Table 27.2 pK, Values for lonizable Groups of Amino Acids pK of a-COOH pK, of a-NH, pK, of Side Chain Isoelectric Amino Acid Point (pl) Alanine 2.35 9.87 6.11 Arginine 2.01 9.04 12.48 10.76 Asparagine 2.02 8.80 5.41 Aspartic acid 2.10 9.82 3.86 2.98 Cysteine 2.05 10.25 8.00 5.02 Glutamic acid 2.10 9.47 4.07 3.08 Glutamine 2.17 9.13 5.65 - Glycine 2.35 9.78 6.06 - Histidine 1.77 9.18 6.10 7.64 Isoleucine 2.32 9.76 6.04 Leucine 2.33 9.74 6.04 Lysine 2.18 8.95 10.53 9.74 Methionine 2.28 9.21 5.74 Phenylalanine 2.58 9.24 5.91 Proline 2.00 10.60 6.30 Serine 2.21 9.15 5.68 Threonine 2.09 9.10 5.60 Tryptophan 2.38 9.39 5.88 Tyrosine 2.20 9.11 10.07 5.63 Valine 2.29 9.72 6.00
Table 27.2 pK, Values for lonizable Groups of Amino Acids pK of a-COOH pK, of a-NH, pK, of Side Chain Isoelectric Amino Acid Point (pl) Alanine 2.35 9.87 6.11 Arginine 2.01 9.04 12.48 10.76 Asparagine 2.02 8.80 5.41 Aspartic acid 2.10 9.82 3.86 2.98 Cysteine 2.05 10.25 8.00 5.02 Glutamic acid 2.10 9.47 4.07 3.08 Glutamine 2.17 9.13 5.65 - Glycine 2.35 9.78 6.06 - Histidine 1.77 9.18 6.10 7.64 Isoleucine 2.32 9.76 6.04 Leucine 2.33 9.74 6.04 Lysine 2.18 8.95 10.53 9.74 Methionine 2.28 9.21 5.74 Phenylalanine 2.58 9.24 5.91 Proline 2.00 10.60 6.30 Serine 2.21 9.15 5.68 Threonine 2.09 9.10 5.60 Tryptophan 2.38 9.39 5.88 Tyrosine 2.20 9.11 10.07 5.63 Valine 2.29 9.72 6.00
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter21: Biochemistry
Section: Chapter Questions
Problem 6A
Related questions
Question
Write the form of lysine most prevalent at pH 1.0 and then show its reaction with the following. Consult Table 27.2 for pKa values of the ionizable groups in lysine.
Q.) 2 mol NaOH
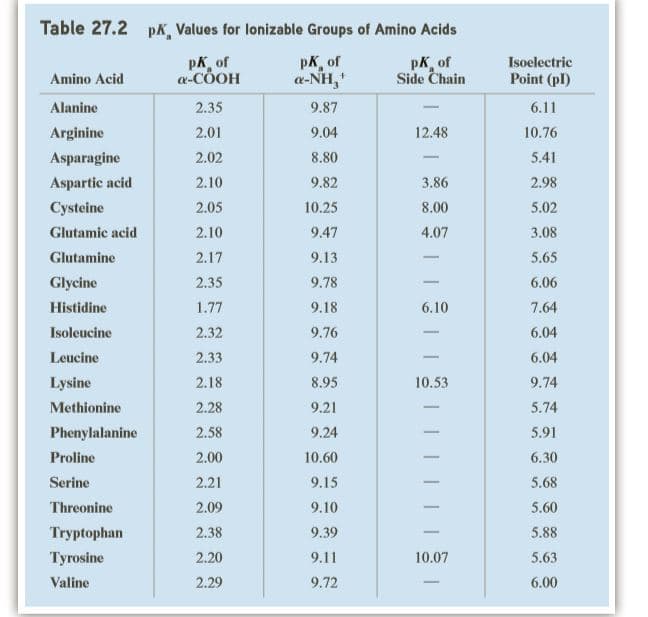
Transcribed Image Text:Table 27.2
pK, Values for lonizable Groups of Amino Acids
pK of
a-COOH
pK, of
a-NH,
pK, of
Side Chain
Isoelectric
Amino Acid
Point (pl)
Alanine
2.35
9.87
6.11
Arginine
2.01
9.04
12.48
10.76
Asparagine
2.02
8.80
5.41
Aspartic acid
2.10
9.82
3.86
2.98
Cysteine
2.05
10.25
8.00
5.02
Glutamic acid
2.10
9.47
4.07
3.08
Glutamine
2.17
9.13
5.65
-
Glycine
2.35
9.78
6.06
-
Histidine
1.77
9.18
6.10
7.64
Isoleucine
2.32
9.76
6.04
Leucine
2.33
9.74
6.04
Lysine
2.18
8.95
10.53
9.74
Methionine
2.28
9.21
5.74
Phenylalanine
2.58
9.24
5.91
Proline
2.00
10.60
6.30
Serine
2.21
9.15
5.68
Threonine
2.09
9.10
5.60
Tryptophan
2.38
9.39
5.88
Tyrosine
2.20
9.11
10.07
5.63
Valine
2.29
9.72
6.00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning