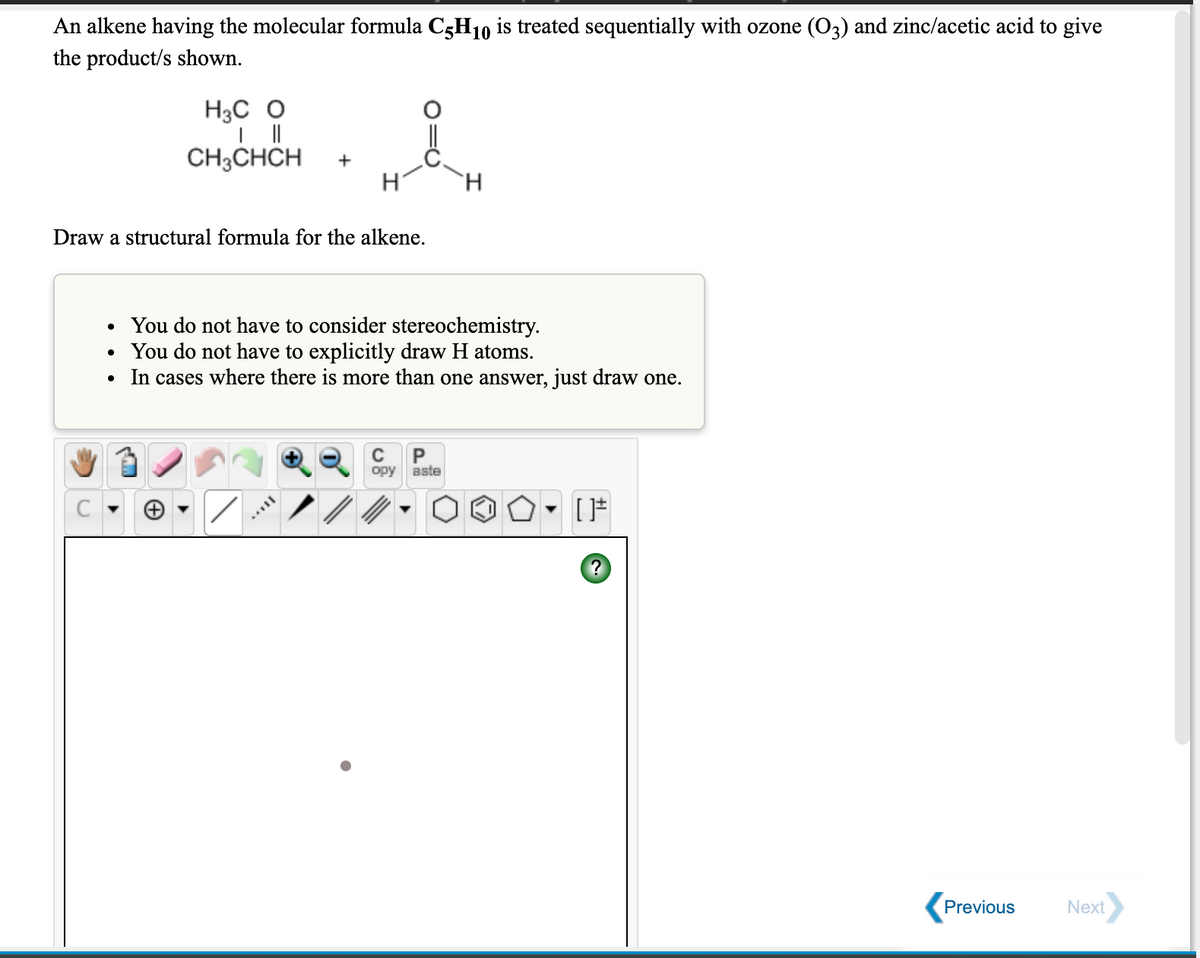
An alkene having the molecular formula C3H10 is treated sequentially with ozone (O3) and zinc/acetic acid to give the product/s shown. H3C O CH3CHCH + H H. Draw a structural formula for the alkene.
Q: For each compound, give the product(s) expected from (1) HgSO4>H2SO4@catalyzedhydration and (2)…
A: The given alkyne is hex-3-yne. The alkyne on hydration in the presence of HgSO4/ H2SO4 gives…
Q: Draw structural formulas for all alkenes that could be used to prepare the alcohol shown below by…
A: ->In oxymercuration-demercuration overall morkovnikov addition of H2O takes place.
Q: Which of the following alcohols undergoes the most rapid dehydration upon treatment with H 2SO 4 to…
A: Dehydration of alcohol: The reaction of alcohol in presence of acid produces alkene. It is an…
Q: Propose a mechanism for formation of product (1), and account for its configuration.
A:
Q: НС енз т CH-CH=CH₂₂ нз' Br₂/cala (Bromination) Вр CH₂ CH CH - Br Dehalogenation "1 енз CH CE CH CH₂…
A:
Q: The alkene shown below is treated sequentially with ozone (O3) and zinc/acetic acid. Draw structural…
A: The above reaction is addition of ozone referring as ozonolysis.
Q: Which reagent is appropriate for the following reaction yclohexene converted to trans-cyclohexane…
A:
Q: Draw structural formulas for all alkenes that could be used to prepare the alcohol shown below by…
A: in oxymercuation reaction, more substituted alchol is formed.
Q: Which of the following compounds will undergo an S,2 reaction most readily? Select one: O a.…
A: 1. Primary alkyl halides more reactive than secondary and tertiary. Compound d is primary alkyl…
Q: In each case below select the synthetic procedure's that could be used to carry out the…
A: The addition of oxygen atom is called oxidation or we can say that the removal of the hydrogen atom.…
Q: Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula C10H16,…
A: The structure of 2,6-dimethyloctane is as follows:
Q: Which of the following alkenes gives rise to the alcohol shown below as the major product of…
A: Conversion of alkene to alcohols is done by dehydrohalogenation reaction. It occurs by the net…
Q: If 1-bromopentane is heated in acetone containing NaOH, what is the alkane produced? Draw and…
A: Here 1- bromopentane is heated in acetone containing NaOH. Here acetone is a polar aprotic solvent ,…
Q: How many alkene products, including E, Z isomers, might be obtained by dehydration of…
A:
Q: 10. Classify the following alkenes as E or Z: CH3 Cilz0H • CH2 Br Citz0 NHz
A: The absolute configuration of alkenes are substituent dependent. The arrangement of substituents of…
Q: An alkene having the molecular formula CH20 is treated sequentially with ozone (O3) and zinc/acetic…
A:
Q: 1. Deduce the structure of each compound from the information given. All unknowns in this problem…
A: The organic reaction in which the unsaturated bonds of alkynes, alkenes and azo compounds are break…
Q: OH 1. Hg(OAc)2, H2O 2. NABH4 CH3 .CH3 H2C° H3C d-catalyzed addition of water to an alkene yields an…
A:
Q: A certain hydrocarbon, C5H6, yields the two products shown below upon ozonolysis with 1. O3/CH2Cl2…
A: The molecular formula of the given compound is C5H6. The two products obtained upon ozonolysis are:…
Q: The Claisen condensation converts two molecules of an ester into a ß-keto ester. The reaction starts…
A: Claisen condensation reaction takes place between two ester molecules when treated with a strong…
Q: Benzene reacts with CH3CH2CI in the presence of AICI3 to form ethylbenzene (Friedel- Crafts…
A:
Q: One compound that contributes to the “seashore smell” at beaches in Hawai‘i is dictyopterene D', a…
A:
Q: How many alkenes could you treat with H2, Pd/C to prepare methylcyclopentane?
A: In order to prepare methylcyclopentane by catalytic reduction 4 alkenes are requires. Alkenes with…
Q: 1-Pentene reacts with H+ and H2O to give the product ______.
A:
Q: An alkene having the molecular formula C11H20 is treated sequentially with ozone (O3) and…
A:
Q: Which alkene reacts with methanol in presence of acid catalyst to give the following ether? CH3…
A:
Q: major
A:
Q: ОН 1. Hо(ОАc)2. Н20 2. NABH4 CH3 CH3 H2C H3C Acid-catalyzed addition of water to an alkene yields an…
A:
Q: Epoxidation is typically achieved by treating an alkene with: a. bromine b. NBS с. ТСРВА O d. BH3,…
A: Alkene reacts with each reagent differently to give different products. We will draw each product…
Q: For each compound, give the product(s) expected from (1) HgSO4>H2SO4@catalyzedhydration and (2)…
A: The expected product when the given reactant react with (1) HgSO4>H2SO4-catalyzed hydration and…
Q: An alkene with the molecular formula C 10H 18 is treated with ozone and then with zinc and acetic…
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: In each case below select the synthetic procedure/s that could be used to carry out the…
A: Both of these reagents are hydrating agent resulting in addition of -OH group.
Q: Draw structural formulas for the alkene(s) formed by treatment of each haloalkane or halocycloalkane…
A: The equation for the reaction of given haloalkane with sodium ethoxide (CH3CH2ONa) in ethanol…
Q: Complete the following reaction by drawing a structural formula for the product. + H2 Ni or Pt…
A: Ketones and aldehydes undergo reduction reaction with H2 in presence of transition metal catalysts…
Q: Draw structural formulas for the isomeric carbocation intermediates formed on treatment of each…
A: The reaction of an alkene with HCl will be proceed by the substitution mechanism. It will form one…
Q: Draw a structural formula for the product of each SN2 reaction. Where configuration of starting…
A:
Q: + N₂ + H₂O CH3 Ph CH3 The Wolff-Kishner reaction involves the reaction of an aldehyde/ketone with…
A:
Q: Two substitution products result from the reaction between 3-chloro-3-methyl-1- butene with sodium…
A:
Q: When 4-penten-2-one is reacted with aqueous NABH4 (sodium borohydride), the major substrate product…
A: Given Reactant 4-penten-2-one Reagent Aq NaBH4 Product To be determined
Q: An alkene having the molecular formula CgH12 is treated sequentially with ozone (O3) and zinc/acetic…
A: When alkenes are treated sequentially with ozone (O3) and zinc/acetic acid, they give carbonyl…
Q: CH, -CH=CCH=CHCH3 The Wittig reaction can be used for the synthesis of conjugated dienes. Propose a…
A: The wittig reagent and the aldehydes used to synthesize the double bond denoted b is given below
Q: Ph Ph- CH3 Ph CH3 The Wolff-Kishner reaction involves the reaction of an aldehyde/ketone with…
A: To solve this problem we have to draw the arrow pushing mechanism of given step .
