For each of the following pairs, indicate the stronger and weaker acid by dragging the appropriate label under the structure. Drag the appropriate label to their respective targets. • View Available Hint(s) Reset Help CH,CH,CH,–H or CH,CH,NH–H CH,CH̟O–H or CH,CH,NH–H or CH,CH,CH,-H CH,CH,NH-H or F-H Stronger acid F-H Weaker acid CH̟CH,O-H or CH,CH,N–H H. H.
For each of the following pairs, indicate the stronger and weaker acid by dragging the appropriate label under the structure. Drag the appropriate label to their respective targets. • View Available Hint(s) Reset Help CH,CH,CH,–H or CH,CH,NH–H CH,CH̟O–H or CH,CH,NH–H or CH,CH,CH,-H CH,CH,NH-H or F-H Stronger acid F-H Weaker acid CH̟CH,O-H or CH,CH,N–H H. H.
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter4: Polar Bonds, Polar Reactions
Section: Chapter Questions
Problem 23E: Summarize the relationship between pKa and acid strength by completing the following sentences: a....
Related questions
Question
For each of the following pairs, indicate the stronger and weaker acid by dragging the appropriate label under the structure.
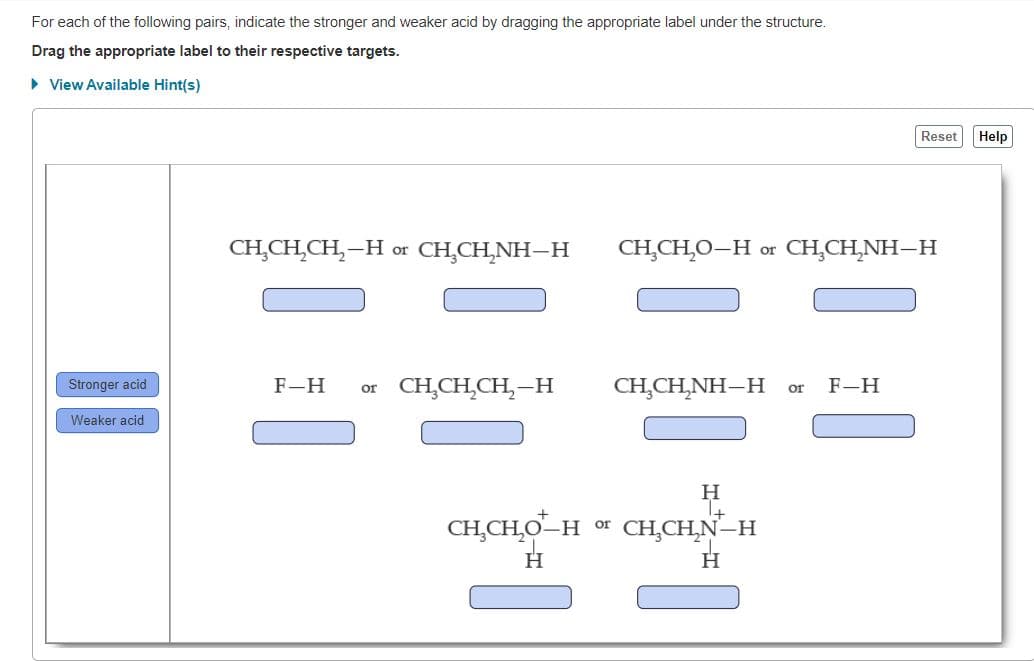
Transcribed Image Text:For each of the following pairs, indicate the stronger and weaker acid by dragging the appropriate label under the structure.
Drag the appropriate label to their respective targets.
• View Available Hint(s)
Reset
Help
CH,CH,CH,–H or CH,CH,NH–H
CH,CH̟O–H or CH,CH,NH–H
F-H
or CH,CH,CH,-H
CH,CH,NH–H
F-H
Stronger acid
or
Weaker acid
CH̟CH,O–H or CH,CH,N–H
H.
H.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning