Gibbs energy change, A:G®, is related to cell potential, E, by the equation Calculate the standard Gibbs energy change at 25 °C for the following reaction: A;G° = -nFE Mg(s) + Fe2+(aq)→Mg²+(aq) +Fe(s) Express your answer to three significant figures and include the appropriate units. where n is the number of moles of electrons transferred and F = 96 500 C mol- is the Faraday's constant. > View Available Hint(s) HẢ ? A,G" = 370.56 kJmol-1 Submit Previous Answers Part B Calculate the standard cell potential at 25 °C for the reaction X(s) +2Y*(aq)¬→X²*(aq) +2Y(s) where ArH° = -729 kJ mol- and A,S° = -325 JK' mo]1 Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) Eo = Value Units
Gibbs energy change, A:G®, is related to cell potential, E, by the equation Calculate the standard Gibbs energy change at 25 °C for the following reaction: A;G° = -nFE Mg(s) + Fe2+(aq)→Mg²+(aq) +Fe(s) Express your answer to three significant figures and include the appropriate units. where n is the number of moles of electrons transferred and F = 96 500 C mol- is the Faraday's constant. > View Available Hint(s) HẢ ? A,G" = 370.56 kJmol-1 Submit Previous Answers Part B Calculate the standard cell potential at 25 °C for the reaction X(s) +2Y*(aq)¬→X²*(aq) +2Y(s) where ArH° = -729 kJ mol- and A,S° = -325 JK' mo]1 Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) Eo = Value Units
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 3RQ: Table 17-1 lists common half-reactions along with the standard reduction potential associated with...
Related questions
Question
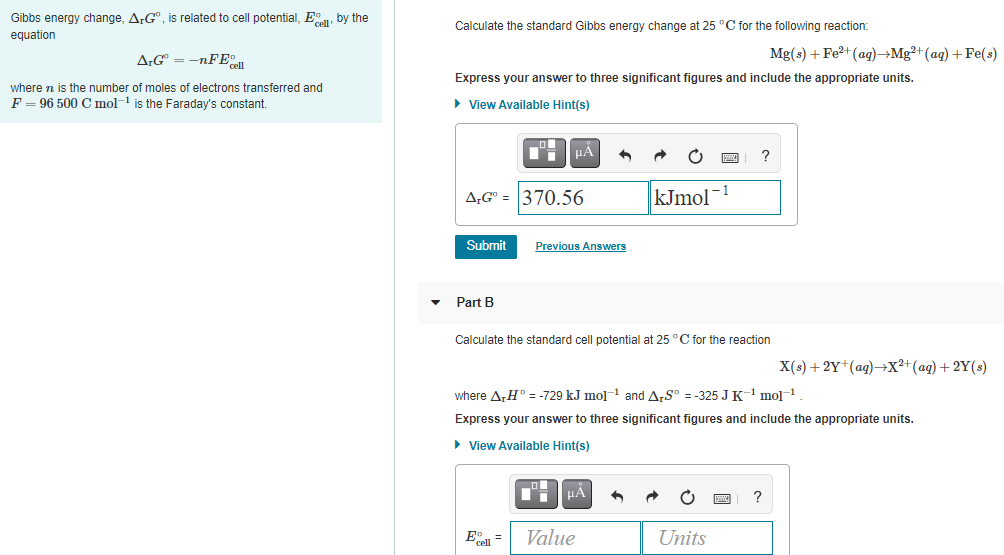
Transcribed Image Text:Gibbs energy change, A;G", is related to cell potential, E, by the
Calculate the standard Gibbs energy change at 25 °C for the following reaction:
equation
A:G = -nFE
Mg(s) + Fe?+(ag)→Mg²+(ag) + Fe(s)
Express your answer to three significant figures and include the appropriate units.
where n is the number of moles of electrons transferred and
F = 96 500 C mol-1 is the Faraday's constant.
• View Available Hint(s)
kJmol
- 1
A,G" = 370.56
Submit
Previous Answers
Part B
Calculate the standard cell potential at 25 °C for the reaction
X(s) + 2Y*(ag)→X²+(ag) + 2Y(s)
where A,H° = -729 kJ mol-1 and ArS" = -325 JK' mol1
Express your answer to three significant figures and include the appropriate units.
• View Available Hint(s)
E =
Value
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning