For the following cyanine dye molecule, particle-in-a-box (PIB) theory works well, H₂C CH3 Based on the PIB modeling, which of the following statements is (are) correct? 1. The box length is taken as the distance between the two nitrogen atoms. II. The length of chain between the atoms nitrogen could not be estimated using number of carbon atoms in the conugated system. III. The carbon atoms in the fused rings disrupt the conjugated linear system of PIB OA. All the three statements are correct B. Both I and III are correct C. Both II and III are correct O D.Only I is correct O E. Only III is correct
For the following cyanine dye molecule, particle-in-a-box (PIB) theory works well, H₂C CH3 Based on the PIB modeling, which of the following statements is (are) correct? 1. The box length is taken as the distance between the two nitrogen atoms. II. The length of chain between the atoms nitrogen could not be estimated using number of carbon atoms in the conugated system. III. The carbon atoms in the fused rings disrupt the conjugated linear system of PIB OA. All the three statements are correct B. Both I and III are correct C. Both II and III are correct O D.Only I is correct O E. Only III is correct
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter4: Introduction To Quantum Mechanics
Section: Chapter Questions
Problem 38P: Using a simple particle-in-a-box model for the multiple bonding in 1,2-butadiene (see Example 4.7)...
Related questions
Question
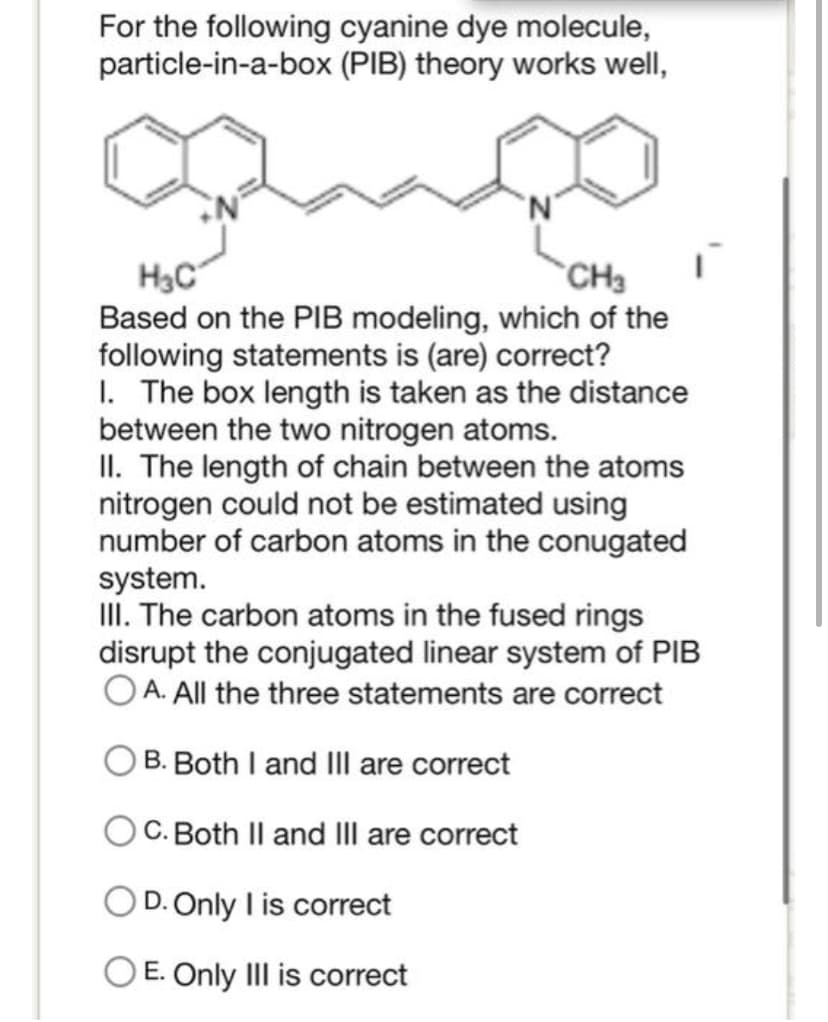
Transcribed Image Text:For the following cyanine dye molecule,
particle-in-a-box (PIB) theory works well,
H₂C
CH3
Based on the PIB modeling, which of the
following statements is (are) correct?
1. The box length is taken as the distance
between the two nitrogen atoms.
II. The length of chain between the atoms
nitrogen could not be estimated using
number of carbon atoms in the conugated
system.
III. The carbon atoms in the fused rings
disrupt the conjugated linear system of PIB
OA. All the three statements are correct
B. Both I and III are correct
C. Both II and III are correct
O D.Only I is correct
O E. Only III is correct
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,