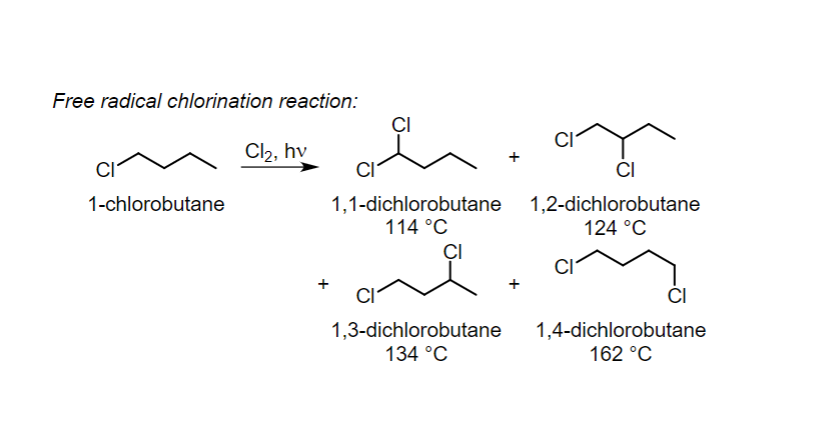
Free radical chlorination reaction: 1-chlorobutane Cl₂, hv CI 1,1-dichlorobutane 114 °C CI CI 1,3-dichlorobutane 134 °C CI 1,2-dichlorobutane 124 °C CI 1,4-dichlorobutane 162 °C
Q: Problem 4. A recent synthesis required the following transformation. Provide a synthetic sequence.…
A: In the given problem we need to provide a synthetic sequence to synthesie the desired from the given…
Q: 1.72. Calculate the average grade using equation 1.32 if the scores on a 10-point quiz are 5, 5, 5,…
A: Since, Average of the given sample or data is calculated as sum of all the data divided by the…
Q: If 0.105 moles of NaCl are dissolved in a 250.0 mL solution, what is the concentration of the NaCl…
A: Molarity: The molarity of the solution can be defined as the number of moles of the solute present…
Q: A gas is at 1.3 atm and 45.1°C. What is the concentration of the gas in units of mol per liter…
A:
Q: What mass of H₂ gas is produced during the electrolysis of water if a current of 12.7 A is used for…
A: Given, 2H+(aq) + 2e- ----> H2(g) Current (I) = 12.7 A Note: 1 min = 60 s Time (t) = 30 min = 30…
Q: Draw a reasonable mechanism for this reaction: O: H Но O -H -ÖH -H -ÖH :O: H+ H HÖ- H -он -H -ӧн…
A: The 5th hydroxy group attacks the carbonyl group of the given fisher projection and followed by…
Q: Please indicate if it will also be an endo or exo product or both (50:50) and why.
A: Diels-Alder reaction: The Diels–Alder reaction is a reaction between a conjugated diene and a…
Q: What is the E-factor for the reaction shown? The product is isolated by filtration with no…
A: Solution- The density of ethyl L-lactate is 1.03 g/mL. What is the E-factor for the given reaction…
Q: 12 - -2-2-2-2-2--2, Br 12
A: This question is related to IUPAC nomenclature. IUPAC stands for International union of pure and…
Q: How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘C to water at 25.0…
A: Mass of steam = 18.0 g (one mole) Initial temperature of the steam = 100oC Final temperature of…
Q: Match the following aqueous solutions with the appropriate letter from the column on the right.…
A: with increasing the number of particles or ions, depression of freezing point occurs. Thus solution…
Q: The decomposition of a generic diatomic element in its standard state is represented by the equation…
A: According to the question, The standard Gibbs free energy is given by = ∆G° = 5.09 kJ/mol The…
Q: Humidity is the percentage of partial pressure of water in air over the total possible water in the…
A: Introduction Humidity is an important concept in chemistry. It refers to the amount of water vapor…
Q: Gaseous ethane (CH,CH₂) will react with gaseous oxygen (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: Consider these hydrocarbons: Butane, Hexane, Methane, Propane First look up the formulas, and write…
A: Introduction Hydrocarbons are organic compounds composed of only hydrogen and carbon atoms. They are…
Q: aqueous layer OH (acid) "HCI layer" + dilute NaOH (aq) solution + NaCl (aq) (neutral) (base) +…
A: In aqueous solution, ionic compounds are mostly soluble where in organic layer organic compounds are…
Q: What is the bond angle between the 2px, 2py, and 2pz atomic orbitals? What would be the allowable…
A: There are three 2p atomic orbitals for an atom. They have the same shape but have different…
Q: an dentify the following properties as being intensive (I) or extensive (E) of a substance. - The…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: Please answer the following study question: Please describe and compare three types of batteries…
A: A battery is a device that stores and provides electrical energy through a chemical reaction.
