Four voltaic cells are set up. In each, one half-cell contains a standard hydrogen electrode (SHE). The second half-cell is one of the following. ,3+ Al"" (aq, 1.0 M) | Al(s) 2+ Cu (aq, 1.0 M) | Cu(s) 3+ • Ga (aq, 1.0 M) | Ga(s) 2+ • Ca (aq, 1.0 M) | Ca(s) (a) In which of the voltaic cells does the hydrogen electrode serve as the cathode?. O SHE & Al" (aq, 1.0 M) | Al(s) 2+ O SHE & Cu (aq, 1.0 M) | Cu(s) 3+ O SHE & Ga"" (aq, 1.0 M) | Ga(s) O SHE & Ca 2+ (aq, 1.0 M) | Ca(s) (b) Which voltaic cell produces the highest voltage? Which produces the lowest voltage? The highest voltage will be produced by combining the SHE and the - half-cell. The lowest voltage will be produced by combining the SHE and the v half-cell.
Four voltaic cells are set up. In each, one half-cell contains a standard hydrogen electrode (SHE). The second half-cell is one of the following. ,3+ Al"" (aq, 1.0 M) | Al(s) 2+ Cu (aq, 1.0 M) | Cu(s) 3+ • Ga (aq, 1.0 M) | Ga(s) 2+ • Ca (aq, 1.0 M) | Ca(s) (a) In which of the voltaic cells does the hydrogen electrode serve as the cathode?. O SHE & Al" (aq, 1.0 M) | Al(s) 2+ O SHE & Cu (aq, 1.0 M) | Cu(s) 3+ O SHE & Ga"" (aq, 1.0 M) | Ga(s) O SHE & Ca 2+ (aq, 1.0 M) | Ca(s) (b) Which voltaic cell produces the highest voltage? Which produces the lowest voltage? The highest voltage will be produced by combining the SHE and the - half-cell. The lowest voltage will be produced by combining the SHE and the v half-cell.
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.18QAP
Related questions
Question
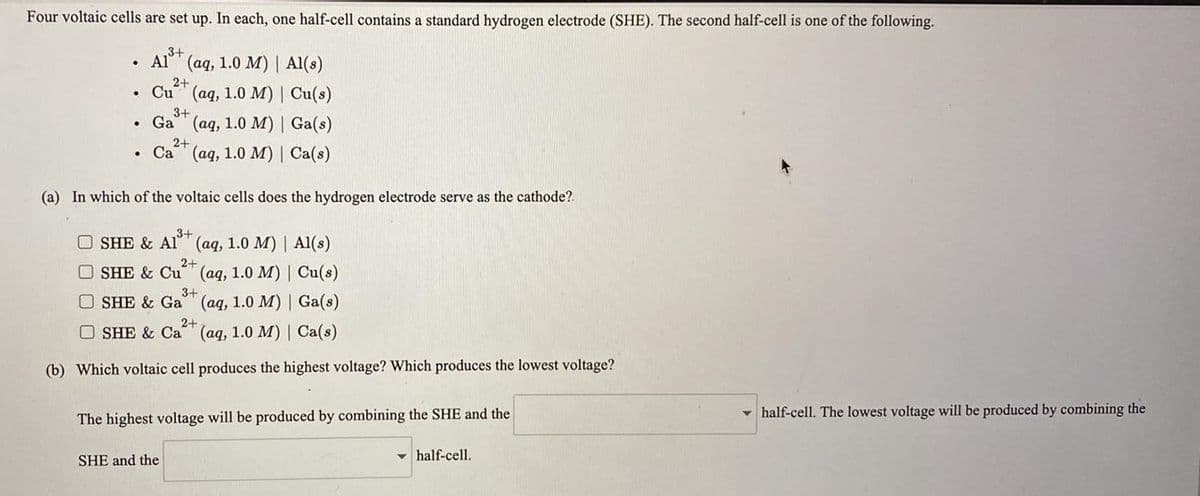
Transcribed Image Text:Four voltaic cells are set up. In each, one half-cell contains a standard hydrogen electrode (SHE). The second half-cell is one of the following.
3+
(aq, 1.0 M) | Al(s)
2+
Cu" (aq, 1.0 M) | Cu(s)
3+
Ga" (aq, 1.0 M) | Ga(s)
2+
Ca
(ад, 1.0 М) | Са(s)
(a) In which of the voltaic cells does the hydrogen electrode serve as the cathode?.
3+
O SHE & Al" (aq, 1.0 M) | Al(s)
2+
O SHE & Cu
(ад, 1.0 М) | Сu(8)
3+
O SHE & Ga (aq, 1.0 M) | Ga(s)
2+
O SHE & Ca (aq, 1.0 M) | Ca(s)
(b) Which voltaic cell produces the highest voltage? Which produces the lowest voltage?
The highest voltage will be produced by combining the SHE and the
- half-cell. The lowest voltage will be produced by combining the
SHE and the
half-cell.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning