Q: 3. Which conformation is lower in energy? Explain.
A: Answer is the one at right is at less energy we know that group at axial position is less stable,…
Q: For the molecule shown below, draw both chair conformations and circle which is the lower energy…
A:
Q: H3C CH,CH3 -H H3C H3C' А H3C B
A: Both the given conformations are eclipsed conformations. Eclipsed conformation in which two…
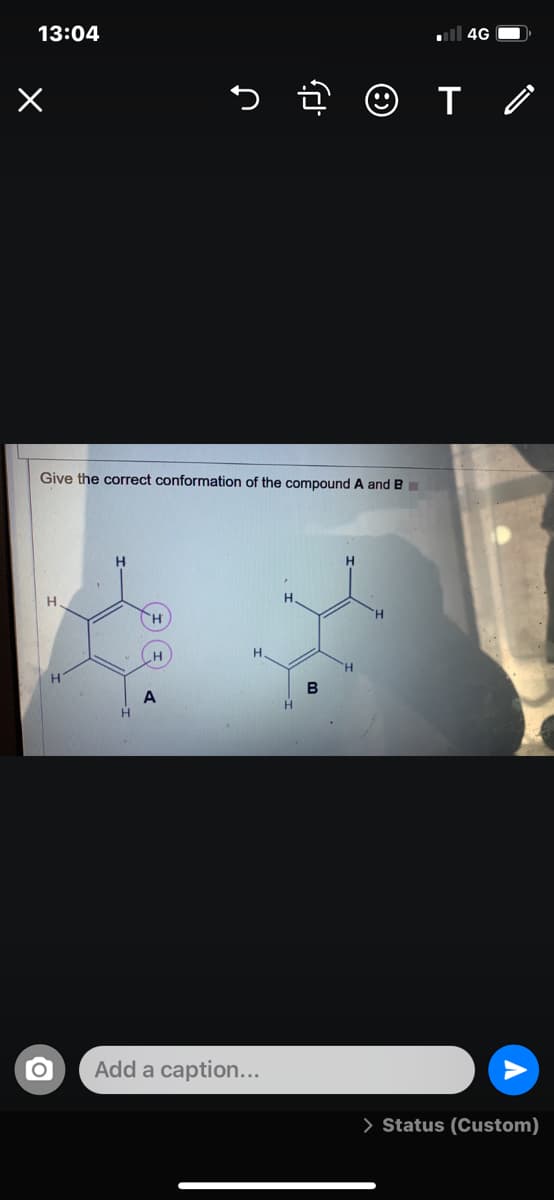
Q: give the most stable chair conformation given bleow structures
A: The structure of the cyclohexane molecule can be expressed in two conformations: chair and boat. The…
Q: a) How many degrees of unsaturation (double bond equivalents) are represented by the molecular…
A:
Q: Define conformation
A: Stereochemistry can be divided into two parts i.e. conformational and configurational.…
Q: Match the images with the correct conformation term. CH3 CH3 H,C. H,c CH, Answer Bank anti eclipsed…
A:
Q: Which of the following chair structures represents the most stable conformation of the…
A: If the substitutents are at 1 and 4 positions and cis to each other in cyclohexane, then the most…
Q: Look at the model of chloroethane. a) Do all chloroethane molecules spend all of their time in this…
A: Model of chloroethane
Q: cis-Decalin is a conformationally mobile molecule. a) Briefly explain what is meant by this…
A: Decalin consists of two cyclohexane rings fused together, i.e., the ring junctions are adjacent.…
Q: What is the difference in stability between the two staggered conformations?
A: The staggered conformations can be classified into gauche and anti conformation.
Q: a. b.
A: Constitutional isomers have same molecular formula but have different bonding patterns.
Q: What is the total number of hydrogen in the conformation below
A: Totel number of hydrogen in the given conformation is calculated below: Number of hydrogen molrcule…
Q: A disubstituted cyclohexane compound is given below in its chair conformation. Draw the…
A: The given compound contains two substituents on the cyclohexane ring. The chlorine and bromine…
Q: a. Complete the Newman projection to show the molecule in its lowest energy conformation (D). F b.…
A:
Q: CI- and CI
A: Write name of the given chair conformations=?
Q: Which conformation is lower in energy? Explain.
A: Which conformar is most stable that has lowest energy.
Q: which if the structure of trans-1,2-dimethylcyclopentane? (b) which is the most stable conformation…
A:
Q: How the relative stability of the two conformations of any disubstituted cyclohexane can be analyzed…
A: The relative stability between 2 conformers of any disubstituted cyclohexane are analyzed as…
Q: (i) Gauche conformation of ethane, (ü) felipsed conformation of ethene, (iii) Staggered conformation…
A:
Q: Among the figure, (a) what is the most stable conformation of n-pentane? (b) gauche conformation of…
A: Firstly we will see which structure is butanes and which structure is pentanes. A) Butane…
Q: Which molecule do you expect to be more stable, a or b)? Use both chair conformations of each…
A: In order to draw the chair conformation of the given cyclohexane molecule. First draw the chair form…
Q: Which of the following is the more stable chair conformation of the substituted cyclohexane shown in…
A: Given structure,
Q: Is the 1st compound ( more, less or equally) stable than B? Pls show through illustrations of chair…
A:
Q: Br
A:
Q: Please match the appropriate chair conformation to the compound given below;
A: All three groups are cis to each other . In chair form they should be cis to each other.