Q: You have a sample of 121.5 g of C₂H6. How many moles of C₂H6 are in the sample?
A:
Q: The activation energy, Ea, for a particular reaction is 37.8 kJ/mol. If the rate constant at 280 K…
A:
Q: Q/A transparent medium has a refractive index of 1.5 and the wavelength of the magnetic ray passing…
A: Given data is Refractive index of the transparent medium=1.5 The wavelength of the magnetic ray…
Q: 4. COOH COOH
A: The given reaction is Diels-Alder reaction.this is also called (4+2) cycloaddition reaction.
Q: B LOH A OH NaH PBr3 C D F (a) Name the functional groups in compounds A, B and F, including, if…
A: A functional group is a specific arrangement of atoms within a molecule that is responsible for the…
Q: Kp for the following reaction at a give temperature is 0.263 at 1000 K. C(s) + 2 H₂(g) CH4(g) What…
A:
Q: Use the data to determine the entropy change ASrxn for this process: NH3(aq) + HOCl(aq) = NH₂Cl(aq)…
A: Given -> NH3(aq) + HOCl(aq) ---> NH2Cl(aq) + H2O(l)
Q: In the following reaction, oxygen is the excess reactant. SiCl4 + O2 → SiO2 + Cl2 The table shows…
A: Given that: For trial 1, Mass of SiCl4 = 120 g Mass of O2 = 240 g Actual yield = 38.2 g For trial 2,…
Q: 21. How many total atoms are present in 1.60 mol of sulfur dioxide? A) 9.63 X 1023 B) 1.54 X 1023 C)…
A: Since, Moles is the ratio of mass to the molar mass of that atom or molecules. Also, Number of atoms…
Q: H-Br Br H 1.2 Br 1,4 H
A: A 1,4-addition is also called a conjugate addition. Here the nucleophile adds to the carbon which is…
Q: Use the initial rates method with the given data to determine the rate law for the hypothetical…
A:
Q: At 500 C, cyclopropane (CaHe) reamanges to propene (CH₂-CH=CH₂). The reaction is first order, and…
A: Given, The rate constant of the reaction (k) = 6.7×10-4 S-1 The initial concentration of C3H6 =…
Q: N₂04(9) 2 NO₂(9) Three experiments were run starting with different initial amounts of N₂04(9)…
A: Answer: In the given problem on the basis data provided for the dissociation reaction of N2O4 into…
Q: e) Using your answers to calculations b and d and the above chemical equation, determine the mole…
A: Answer: When hydrated salt of magnesium sulfate is heated then it loses the water molecule and forms…
Q: Propose an efficient synthesis for the following transformation: The transformation above can be…
A: Answer:- This question is answered by using the simple concept of chemical reactions of organic…
Q: Evaluate the specific rate constant for the reaction at the temperature for which the data were…
A:
Q: 0.700 mol Na₂SO STARTING AMOUNT 0.700 mol Na₂SO ADD FACTOR x( ) 0.700 0.0833 mL Convert the…
A: Conversion factor - When we have to convert one unit to another unit we must use conversion factor.…
Q: h) CHCI3 KOtBu 1 enantiomer (1 chiral compound) 1:1 mix of enantiomers mixture of diastereomers…
A: MIXTURE OF DIASTEREOMER.