Q: 2. Give the products when 1-methyl-2-deutero-cyclohexene is treated with the following reagents. Be…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three…
Q: OH 1. Hg(OAc)2, H2O 2. NaBH4 CH3 .CH3 H3C Acid-catalyzed addition of water to an alkene yields an…
A: Given reaction is oxymercuration- demercuration reaction of alkene.
Q: CH HCHS 2. NAEH,
A:
Q: Which of these compounds is obtained as the major product from the reaction of the following alkene…
A: The reaction between aqueous sulphuric acid and alkene takes place according to the Markonikove's…
Q: An alkene having the molecular formula CHs is treated sequentially with ozone (O3) and zinc/acetic…
A:
Q: CH3 CH;CHCHCHCH2CSCH
A: Given alkyne is : What organic product(s) would you expect when the following compound is…
Q: 1. Synthesize the following alkene through the Wittig reaction of a carbonyl compound and a Wittig…
A: 1
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Please tell the outcome of the addition of HBr to (a) trans-2-pentene, (b) 2-methyl-2-butene, and (c) 4-methylcyclohexene. Also please list how many isomers can be formed in each case.OChem HELP... Draw the conformational structures (chair or boat conformations) for the MAJOR product formed when 1-tert-butylcyclohexene reacts with each of the following reagents. Also, indicate if the product obtained is racemic form. a) Br2, CCl4 b) Br2, H2O c) OsO4, then aqueous NaHSO3 d) ICl e) mCPBA, then H3O+, H20 f) O3, then Me2S (conformational structure not required) g) BH3:THF, then H2O2, HO- h) D2, Pt i) Hg(OAc)2 in THF-H2O, then NaBH4, HO- j) BD3 :THF, then CH3CO2T(S)-2-Tosylpentane (stereochemistry not shown in the figure below) is converted to one of the stereoisomers of 2-bromopentane. Provide the formula of the reagent you would use to accomplish this transformation, and draw the correct stereoisomer product.
- Name the alkene below.Use only E/Z designators to indicate stereochemistry.Please predict the outcome of the addition of HBr to (a) trans-2-pentene, (b) 2-methyl-2-butene, and (c) 4-methylcyclohexene. Also please list how many isomers can be formed in each case? Thank youA chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions
- Most methods of making alkenes yield predominately the more stable isomer, usually the trans. Outline all steps in the conversion of a mixture of 75% trans-2-pentene and 25% cis-2-pentene into essentially pure cis-2-pentene.Which alkene from the choices can react with excess Hbr to yield the major product shown in the first photo?Like other electrophiles, carbocations add to alkenes to form new carbocations, which can then undergo substitution or elimination reactions depending on the reaction conditions. With this in mind, consider the following reactions of nerol, a natural product isolated from lemon grass and other plant sources. Treatment of nerol with TsOH forms α-terpineol as the major product, whereas treatment of nerol with chlorosulfonic acid, HSO3Cl, forms a constitutional isomer, α-cyclogeraniol. Write stepwise mechanisms for both processes. Each mechanism involves the addition of an electrophile—a carbocation—to a double bond.
- Like other electrophiles, carbocations add to alkenes to form new carbocations, which can then undergo substitution or elimination reactions depending on the reaction conditions. With this in mind, consider the following reactions of nerol, a natural product isolated from lemon grass and other plant sources. Treatment of nerol with TsOH forms α-terpineol as the major product, whereas treatment of nerol with chlorosulfonic acid, HSO3Cl, forms a constitutional isomer, αcyclogeraniol. Write stepwise mechanisms for both processes. Each mechanism involves the addition of an electrophile— a carbocation—to a double bond.Based on the more stable conformation (conformation A), need help providing two mechanisms for the E2 elimination of the protonated version of cis-1 using a general base (B:) to give alkenes 2 and 3. Clearly show which hydrogens meet the stereochemical requirements for E2-elimination Using Zaitsev’s rule, indicate which is favoured. Pictured is also a reaction mechanism to show what the alkenes 2 and 3 are. Thank you :)1)Chemistry students are taking an experimental course in organic chemistry at a public university. During an experiment involving conjugated dienes, some doubts arose when discussing the results obtained so far: (a) A student obtained two products from the reaction of 1,3-cyclohexadiene with Br2. His lab partner was surprised to get only one product from the reaction of 1,3 - cyclohexadiene with HBr. Explain these distinct results. (b) One student, seeing the discussion of colleagues, commented that she obtained two distinct products when reacting 1,3,5-hexatriene with HBr, with different yields just by changing the reaction temperature. Explain the results she obtained using reaction mechanism and based on kinetic and thermodynamic control involving conjugated dienes.



