Give the expression for Keg for the forward reaction. All are gases in a closed container. C2H6 + 2 Cl2 <-> C2H4CI2 + 2 HCI The concentrations for both Cl2 and HCl are squared due to their coefficients. [ 12 Keg [ a. H30*1 b. Он1 С. С2Н6 d. C2HĄC12 e. Cl2 f. HCI g. H2SO3 h. HC2H3O2 i. C2H3O21 j. H2S k. HS-1 I. s2 m. HX n. X-1 О. 1 р. 2 q. 3 r. 0.5000 s. 5.5 X 10-5 t. 3.5 X 10-4 u. 8.00 x 105 v. 1.75 x 10-6 w. 1.11 x 10-8 х. 5.5 х 102 y. 0.7000 z. 1.60 x 10-5 аа. 1.143 х 10-5 bb. 0.4900 СС. 1.104 х 10-5 dd. 0.7100 ее. 1.26 х 105 ff. 4.901 gg. 7.93 hh. 4.942 ii. 4.957
Give the expression for Keg for the forward reaction. All are gases in a closed container. C2H6 + 2 Cl2 <-> C2H4CI2 + 2 HCI The concentrations for both Cl2 and HCl are squared due to their coefficients. [ 12 Keg [ a. H30*1 b. Он1 С. С2Н6 d. C2HĄC12 e. Cl2 f. HCI g. H2SO3 h. HC2H3O2 i. C2H3O21 j. H2S k. HS-1 I. s2 m. HX n. X-1 О. 1 р. 2 q. 3 r. 0.5000 s. 5.5 X 10-5 t. 3.5 X 10-4 u. 8.00 x 105 v. 1.75 x 10-6 w. 1.11 x 10-8 х. 5.5 х 102 y. 0.7000 z. 1.60 x 10-5 аа. 1.143 х 10-5 bb. 0.4900 СС. 1.104 х 10-5 dd. 0.7100 ее. 1.26 х 105 ff. 4.901 gg. 7.93 hh. 4.942 ii. 4.957
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter12: Gaseous Chemical Equilibrium
Section: Chapter Questions
Problem 61QAP: Hemoglobin (Hb) binds to both oxygen and carbon monoxide. When the carbon monoxide replaces the...
Related questions
Question
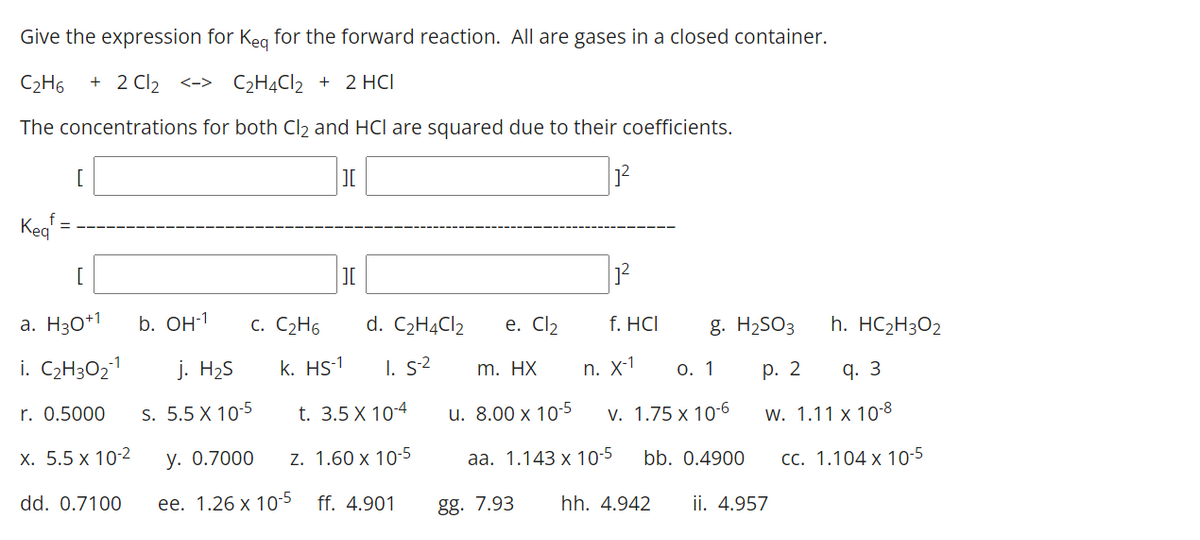
Transcribed Image Text:Give the expression for Keg for the forward reaction. All are gases in a closed container.
C2H6
+ 2 Cl2 <-> C2H4CI2 + 2 HCI
The concentrations for both Cl2 and HCl are squared due to their coefficients.
[
Kea
[
a. H30*1
b. OH-1
c. C2H6
d. C2H4CI2
е. Clz
f. HCI
g. H2SO3
h. HC2H3O2
i. C2H3O21
j. H2S
k. HS-1
I. s2
n. X-1
О. 1
р. 2
q. 3
m. HX
r. 0.5000
s. 5.5 X 10-5
t. 3.5 X 10-4
u. 8.00 x 10-5
v. 1.75 x 10-6
w. 1.11 x 10-8
х. 5.5 х 10-2
у. О.7000
z. 1.60 x 10-5
aа. 1.143 х 10-5
bb. 0.4900
CC. 1.104 x 10-5
dd. 0.7100
ее. 1.26 х 10-5
ff. 4.901
gg. 7.93
hh. 4.942
ii. 4.957

Transcribed Image Text:Give the expression for Keg for the reverse reaction. All are gases in a closed container.
C2H6
+ 2 Cl2
<-> C2H4CI2 + 2 HCI
The concentrations for both Cl2 and HCl are squared due to their coefficients.
[
I
Keg
=
[
JI
a. H30+1
b. Он1
c. C2H6
d. C2H4C12
e. Cl2
f. HCI
g. H2SO3
h. HC2H3O2
i. C2H3O21
j. H2S
k. HS-1
I. s2
m. HX
n. X-1
0. 1
р. 2
q. 3
r. 0.5000
S. 5.5 X 10-5
t. 3.5 X 10-4
u. 8.00 x 10-5
v. 1.75 x 10-6
w. 1.11 x 10-8
х. 5.5 х 10:2
у. О.7000
z. 1.60 x 10-5
aа. 1.143 х 10-5
bb. 0.4900
CC. 1.104 x 10-5
dd. 0.7100
ee. 1.26 x 105 ff. 4.901
gg. 7.93
hh. 4.942
ii. 4.957
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning