For a certain system, the equilibrium constant, K, is 8.73 x 1012. What is true about this system once it reaches equilibrium? O Products and reactants continue to form and the concentration of products will increase O Products and reactants continue to form and the concentration of reactants will increase O Products and reactants continue to form at equal rates. O The rate of formation of products is greater than the rate of formation of reactants, because K>>> 1 O None of the above statements are true once the system reached equilibrium.
For a certain system, the equilibrium constant, K, is 8.73 x 1012. What is true about this system once it reaches equilibrium? O Products and reactants continue to form and the concentration of products will increase O Products and reactants continue to form and the concentration of reactants will increase O Products and reactants continue to form at equal rates. O The rate of formation of products is greater than the rate of formation of reactants, because K>>> 1 O None of the above statements are true once the system reached equilibrium.
Chapter1: Excel Basics
Section: Chapter Questions
Problem 5P
Related questions
Question
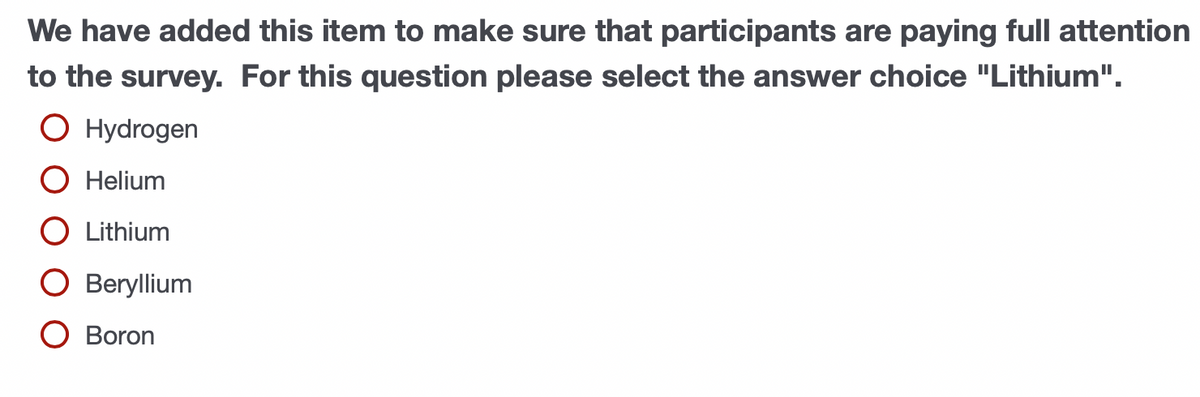
Transcribed Image Text:We have added this item to make sure that participants are paying full attention
to the survey. For this question please select the answer choice "Lithium".
O Hydrogen
O Helium
O Lithium
O Beryllium
O Boron

Transcribed Image Text:For a certain system, the equilibrium constant, K, is 8.73 x 1012. What is true
about this system once it reaches equilibrium?
O Products and reactants continue to form and the concentration of products will
increase
O Products and reactants continue to form and the concentration of reactants will
increase
O Products and reactants continue to form at equal rates.
The rate of formation of products is greater than the rate of formation of reactants,
because K >>> 1
None of the above statements are true once the system reached equilibrium.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning