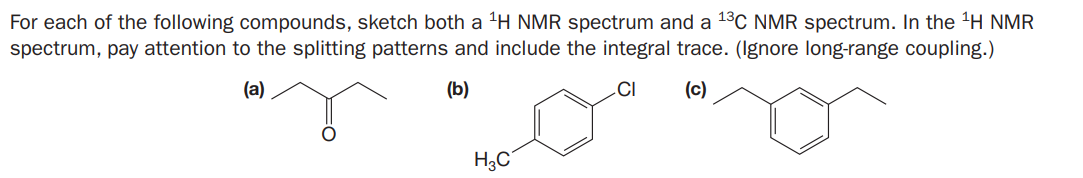
For each of the following compounds, sketch both a 'H NMR spectrum and a 1°C NMR spectrum. In the 'H NMR spectrum, pay attention to the splitting patterns and include the integral trace. (Ignore long-range coupling.) (a) (b) .CI (c) H3C
Q: Compound A with molecular formula C6H10 has two peaks in its 1H NMR spectrum, both of which are…
A:
Q: How could 1H NMR spectroscopy be used to distinguish among isomersA, B, and C?
A: It is type of spectroscopy called as Nuclear Magnetic resonance spectroscopy. It is analytical tool…
Q: The spectrum of compound F with molecular formula C5H602 is shown below. IR spectrum shows a medium…
A:
Q: Provide a detailed analysis of IR, Mass, 1H NMR and 13C NMR data for the following compound
A:
Q: Deduce a possible structure for the compound with the IR absorptions of C3H3Br:3300,2900,2100Cm
A:
Q: Propose structures that are consistent with the following spectra. (Integral ratios are given from…
A: The 1H NMR spectrum of a compound with molecular formula C4H10O2 has two singlets with integral…
Q: How could you use chemical shift and integration data in 1H NMR spectroscopy to distinguish between…
A: Due to the presence of six protons of two methyl groups and four protons of two methylene groups,…
Q: Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or…
A:
Q: Compound A has prominent infrared absorptions at 1050, 1786, and 1852 cm and shows a single…
A:
Q: 1- FROM structure above Which kinds of transition could you expect from the given compounds?…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: 1(a) Base on the information provided, determine the structures of compounds. (b) Assign all peaks…
A:
Q: What is the possible unknown compound based on the IR Spectrum, please give detailed explanation on…
A: Ans: 3400 cm-1, 3300 cm-1 N-H stretch. Primary amine present in the compound. 3500 to 3000 cm-1…
Q: Compound A has molecular formula C9H18O but shows only one singlet in the 1H-NMR spectrum. Suggest a…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: :) Propose a structure for compound A that satisfies ALL the data provided: (GIVE REASONS FOR YOUR…
A: Note : MF = C5H10 (DBE= 5-10/2+1 = 6-5 = 1). Compound A do not decolorizes the bromine solution…
Q: Compounds C and D are isomers with a molecular formula of CsH100 that give a negative Tollens' test.…
A: C5h10o
Q: Calculate the index of hydrogen deficiency (degrees of unsaturation) for the following molecule with…
A: The structure of the following is:
Q: (a) Show, for the example of paracetamol and its meta-isomer (i.e. para- acetamidophenol and…
A: Every nuclei will have spin and if an external magnetic field is applied, there will be an energy…
Q: Annotate the NMR spectra. Assign the hydrogens to the structure
A:
Q: A chemist is summoned to an abandoned waste-disposal site to determine the contents of a leaking,…
A: (a)
Q: a) Using the molecular formula shown, calculate the elements of unsaturation for this compound.…
A: Solution: The step-by-step solution of the given questiona are shown below ---
Q: (d) THE UV-spectra of two isomers of cinnamic acid absorb at 272 nm and 268 nm. Identify the isomers…
A:
Q: Propose a structure consistent with following spectral data for a compound C8H18O2
A: Solution-
Q: The 1H-NMR spectrum of lycopene is provided below. Comment on the general features of the NMR…
A: NMR stands for nuclear magnetic resonance spectroscopy. NMR spectroscopic technique is used to…
Q: 1. Predict the maximum total number of possible peaks in the 13C NMR spectrum of sesamin (Hint: ,…
A: If the carbon atoms are in different environments then we will get signals for these carbon in 13C…
Q: Draw the structure of a compound with the formula C5H10O2 (along with the reasons of choosing it)…
A:
Q: Based on the included NMR spectrums of ferrocene and ferrocenecarboxylic acid, how many “peaks”…
A: C-13 NMR of Ferrocene and Ferrocenecarboxylic acid
Q: A compound with a formula of C8H100 has the IR and NMR below. Propose a structure.
A: C8H10O DBE = 8 + 1 - 10/2 = 4
Q: Identify the compound (C,H1|NO2) that would give the spectral data (IR, ³C NMR and 'H) below. The…
A: Combined spectroscopic problems are relatively easy to solve as you can confirm the observation from…
Q: ) Propose the possible substitution patterns for each of the derivatives based on the observed…
A: A) Based of splitting pattern, we can say that Derivative 1 1CH2 = 2CHX The hydrogen of carbon(1)…
Q: Compound C has the molecular formula C5H8O. The IR, 1H, 13C, and DEPT NMR spectra of this compound…
A: It is given that the molecular formula of a compound is C5H8O and its NMR, IR and DEPT predictions…
Q: The following IR spectrum corresponds to which compound? Explain. Tranamittance, %
A: Given IR data : 2969 cm-1 belongs to sp3 C-H Stretch of alkane 1715 cm-1 belongs to C=O stretch of…
Q: Compound A (C1,H14) shows prominent peaks in its mass spectrum at m/z 134 and 91. Compound B (also…
A: The double bond equivalent of compound A and compound B is same. The double bond equivalent of the…
Q: What is the structures of the compounds with the NMR data below?
A: Answer: The name of C16H18O6S2 is 1,2-Bis(tosyloxy)ethane OR Ethane-1,2-diyl…
Q: cyclohexanone 1-methylcyclopentanecarbaldehyde
A: We know that the 1H- NMR spectroscopy predicts the number of chemically distinct protons present in…
Q: Which method, polarimetry or NMR spectroscopy, gives the more accurate results in this experiment?…
A: In this case, we are attempting to determine the degree of optical purity. NMR spectroscopy will be…
Q: Compound D has the molecular formula C5H8O. Assign all peaks in 1H and 13C NMR spectrum of compound…
A: The given molecular formula is C5H8O Data given for 1H NMR spectra are given below: 2 Hydrogens have…
Q: 3H, t 1Η ΝMR 2H, q 1H,broad Disappears when 1 drop of D20 was added. 12 11 10 8. 6. 4 2
A: The correct structure of the above spectral data is propanoic acid. this is mostly confirmed by the…
Q: Give the structure for two isomers of molecular formula C4H10O which are consistent with the ^1H-NMR…
A: Note : compound b is isobutyl alcohol. It is having 4 signals. Compound c is having diethyl ether…
Q: Below is a zoomed in portion of a 13C{'H} NMR spectrum of the quaternary carbons of Compound 1. The…
A: In this example there is C-F and C-P coupling Multiplicity = (2nI + 1) Where, I = nuclear spin…
Q: Set 1: cyclohexanone 1-methylcyclopentanecarbaldehyde
A: The given molecule are ketone and aldehyde respectively. Hence we can differentiate between them…
Q: When a sample of the compound below is analyzed by mass spectrometry, the molecular ion will appear…
A: Mass spectrometry is used to find out the molecular weight of the compound. In this technique sample…
Q: A B (a) Identify which of the structures (A and B) gives to the *H NMR spectrum below. Explain your…
A: The instrumental method which is used to determine the structure of chemical compound using…
Q: Calculate the IHD and explain (elucidate) the structure using the 1H and 13C NMR data.
A:
Q: . Deduce a possible structure for the compound with the IR absorptions below. а. С3H5N: 2950, 2250…
A: IR spectra: Infrared spectra used to determine functiona groups in compounds Based on stretching…
Q: Please provide the structure of a compound with the molecular formula C8H8O2 using the IR, 1H-NMR.…
A: The question is based on concept of molecular Spectroscopy. we have to have identify IR and NMR…
We’ll answer the first question since the exact one wasn’t specified. Please submit a new question specifying the one you’d like answered.
Step by step
Solved in 2 steps with 2 images

- a) Interpreted the analytical data reported, you should include reference to the IR, and NMR chemical shifts, integration and observed splitting patterns?b) Identify the compound which corresponds to the unknown, and provide a reason?c) Provide reasons why the other 8 structures do not fit with the reportedanalytical data?Part 3B Set 1. Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. Predict the 1H NMR spectrum for each compound (include integration, multiplicity, and approximate chemical shift). Either draw the actual spectrum or put in a data table format.The HNMR , CNMR , 2D NMR SPECTRA OF COMPOUND M1 ARE ATTACHED. WHAT IS STRUCTURE OF IT ? IS IT SYMMETRICAL REGARDS TO ITS CNMR SPECTRUM ? WHAT IS THE STRUCTURE SUGGESTION AND WHY ?
- Please provide a structure consistent with the following IR, 13C NMR, and 1H NMR spectra. Assign at least two bands in the IR and assign all protons in the 1H NMR/13C NMR spectrum. Show calculation of degree of unsaturation and explain your reasoning.Provide a detailed analysis of IR, Mass, 1H NMR and 13C NMR data for the following compound.Provide the structure or SMILES of the compound with molecular formula C8H8O and correlates with the NMR shown in the image
- (a) Compound A has molecular formula C9H18O but shows only one singlet in the 1H-NMR spectrum. Suggest a structure for A and explain your reasoning. (b) Compound B has molecular formula C10H14. The IR, mass, 1H-NMR, and 13C-NMR speca are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Suggest a structure for B and explain your reasoning.Shown on the next two pages are the mass, IR and NMR spectra for anunknown organic molecule. Based on the spectra provided, determine thestructure of the molecule. You do not need to assign spectral peaks.Remember to ignore the 13 C NMR triad at 77 ppm that comes from the NMRsolvent.Please give the correct answer to the following NMR spectroscopy question and provide detailed solutions of the answer.
- Analyze the 1H NMR data and talk about the coupling, splitting pattern, equivalents, and the j coupling, and the hydrogensThe molecular weight of the compound is 126. Deduce its structure based on molecular weight. HNMR,IR, and 13C NMR data provided..Give the structure for two isomers of molecular formula C4H10O which are consistent with the ^1H-NMR spectra shown below.

