Q: Indicate anode, cathode, half cell potential, overall potential, identify the reducing and oxidizing...
A: A question based on electrochemical cells that is to be accomplished.
Q: Experiment No: 10 SPECTROPHOTOMETRIC DETERMINATION OF IRON RESULTS AND INTERPRETATIONS Table 10.1. M...
A: Given:
Q: volume
A:
Q: The K, for pyridine C,H,N is 1.7 x 10°. а) Calculate K, for its conjugate acid C,H,NH". b) Complete ...
A: Pyridine acts as base as follows; C5H5N + H2O = C5H5NH+OH- ↔C5H5NH+ + OH- As it weakly dissociates ...
Q: LINO3 is added to water. Will it change the pH, and if so, how? Yes, and the pH will be 7. You want...
A: We can determine if a salt added to water will make pH of water more acidic,basic or will remain unc...
Q: 1. Half-fill a 50-ml beaker with water. Warm gently with a small flame but do not boil. What appears...
A: Evaporation is the phenomenon where the liquid turns into a vapor.
Q: PROCEDURE 1000 mg L-asocrbic acid tablet (pulverized into fine powder) Molar mass of tablet = 176....
A: A question based on introduction to chemistry that is to be accomplished.
Q: Use the following two half-reactions to write balanced net ionic equations for one spontaneous react...
A:
Q: Why does freezing point decrease and boiling point increase when solute is added? Please answer in n...
A: Please find the below attachment.
Q: 0.512 g sample of CaCO3 is dissolved in 12 M HCl and the mixture is diluted to 250 mL. A small amoun...
A: CaCO3 + 2HCl --> CaCl2 + H2O + CO2 Mass of CaCO3 = 0.512 g Molar mass of CaCO3 = 100 g/mol
Q: For the reaction A+B+C→D+EA+B+C→D+E, the initial reaction rate was measured for various initial conc...
A:
Q: Discuss the benefits and disadvantages of each of the following types of milk containers ( glass, t...
A:
Q: What is the Kohlrausch plot of a weak acid? 1 2 O 2
A: Kohlrausch's law is used to calculate molar conductance of weak acid by graphical methods. It has t...
Q: What is the pressure in Pa due to the seawater at a depth of 10 m? The density of seawater is 1.025 ...
A:
Q: What volume of 1.44 M KMN04, in liters, contains 292 g of solute? Volume = L. %3D
A: Given that, Molarity of KMnO4 = 1.44 M Molar mass = 158.034 g mol-1 Mass of KMnO4 = 292 g Volume (...
Q: D: Define and draw the Unit Cell and give an example.
A: Unit cell can be defined as the smallest repeating unit in space lattice which, when repeated over a...
Q: Consider the following rate law: rate = k[acetone][l2]°[H*]ª What effect would decreasing the concen...
A: Given, Rate law expression: rate = k[acetone]1[I2]0[H+]1 Here, k = rate constant [acetone]1 = concen...
Q: Is 4-bromo-4-ethylhept-1-ene capable of hydrogen bonding? if no explain why?
A: Hydrogen bonding : is the strongest intermolecular force formed by the attractive force between a hy...
Q: A student mixes 20 mL of 0.1 M HCl and 20 mL of 0.1 M KOH in a coffee cup calorimeter. Upon complete...
A:
Q: Does an electromagnetic spectrophotometer using photoelectron spectroscopy exist? Explain why or why...
A: A spectrophotometer is an instrument which measures the absorption of light at each wavelength of li...
Q: You need to selectively precipitate and separate the metal jons Co", Pb*?, and Sr² in a tap water sa...
A: Ksp is the solubility product and it tells the measure of ionic concentration required to precipitat...
Q: ii) What are the adjusted retention times for components A and B? iv) What is the relative retention...
A: 3 . For component A = 8-1.2=6.8 For component B = 10-1.2 = 8.8 4. Separation factor = 1.29
Q: 10. Differentiate between double salt and complex salts with suitable examples. Based on Werner's th...
A: Double Salts : These exist only in solid state and dissociate into constituent species in their so...
Q: what reagents can you use to connect the cl to the double bond to make it all one product? CI 3D0
A:
Q: What is the pH of a weak base-strong acid titration when 20.0 mL of 0.56 M of NaOCl (Ka = 3.0*10-8) ...
A:
Q: Shonld be mixed rith s.0 lth pH 10.00? 9-13 How mony grams of maco, (Fm 1o5.9) 5.009 g buffer hith p...
A: Volume of buffer solution = 100 mL pH = 10.00
Q: Please provide a synthetic route for this molecule Br
A:
Q: Question 23 The equilibrium constant Kc for the reaction fructose-1,6-diphosphate = glyceraldehyde-3...
A: Introduction : The equilibrium constant, K, is the concentration of the reactants over the concentr...
Q: A sample of 1.00 mol of perfect gas molecules with Cp = 7/2 R is put through the following cycle: (a...
A: Given 1.00 mol of a perfect gas having Cp = 7/2 R Pressure =1.00 atm Q, W, dU, and dH for each ste...
Q: 0.1 g of the mixture of na2so4 and k2so4 is taken and 100 ml of solution is prepared. 10 ml of this ...
A:
Q: D 11. What is the molecular formula for the following compound?as a. C,H, b. CH12 c. C,H12 d. C;H14 ...
A: Molecular formula: In a molecular formula we describe the number of atoms of all the elements presen...
Q: Briefly explain why Fe* is a Lewis acid, but Fe" is not a Brønsted-Lowry acid.
