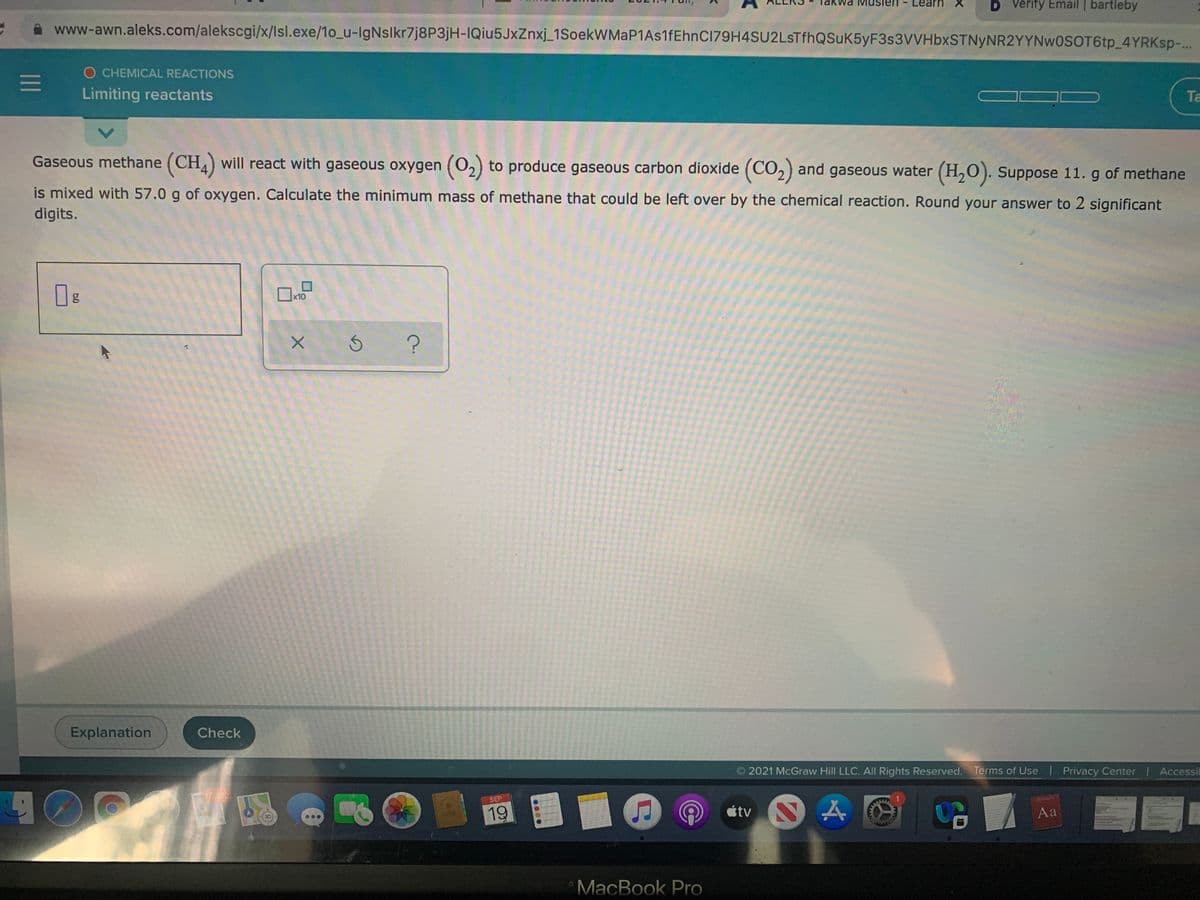
F3S3VVHD TNyNRZYYNWOSOT6tp_4Y O CHEMICAL REACTIONS Limiting reactants seous methane (CH) will react with gaseous oxygen (0,) to produce gaseous carbon dioxide (CO,) and gaseous water (H,0). Suppose 11. g of me nixed with 57.0 g of oxygen. Calculate the minimum mass of methane that could be left over by the chemical reaction. Round your answer to 2 significa its.
F3S3VVHD TNyNRZYYNWOSOT6tp_4Y O CHEMICAL REACTIONS Limiting reactants seous methane (CH) will react with gaseous oxygen (0,) to produce gaseous carbon dioxide (CO,) and gaseous water (H,0). Suppose 11. g of me nixed with 57.0 g of oxygen. Calculate the minimum mass of methane that could be left over by the chemical reaction. Round your answer to 2 significa its.
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
Transcribed Image Text:b Verify Email | bartleby
www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IQiu5JxZnxj_1SoekWMaP1As1fEhnCI79H4SU2LsTfhQSuK5yF3s3VVHbxSTNyNR2YYNwOSOT6tp_4YRKSP-..
O CHEMICAL REACTIONS
Limiting reactants
Ta
Gaseous methane (CH) will react with gaseous oxygen (O,) to produce gaseous carbon dioxide (CO,) and gaseous water (H,0). Suppose 11. g of methane
is mixed with 57.0 g of oxygen. Calculate the minimum mass of methane that could be left over by the chemical reaction. Round your answer to 2 significant
digits.
x10
Explanation
Check
© 2021 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessil
100
SEP
Detion
19
étv S
Aa
100
MacBook Pro
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

