Functional Soluble Soluble in Soluble in Soluble in Specific Compound Group in Water? 1 M HCI? 1 M NaOH? 1 M NaHCO3? Amine & m-Aminoacetophenone No Yes No No Carbonyl Cholesterol OH Group No No No No 4-t-Butylphenol ОH Group No Yes No No Benzoic acid COOH Group No No Yes Yes
Functional Soluble Soluble in Soluble in Soluble in Specific Compound Group in Water? 1 M HCI? 1 M NaOH? 1 M NaHCO3? Amine & m-Aminoacetophenone No Yes No No Carbonyl Cholesterol OH Group No No No No 4-t-Butylphenol ОH Group No Yes No No Benzoic acid COOH Group No No Yes Yes
Chapter23: Carbonyl Condensation Reactions
Section23.SE: Something Extra
Problem 35MP: Isoleucine, another of the twenty amino acids found in proteins, is metabolized by a pathway that...
Related questions
Question
Q⁴
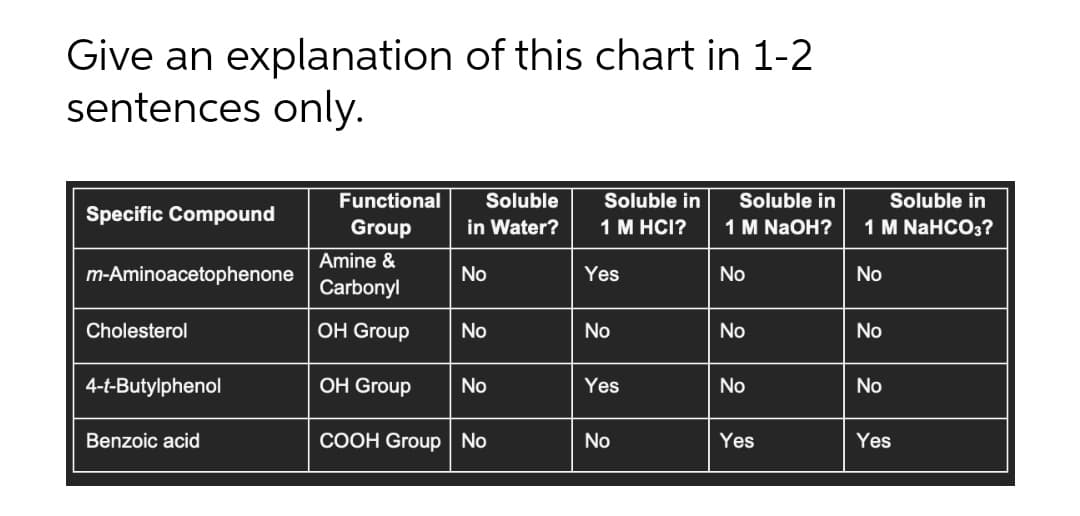
Transcribed Image Text:Give an explanation of this chart in 1-2
sentences only.
Functional
Soluble
Soluble in
Soluble in
Soluble in
Specific Compound
Group
in Water?
1 M HCI?
1 M NaOH?
1 M NaHCO3?
Amine &
m-Aminoacetophenone
No
Yes
No
No
Carbonyl
Cholesterol
ОH Group
No
No
No
No
4-t-Butylphenol
ОH Group
No
Yes
No
No
Benzoic acid
COOH Group No
No
Yes
Yes
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