A:
Q: Identify the strongest IMF acting on the pair of substances given. Cl2and HCl HF and NH3 CH3OH and ...
A: An intermolecular force is the force that mediates interaction between molecules, including the elec...
Q: 1.Consider the complete combustion of methane gas, CH4(g): Which of the following BEST describes the...
A: Entropy is defined as measure of randomness or disorder of system.
Q: An unknown compound has the following chemical formula: PtCly where x stands for a whole number. Mea...
A: Here, the unknown compound given is PtClx. No of moles of Pt and Cl are given .we have to find chemi...
Q: Cite some advantages of using the pH paper than the pH meter and vice versa.
A: pH paper and pH meter both are use to determine the acidity of the sample.
Q: For copper (Cu, Z= 29), provide the electron configuration
A: Electrons are filled in a different atomic orbital in an atom. According to the Aufbau rule, the ele...
Q: For the following reaction, 5.92 grams of hydrogen gas are mixed with excess nitrogen gas . The reac...
A:
Q: Computation. Compute for the amount of NaHCO3 that reacted with 6.2 g citric acid. 3 NaHCO3 (MW:84g...
A:
Q: а. 0.0 mL pH = b. 20.0 mL pH = с. 30.0 mL pH = d. 40.0 mL pH = е. 90.0 mL PH =
A:
Q: Referring to Question 2 in the Feedback Form, calculate the rate constant (k) for the reaction of Ye...
A: We would use the equation which is used to plot ln(abs) vs time plot.
Q: the attraction forces that allow water to function as a universal solvent are? a. van der waals forc...
A: The characteristics of van der Waals forces are, Weak intermolecular forces Depend on the distance...
Q: PROCEDURE 1000 mg L-asocrbic acid tablet (pulverized into fine powder) Molar mass of tablet = 176....
A:
Q: For the following reaction, 65.2 grams of barium hydroxide are allowed to react with 35.2 grams of s...
A: Limiting reagent is the reactant that get Completely used up in the reaction and hence ends the reac...
Q: A 8.25 g sample of an aqueous solution of hydrobromic acid contains an unknown amount of the acid. I...
A:
Q: The Gibbs Free Energy, Enthalpy, and Entropy are state functions; hence, path-dependent. True Fa...
A: State functions are those functions which depends on initial and final state of the substance.
Q: Define 1. Molecular mass 2. Mole
A: Molecular mass: Molecular mass is a number which indicates how many times one molecule of a substan...
Q: Summarize the possible values of all the quantum number is n=1,2 and 3
A: Summarize the possible values of all the quantum number is n=1,2 and 3
Q: In some cases, the rate law of a reaction depends on the concentration of three reactants. Such is t...
A:
NEED ASAP
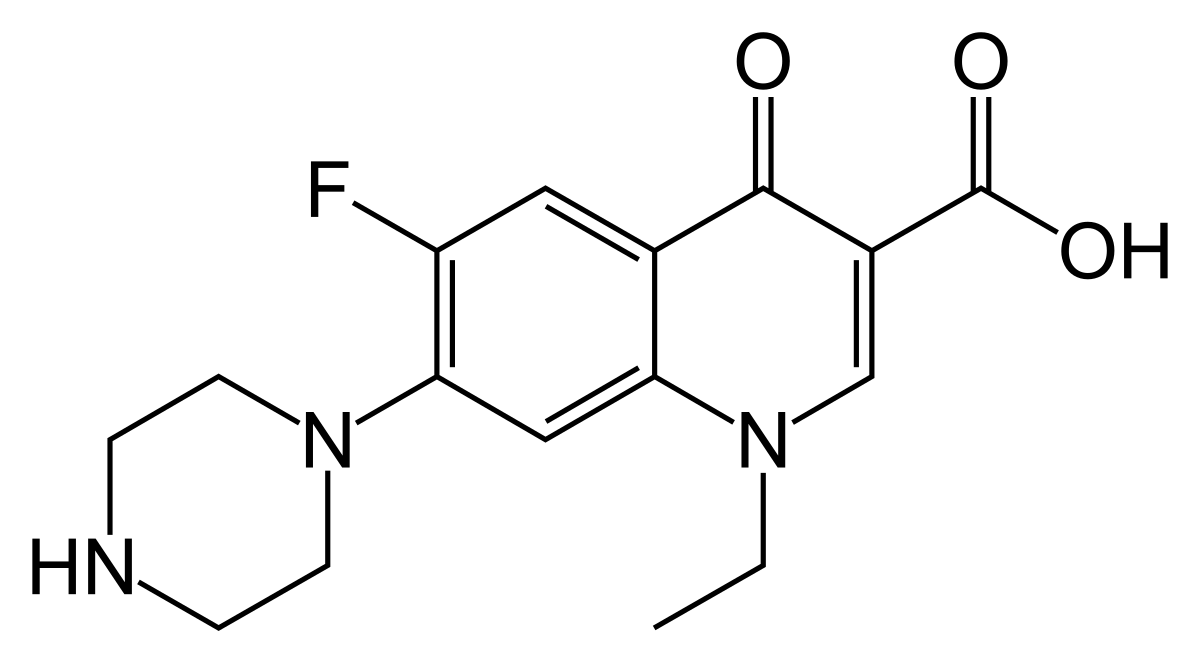
What are the present Functional Groups in the drug molecule Norfloxacin ? (pls label if possible, thanks!)
Step by step
Solved in 2 steps with 1 images


