For the following reaction, 6.75 grams of barium hydroxide are mixed with excess hydrochloric acid. The reaction yields 7.02 grams of barium chloride. hydrochloric acid (aq) + barium hydroxide (aq) barium chloride (aq) + water (1) What is the theoretical yield of barium chloride ? grams What is the percent yield of barium chloride ? %
For the following reaction, 6.75 grams of barium hydroxide are mixed with excess hydrochloric acid. The reaction yields 7.02 grams of barium chloride. hydrochloric acid (aq) + barium hydroxide (aq) barium chloride (aq) + water (1) What is the theoretical yield of barium chloride ? grams What is the percent yield of barium chloride ? %
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 56A
Related questions
Question
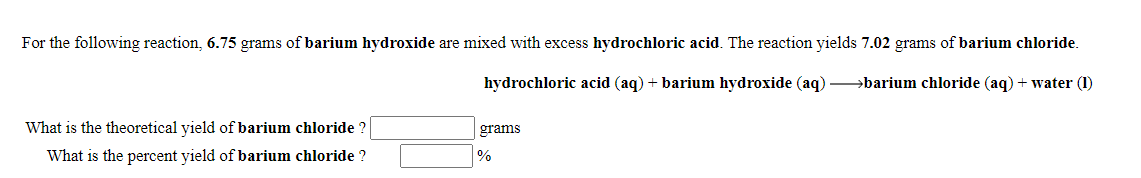
Transcribed Image Text:For the following reaction, 6.75 grams of barium hydroxide are mixed with excess hydrochloric acid. The reaction yields 7.02 grams of barium chloride.
hydrochloric acid (aq) + barium hydroxide (aq) barium chloride (aq) + water (1)
What is the theoretical yield of barium chloride ?
grams
What is the percent yield of barium chloride ?
%
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co