Q: Ethylene glycol is unusual in that the gauche conformation is more stable than the anti…
A: Ethylene glycol (IUPAC name: ethane-1,2-diol) is an organic compound containing the formula…
Q: which chair conformation is more stable and why?
A: Different conformations of cyclohexane are formed by the rotation around the carbon-carbon single…
Q: Convert the following Newman projection of compound J to a three dimensional line structure in the…
A:
Q: Discuss and compare two possible chair conformations of cis-1,3-dibromocyclohexane compound. Which…
A:
Q: conformer, however B is not. Please use Newman projection to of the substituted cyclohexane to draw…
A:
Q: Draw an alkene (minimum of 4 carbons) that is in the E conformation and a different alkene (also 4…
A: Let's consider our alkene is 3-methylpent-2-ene
Q: Which conformation of ethane is more stable?
A: There are infinite number of conformations of ethane. But there are two conformations which are…
Q: Br A. Redraw the bond-line structure of the molecule above on a separate sheet of paper. B. Draw a…
A: More syn diaxial strain more will be the energy and less will be the stability.
Q: , Which of the following compounds is more stable? To get credit you must draw the most stable chair…
A:
Q: The anti-conformation is the most stable n-butane conformation. If you could take a snapshot of a…
A: Anti-conformation of n-butane is the most satble conformation. To determine, for 1 mole of n-butane…
Q: a) State two (2) types of stereoisomer. b) Explain briefly, which compound hexane or…
A: a. Stereoisomerism is the arrangement of atoms in molecules whose connectivity remains the same but…
Q: ) Using chair conformation action that would take place when trans- n. (Show the most stable…
A: Trans-1-bromo-4-tert-butyl cyclohexane on reaction with iodide ions(I -) follows SN2 mechanism and…
Q: CH
A: We are given the two conformations of a compound. ...................................(1).…
Q: 1) Draw both chair conformations of the following compound
A: The stable configuration of cyclohexane is the one where the bulky groups are on equatorial…
Q: Please draw the two most stable conformations (chair conformatoin I beleive) and label the most…
A: A most stable chair conformation is possible when, There is no 1,3 diaxial interaction and, The…
Q: When you obtained a geometry optimized structure of methylcyclopentane, did you determine the energy…
A: The chemical name of an organic compound provides information about the functional groups present in…
Q: Explain why alkene A is more stable than alkene B, even though Bcontains more carbon atoms bonded to…
A: Given compounds,
Q: Label each attached compound as cis or trans. Then draw the second chair conformation.
A: This given compound is 1,2 axial/equatorial trans dichlorocyclohexane. Axial means above the plane…
Q: When you obtained a geometry optimized structure of methyleyclopentane, did you determine the energy…
A: Methylcyclopentane exist in two conformation. Cs Envelope and Cn Half chair.
Q: Which of compounds A-C would you expect to have the greatest heat of combustion? The smallest?…
A: Heat of combustion of the substance can be defined as the energy value or calorific value is the…
Q: Which isomer, A or B, would you expect to have the greater heat of combustion? Explain. A B
A:
Step by step
Solved in 2 steps with 1 images

- Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.a model of cyclohexane in a chair conformation, and explain why the names “axial” and“equatorial’ are appropriate.Two conformations of cis-l, 3-dimethylcyclobutane are shown. What is the difference between them, and which do you think is likely to be more stable?
- Fill in the blanks: cis-1,3-Dimethylcyclohexane has two different chair conformations: one withboth methyl groups in __________ positions and one with both methyl groups in ____________ positions.Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of methylcyclohexane in the chair conformation: a. When the methyl group is in an axial or equatorial (circle one) position, the molecule is inits lowest potential energy conformation. b. Label one Newman projection above anti and the other gauche to describe the relationshipbetween the methyl group and C3 of the ring. c. In general, which is a lower PE conformation, anti or gauche? d. Explain how your answer to b and c provide an explanation for why it is more favorable fora large group to be in an equatorial than an axial position.Is the 1st compound ( more, less or equally) stable than B? Pls show through illustrations of chair conformations
- What is the least and stable conformer among the choices below?Starting from the Newman projection below, rotate the back carbon to provide the structure in a gauge staggered conformation.True or False: 1. The eclipsed conformation of a linear alkane IS called cis, while the anti conformation of an alkane IS called trans. 2. In the conversion of open-chain D-glucose to the ring form, the aldehyde carbon (carbon #1) bonds to the oxygen on carbon number 5 to form a ring that is both PYRANOSE and UNLOCKED.
- Following is a staggered conformation for one of the enantiomers of 2-butanol Q. viewed along the bond between carbons 2 and 3, draw a Newman projection for this staggered conformation.Which conformation shown below is the most stable conformation of 2-methylbutane?The potential energy of a CH3 group in ethane as it is rotated around the C-C bond can be written V= 1/2V0(1 +cos φ), where φ is the azimuthal angle as shown and V0 = 11.6 kJ mol-1. (a) What is the change in potential energy between the trans and fully eclipsed conformations? (b) Show that for smal lvariations in angle, the torsional (twisting) motion around the C-C bond can be expected to be that of a harmonic oscillator. (d) Estimate the vibrational frequency of this torsional oscil lation.



