactants.
Q: Electrons are transferred In True O False
A: The question is based on the concept of redox reaction. We have to talk about the transfer of electr...
Q: Ammonia is a convenient buffer system in the slightly basic range. (a) What is the pH of a buffer so...
A: pH is used to determine the concentration of hydronium ion.
Q: Calculate the mass of glucose C6H12O6 that contains a million(1.0*10^6 )hydrogen atoms.Be sure your ...
A: 1 mole = 6.0224 x 1023
Q: A reaction with an activation energy of 94.8 kJ/mol is carried out at 281. K and 376. K. The reactio...
A: The Arrhenius equation is given as, lnk2k1=-EaR1T2-1T1 where Ea is the activation energy.
Q: The osmotic pressure of a solution of K2SO4 = 1.400 atm at 13.78 °C. What is the analytical (not the...
A: Osmotic Pressure π = osmotic pressure = 1.400 atm T=Temperature = 13.78°C = 286.78 K Let us conside...
Q: 3. What is the density of N2O4 gas at STP? Round your answer to the tenth place; include the correct...
A: The relation between density and molar mass is: dRT=PM Here, d= density, M=molar mass, P=pressure, T...
Q: Write the shorthand electron configurations for each of the follwing. a. iodine b.zinc c. phosphorou...
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If y...
Q: when 20 grams of salt is dissolved in 95 grams of water, the temperature of the solution decreases f...
A: When the solute is dissolved in the solution and the amount of heat released is known as the heat of...
Q: A certain drug has a half-life in the body of 2.5 h. What should the interval between doses be, if t...
A: Consider that the decomposition of the drug follows first order reaction. The half-life of the drug ...
Q: 1. Find the molarity of a 500 L solution that contains 10 moles of fluorine solute.
A: Since you have posted multiple questions, we will solve only first question for you. If you want any...
Q: 4. Given: pKsp of BaF2 = 10.40. calculate molar solubility in g/L. ANSWER: Molar solubility in g/L=
A: The solubility product constant (Ksp) represents the solubility of products at equilibrium for solid...
Q: The half reaction method can be used to simplify equations. O True O False
A: The half reaction method involve a spliting of overall reaction into two half reaction, one involvin...
Q: (a) In the reaction mechanisms given, there are some chemical species which will get produced in one...
A:
Q: Bromine is a diatomic molecule with a boiling point of 332K and a melting point of 266K. Draw a mole...
A: We have been given the melting and boiling point of Br2 and been asked its structure at different te...
Q: An aqueous solution of barium hydroxide is standardized by titration with a 0.115 M solution of nitr...
A: Answer of this question :- Molarity :- Number of mole of solute dissolved in one litre of solution....
Q: In the laboratory you dissolve 21.7 g of copper(II) fluoride in a volumetric flask and add water...
A: Given data contains, Mass of copper (ii) fluoride is 21.7g. Volume of solution is 500ml. The moles o...
Q: From top to bottom, down the periodic table, ionization energy tends to decrease. This is best expla...
A: Applying basics of periodic properties of elements we can determine variation of ionisation energy a...
Q: Please use the values in the resources listed below instead of the textbook values. Calculate the co...
A: A salt will precipitate only when the ionic product of the concentration of ions exceeds its Ksp val...
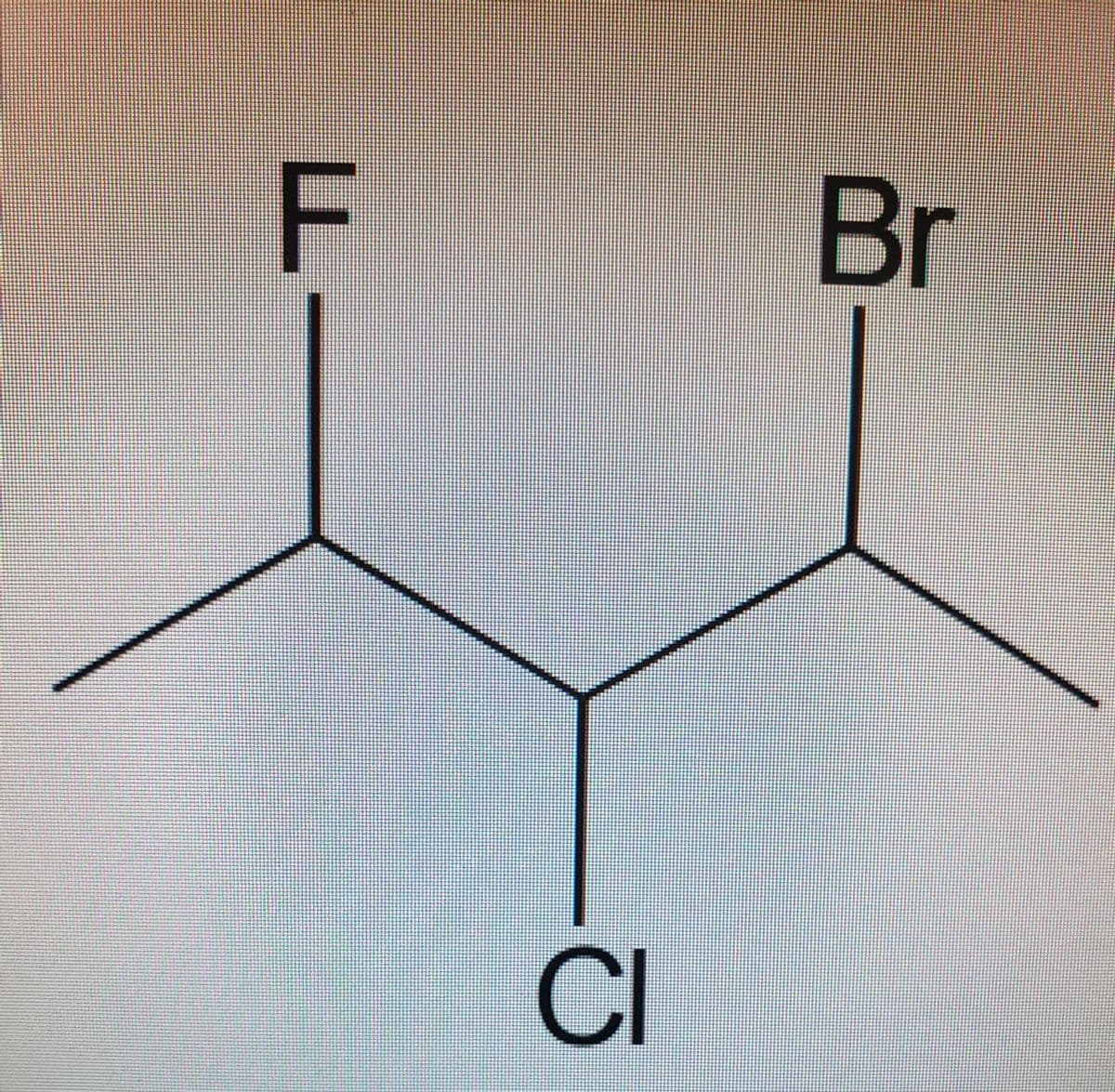
1. Draw all of the possible stereoisomers of the compound shown. Group them as
enantiomeric pairs, or indicate any meso compounds.
2. Label all stereocenters with the absolute stereochemistry (R or S).
Step by step
Solved in 2 steps with 2 images




