Q: How many grams of solid Cu2SO4 5H2O (249.68 g/mol) must be dissolved in 400 mL of water to obtain a ...
A: Given :- molarity of Cu+ ions = 0.0899 M volume of water = 400 mL molar mass of Cu2SO4 5H2O = 249...
Q: What is the molar solubility of Ag2SO4 in a solution that is 0.0522 M AgNO3? Ksp= 1.40x10^-5
A:
Q: When propane, C3H8, is burned, carbon dioxide and water vapor are produced according to the followin...
A: Law of Conservation of Mass: The Law of Conservation of Mass states that in a chemical reaction the ...
Q: Write a nuclear equation to describe the spontaneous fission of Am-244 to form I-134 and Mo-107. Det...
A:
Q: An unidentified ionic compound contains approximately 29.85 g sodium, 67.50 g chromium, and 72.67 g ...
A:
Q: What is the molarity of K+ in a solution that contains 89.72 ppm of K3Fe(CN)6 (329.3 g/mol)? 8.17...
A:
Q: What is the percentage composition by mass of the substance shown below? % C = % S = % H = How many ...
A:
Q: Calculate the volume occupied by 122.541 g of water at 22.35 °C.
A:
Q: 1. Lithium reacts with oxygen to form lithium oxide according to the equation: 4Li + O2 = 2Li2O. If ...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: (a)Write the equilibrium expression for the following reaction: Co^2+ (aq) + 3 NH3(aq) Co(NH3)3^2 + ...
A: Kc is the ratio of the equilibrium concentrations of product over equilibrium concentrations of reac...
Q: 1. A suggested way to organize your data for Part B of the lab is below. 0.00200 M 0.1 M KSCN 0.0020...
A: The data table given is,
Q: I don't understand this problem! Please help! The question is: In the following balanced chemical re...
A: The chemical equation represents the possible reactants that combine to yield the products. In this ...
Q: To what volume should you dilute 30 mL of a 12 M H2SO4 solution to obtain a 0.16 M H2O4 solution?
A: In case of dilution of any electrolyte, the number of moles or millimoles will remain unchanged. i.e...
Q: Increasing the temperature usually causes the rate of a chemical reaction to increase. If the rate o...
A:
Q: What mass of Ba(NO3)2 (261.34 g/mol) is needed to convert 2.33 g of NaIO3 (197.89 g/mol) to Ba(IO3)2...
A: Barium nitrate react with sodium iodate and produced Ba(IO3)2
Q: If 31.4g of NaOH (MM= f Fi 40.00 g/mol) are added to 500.0 mL volumetric flask, and water is added t...
A: Answer: To find out the molar concentration of NaOH, first of all we will find out its number of mol...
Q: Which is bronsted lowry acid and which is bronsted lowry base, and which is no one. Answer and expla...
A:
Q: A group of students were trying to experimentally determine the density of an unknown liquid. They d...
A: The data given is, And the volume of the liquid = 5.00 mL.
Q: In the preparation of the titrant, the solution must be aged for at least 24 hours in the dark. Stat...
A:
Q: Nitrogen and oxygen react to produce nitric oxide according to the following equation: N2 (g) + O2 ...
A:
Q: What is the molarity of a 1.44 (m/v)% solution of HCOOH (formic acid)?
A: Given: %(m/v) of HCOOH = 1.44 % Assuming 100 mL i.e. 0.100 L of the solution.
Q: What does amphoteric mean? Group of answer choices The substance has a negligible conjugate base. Th...
A: The word amphoteric comes from the Greek word amphoteros or amphoteroi meaning "each or both of two"...
Q: What is the mass in grams of 38.50 moles of LiCl?
A:
Q: What volume of methane (CH4) is required to react with 162 liters of carbon tetrachloride according ...
A: The balanced chemical equation for the reaction of methane (CH4) and carbon tetrachloride (CCl4) is ...
Q: A sealed container with a volume of 63.8 mL is filled with a gas at 22°C at 747mmHg. The mass of the...
A: We will use ideal gas equation to find molar mass of gas and then estimate the gas .
Q: PM Tue Mar 1 -e 64% +: 0 9. Sodium carbonate reacts with nitric acid according to the following equa...
A: Na2CO3 + 2HNO3 ------> 2NaNO3 + CO2 + H2O 3Fe + 4H2O ---------> 4H2 + Fe3O4
Q: A solution containing about 0.001 M Ca2+ is being measured using a calcium-specific electrode. If an...