Q: 02A: 2.1-2.5 and Accuracy Handout (70 min) culate Figures of Merit for Trueness ▾ Part A. Absolute…
A: Given data is The concentration of standard ethyl alcohol sample = 0.174 mg/L The average value of…
Q: W Exercices Calculate the solution of 0.01 kildgram of Chlovide in 250 grams of boiling point of a…
A: In the multiple questions, we solve only first question according to the Bartleby guidelines. Check…
Q: The decomposition of N2O5 when heated in carbon tetrachloride solvent is a second order reaction.…
A:
Q: For the image attached make a graph of the average mass vs year. Make sure axes are labeled
A: Solutions- We have to plot the graph of the average mass vs year.
Q: The vapour pressures of the pure components P and Q are 700 Torr and 500 Torr, respectively. When…
A: Given: Vapor Pressure of Pure P = 700 torr Vapor Pressure of Pure q = 500 torr Two phases are in…
Q: 6. Consider the chemical equation: CO2 + H₂O → C6H12O6 + 02 To balance this equation, place…
A: Balanced chemical equation: A balanced chemical equation is one in which the number of atoms on the…
Q: Which functional category CANNOT be an ester's isostere? carbamate. amide urethane. pyrrole.
A: isostere groups have same type of physical and chemical properties. They have same number of…
Q: alcohol. A carbocation intermediate is central to which of these reaction types? (circle) E1 E2…
A: In bimolecular reactions the attack of nucleophiles (in case of SN2) or base (in case of E2) and…
Q: The mass percentage of hydrochloric acid within a solution is 10.00%. Given that the density of this…
A: The given data is The mass percentage of hydrochloric acid with in a solution (mm×%) = 10.00%. The…
Q: 9- Give one example for the hydration of Alkene: That is the Addition of H2O by oxymercuration. Give…
A: 9) The oxymercuration reaction is an electrophilic addition that transforms an alkene into a neutral…
Q: You mieht need: F Calcula How many solutions does the following cquation have? -6y+13+9y=8y-3 Choose…
A: The equation given is as follows, -6y + 13 + 9y = 8y - 3
Q: A KClKCl solution containing 41 gg of KCl per 100.0 g of water is cooled from 70 ∘C to 0 ∘C. What…
A: A saturated solution is one in which a solvent has completely dissolved all of the solutes presents.…
Q: D. Give the quantum numbers for the last electron of each element given in Part II following the…
A: This question is related to quantum numbers. In the given question, five elements are given and it…
Draw the initiation and propagation steps for the formation of 1,3-dichlorobutane in this reaction.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

- Chemical Reactions and Mechanisms: Use the space below for reactions and mechanisms. Initiation Propagation Termination (3) Toluene 1° Benzylic1,1-dichloroethane is the product of free radical substitution reaction of ethane. Show the overall mechanism including initiation, propagation and termination steps.( answer asap with explanation in handwritten)
- As in the free-radical halogenation of alkanes, chlorination of alkylbenzenes is less selective than bromination. Given the relative rates per hydrogen for hydrogen atom abstraction from 1-phenylbutane by chlorine for the elementary step shown, calculate the percentage of 1-chloro-1-phenylbutane in the C10H13Cl product.Calculate ▲S°rxn for the following reaction. the S° for each species is shown below in the reactionGive mechanism of this reaction ASAP.
- Identify this mechanistic step in a radical halogenation reaction. A) initation B) propagation C) terminationAcetic anhydride reacts with water in the following manner: CH3C(O)-O-C(O)CH3 + H2O = 2CH3CO2 If this reaction is carried out in an inert solvent with low concentrations of both acetic anhydride and water, it follows the rate law rate = k [CH3C(O)-O-C(O)CH3] [H2O] with a rate constant of 0.0035 s-1. However, if low concentrations of acetic anhydride are placed in pure water, it follows the rate law rate = K [CH3C(O)-O-C(O)CH3] where K = 0.1944 s-1. Explain why this is happening.a. Give the letter(s) corresponding to the transition state(s). ____________ b. Give the letter(s) corresponding to the reactive intermediate(s). _________
- Define mechanism of haloform reaction ?Draw a reaction-energy diagram for the propagation steps of the free-radical addition of HBr to isobutylene. Draw curves representing the reactions leading to both the Markovnikov and the anti-Markovnikov products. Compare the values of ∆G° and Ea for the rate-limiting steps, and explain why only one of these products is observed.Define the situation when Is the Mechanism SN1 or SN2?

