Find the hybrid orbital type for each atom, the carbon-carbon-oxygen bond angle, the molecular geometry of oxygen, and the number of sigma and pi bonds in the molecule.
Find the hybrid orbital type for each atom, the carbon-carbon-oxygen bond angle, the molecular geometry of oxygen, and the number of sigma and pi bonds in the molecule.
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter1: Covalent Bonding And Shapes Of Molecules
Section: Chapter Questions
Problem 1.61P: Using cartoon representations, draw a molecular orbital mixing diagram for a CO bond. In your...
Related questions
Question
100%
Find the hybrid orbital type for each atom, the carbon-carbon-oxygen bond angle, the molecular geometry of oxygen, and the number of sigma and pi bonds in the molecule.
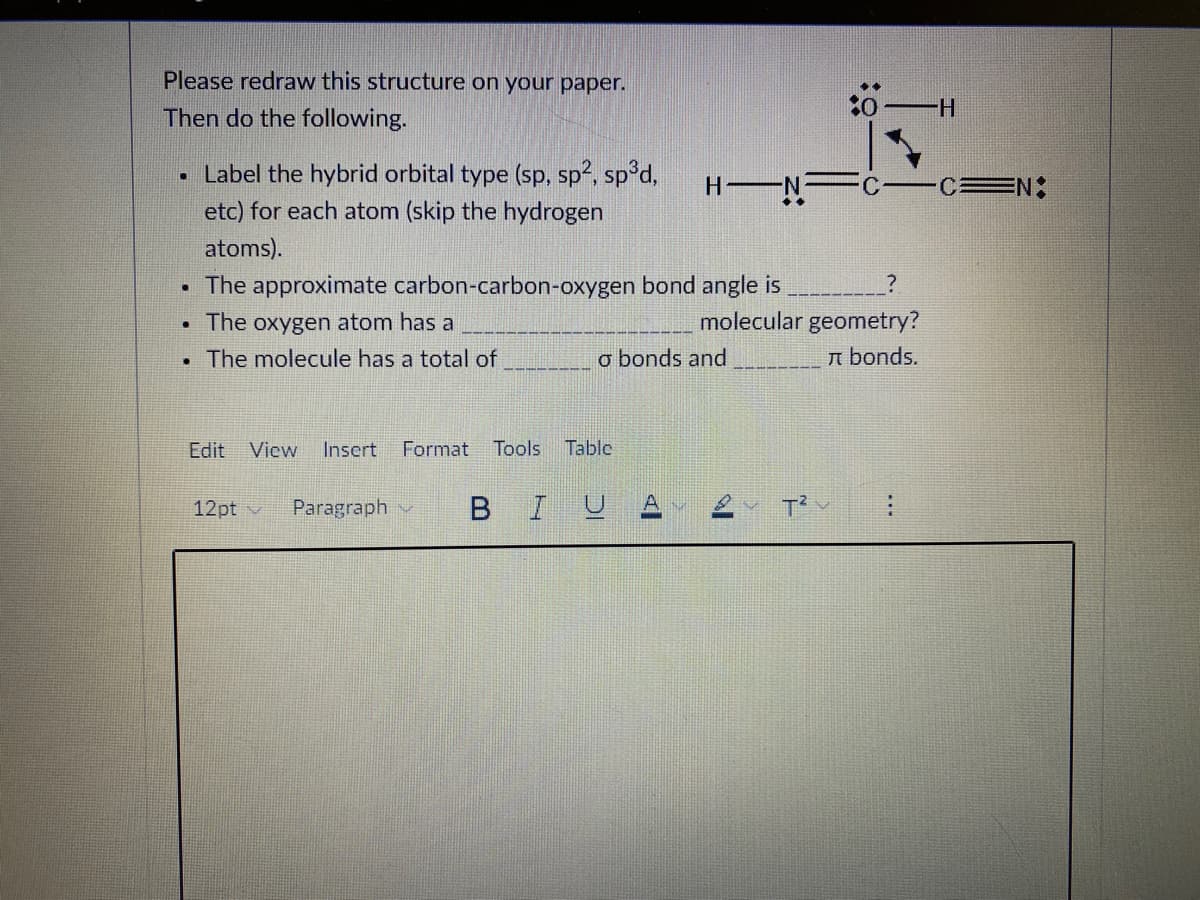
Transcribed Image Text:Please redraw this structure on your paper.
Then do the following.
0 -H
• Label the hybrid orbital type (sp, sp?, sp°d,
etc) for each atom (skip the hydrogen
H N
CEN:
atoms).
• The approximate carbon-carbon-oxygen bond angle is
• The oxygen atom has a
molecular geometry?
• The molecule has a total of
o bonds and
n bonds.
Edit View
Insert
Format
Tools Tablc
12pt v
Paragraph
BIUA
T
...
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning