Following several days at high altitude the body responds to the decreased oxygen content of the atmosphere by increasing the amount of 2,3-bisphosphoglycerate it produces. Based on this information and refering to the graph below, identify the correct response from the options. Y (fraction saturation) 1.0 0.8 0.6 0.4 0.2 0.0 0 20 Oxygen Binding plot 40 60 p02 (torr) 80 100 +
Following several days at high altitude the body responds to the decreased oxygen content of the atmosphere by increasing the amount of 2,3-bisphosphoglycerate it produces. Based on this information and refering to the graph below, identify the correct response from the options. Y (fraction saturation) 1.0 0.8 0.6 0.4 0.2 0.0 0 20 Oxygen Binding plot 40 60 p02 (torr) 80 100 +
Chapter13: Dimensional Analysis/units Conversion
Section: Chapter Questions
Problem 38SST
Related questions
Question
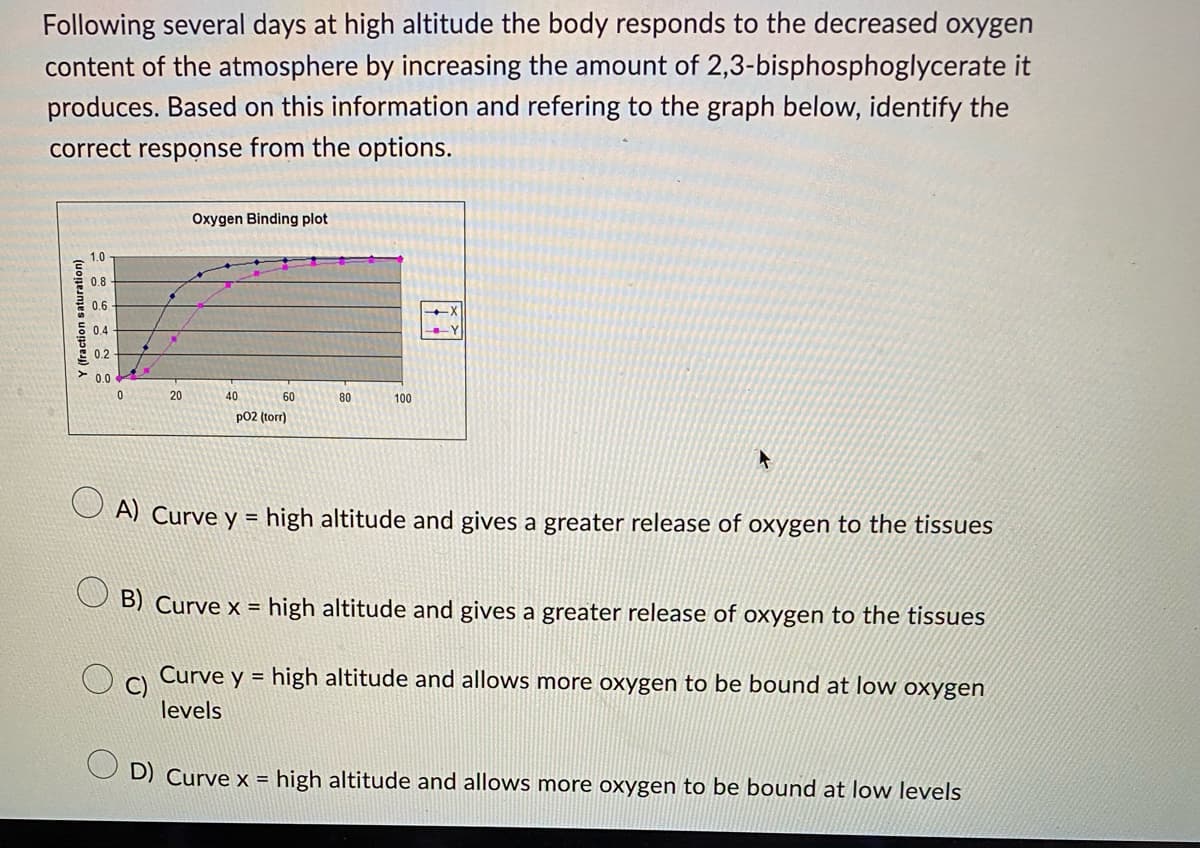
Transcribed Image Text:Following several days at high altitude the body responds to the decreased oxygen
content of the atmosphere by increasing the amount of 2,3-bisphosphoglycerate it
produces. Based on this information and refering to the graph below, identify the
correct response from the options.
Y (fraction saturation)
1.0
0.8
0.6
0.4
0.2
0.0
0
20
Oxygen Binding plot
40
60
p02 (torr)
80
100
A) Curve y=high altitude and gives a greater release of oxygen to the tissues
B) Curve x = high altitude and gives a greater release of oxygen to the tissues
C)
Curve y=high altitude and allows more oxygen to be bound at low oxygen
levels
D) Curve x = high altitude and allows more oxygen to be bound at low levels
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