A: Concentration of Ca2+ = 0.001M Permissible interference from Na+ = 4% We have to calculate the maxim...
Q: Which of the given phenomenon is explained by colligative property? Freezing of ice Evap...
A: Answer Which of the given phenomenon is explained by colligative...
Q: Classify the sigmatropic rearrangement with bracketed numbers.
A: Sigmatropic rearrangement : is a pericyclic intramolecular rearrangement reaction in which one sigma...
Q: Two chemicals A and B are combined to form a chemical C. The rate, or velocity, of the reaction is p...
A:
Q: 1 3 NEXT A 0.60 M aqueous solution of HNO, is prepared. Fill in the ICE table with the appropriate v...
A: Given : We have to fill ICE table for the given reaction.
Q: Give the classification of the following alcohols as to primary, secondary, or tertiary. U (CHals-C-...
A:
Q: A mixture of gases is at equilibrium: C2H6 (g) → C2H4 (g) + H2 (g) ∆H = 136.94 kJ (a) Does the equ...
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on ...
Q: if 1 pint of blood weigh 1lb , calculate 900ml of blood .
A: Given: Mass of 1 pint of blood = 1 lb. And volume of blood = 900 mL.
Q: When 75.06 g of metal is submerged completely, the water in a graduated cylinder rise from 40.52 mL ...
A:
Q: The molar mass of an unknown compound was determined in three repeat experiments. The molar masses o...
A:
Q: Sodium stearate, a product of fatty acid and strong base, has the following structure. What forces o...
A: The detailed answer of the given question is done on the white sheets of paper attached below. Sod...
Q: The equation for the dissolving of lead (II) chloride (including heat) is: PBCI2(s) + heat Pb2+ (a q...
A:
Q: At a certain temperature this reaction follows first-order kinetics with a rate constant of 0.034 s ...
A: Given that, a first-order kinetics reaction is NH4OH (aq) →NH3(aq) + H2O (aq). The rate constant of...
Q: . Arrange the following in the order of increasing boiling point: CO,, KCI, CH,I, CH,OH. Identify th...
A: Boiling point:The temperature at which the vapor pressure of a liquid becomes equal to the atmospher...
Q: In order to form Ba(IO3)2, 500 mL of 0.5000 M Ba(NO3)2 was mixed with 500 mL of 0.0500 M NaIO3. How...
A: Solving only first three subparts in accordance with the guidelines. We will first calculate millimo...
Q: What is the pH of a 0.100 M solution of LiBrO(aq) at 25'C? Assume that LiBrO is a strong electrolyte...
A: Since you have posted multiple questions, we are entitled to answer the first only. Given: Concentra...
Q: Which reaction results in an increase in the entropy of the system? * O H20(g) H20(L) H20(L) H20(s) ...
A: Entropy: Entropy can be defined as the measurement of randomness or disorderness in the thermodynami...
Q: Calculate the enthalpy change for the complete complete combustion of ethane (C2H6). (Note: the onl...
A:
Q: How to improve the yeild in haber process
A: Given: Habers process. It is a process in which the formation of ammonia takes place from nitrogen a...
Q: If I combined 15.0 grams of calcium hydroxide with 75.0 mL of 0.500M HCl, how many grams of calcium ...
A: Number of moles is calculated by dividing the given mass with molecular mass.u
Q: I need help to find out why we use a reflux condenser, vacuum filtration in the Diels-Alder Reacti...
A: In a polycyclic aromatic hydrocarbon, the aromatic rings that are made up of only carbon and hydroge...
Q: Which statement is false regarding labels on chemical containers? a. labels tell the embalmer how t...
A: Label is an indicative of hazardous behaviour and precurtions need to be employed during use.
Q: A mixture ui gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: According to the following reaction: 2H,S (9) + 30, (9) – 2H,0 (1) + 250, (g) What would you multipl...
A: Here, the balanced chemical equation given is 2H2S(g)+3O2(g)------>2H2O(l)+2SO2(g) We will answer...
I need help explaning the
Diels-Alder Reaction mecahnism
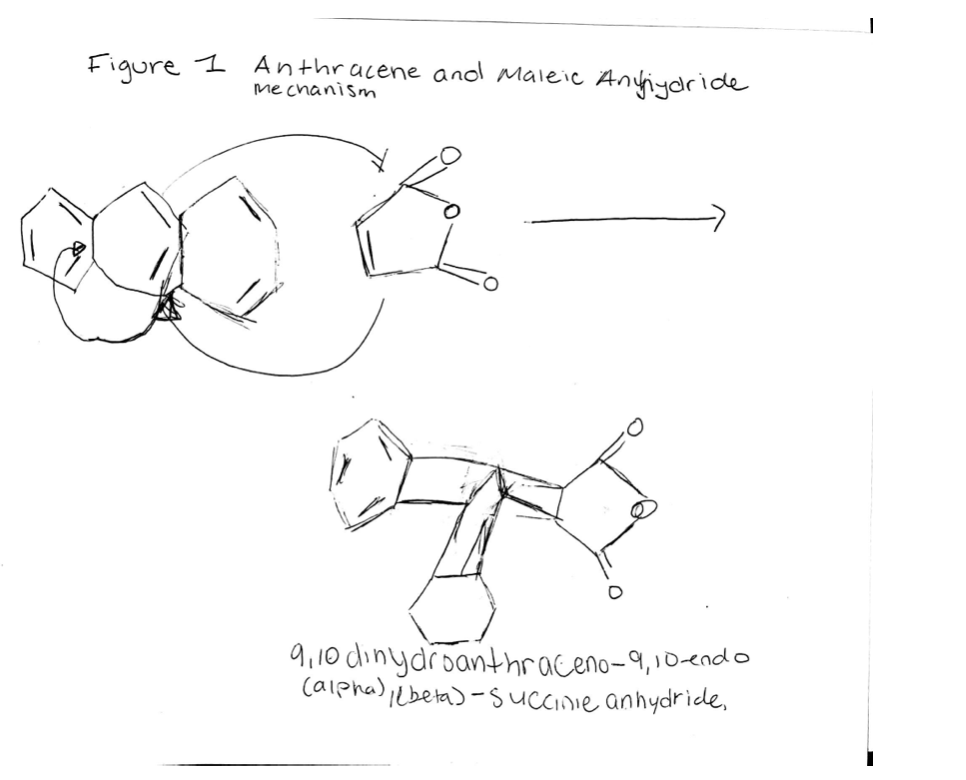
Step by step
Solved in 2 steps with 1 images

- Calculate the Ksp for uraninite (reaction 1) and rutherfordine (reaction 2). UO2 + 4 H+ ---U4+ + 2 H2O (reaction 1) UO2CO3 ---UO22+ + CO32- (reaction 2)whats the stracures of C6H11NO2 in HNMR spectrascupeCalculate the Ksp for uraninite (reaction 1) and rutherfordine (reaction 2). UO2 + 4 H+ ßà U4+ + 2 H2O (reaction 1) UO2CO3 ßà UO22+ + CO32- (reaction 2)
- Write the form of lysine most prevalent at pH 1.0 and then show its reaction with the following. Consult Table 27.2 for pKa values of the ionizable groups in lysine. Q.) 2 mol NaOH1.Which of the following correlations about the analyses of iodine and saponificationnumbers is/are incorrect?a. Alcoholic KOH: dissolve KOH to lipid sampleb. Hanus reagent: react with saturated lipid bondc. Reflux set-up: hasten reactiond. Blank titration: determine total mol of KOH2. A triacylglycerol (Sample X) was subjected to saponification analysis. 1.1 grams of thesample was refluxed with 5% alcoholic KOH and the refluxed solution was titrated with 17.2 mLof 0.44 M HCl titrant. The blank solution was titrated with 25.6 mL of the titrant.i. What is the saponification number of the sample? ii. Which of the following could be the identity of the sample? Show solutions pls a. Tripalmitin (MW: 807.3)b. Trilaurin (MW: 639.8)c. Triolein (MW: 885:4)d. Tristearin (MW: 891.48)Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA. a. Explain why it can be used only under slightly acidic conditions. b. Explain why it can cross-link DNA.
- Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA. a. Explain why it can be used only under slightly acidic conditions.b. Explain why it can cross-link DNA.How to create 50 uL of TER (with compostions 10 mM Tris-HCl, pH 7.4; 1 mM EDTA, pH 8; 100 ug/mL RNase A)? Show calculationsWhy an amino group (NH2) directs electrophilic aromatic substitutionto the ortho and para positions ?

