Give the structure for the compound with the empirical formula C3H3O2 and the following spectral data: 'H NMR Singlet at 3.7 ppm (3H); multiplet at 7.2 ppm (5H) IR 2850 cm (sharp), 1720 cm-1, 1600 cm1, 1503 cm-! 100- `105 80 77 60- M: 136 40- * 20 40 80 120 160 miz % Relative abundance
Give the structure for the compound with the empirical formula C3H3O2 and the following spectral data: 'H NMR Singlet at 3.7 ppm (3H); multiplet at 7.2 ppm (5H) IR 2850 cm (sharp), 1720 cm-1, 1600 cm1, 1503 cm-! 100- `105 80 77 60- M: 136 40- * 20 40 80 120 160 miz % Relative abundance
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter16: An Introduction To Infrared Spectrometry
Section: Chapter Questions
Problem 16.16QAP
Related questions
Question
Transcribed Image Text:C P
opy aste
C
ChemDoodle
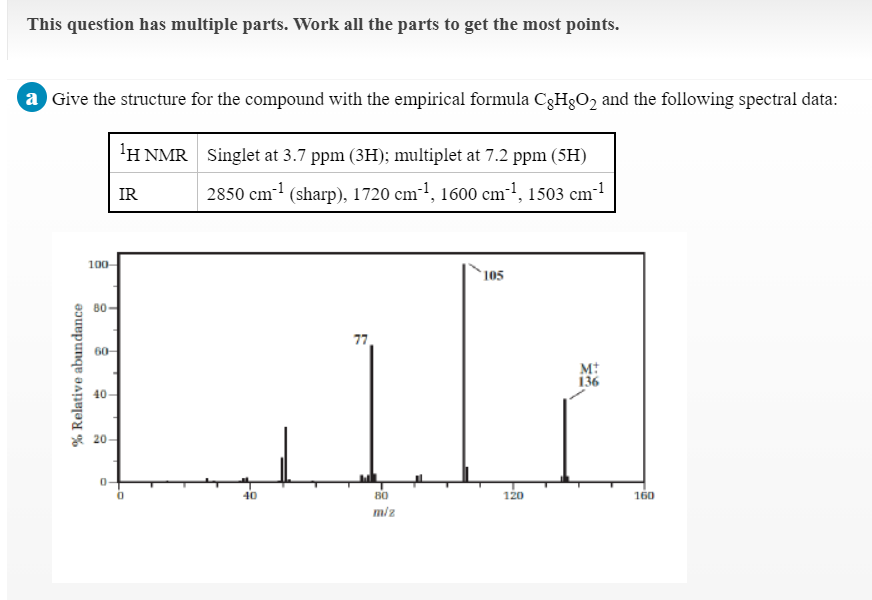
Transcribed Image Text:This question has multiple parts. Work all the parts to get the most points.
a Give the structure for the compound with the empirical formula C3H3O2 and the following spectral data:
1Η ΝMR
Singlet at 3.7 ppm (3H); multiplet at 7.2 ppm (5H)
IR
2850 cm1 (sharp), 1720 cm-1, 1600 cm-1, 1503 cm!
100-
105
80
77
60-
M:
136
40-
ae 20-
40
80
120
160
miz
% Relative abundance
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning