Q: Explain the salt-like behavior of compound A in under thirty words.
A: About tropylium bromide :
Q: Compound A exhibits the following H1 NMR, 13C NMR, and partial mass spectra respectively.…
A:
Q: I I Least reactive Most reactive
A: SN1 reaction is substitution nucleophilic unimolecular reaction. Stability of reaction depend upon…
Q: In section (ii) above an acetal group is present in the starting material. Give a general reaction…
A: An acetyl is an organic compound formed by the addition of two hydroxyl group on an aldehyde or a…
Q: The order of decreasing rectivity towards electrophilic reagent for the following: (a) Benzene (b)…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: Give the structure of the following compound(a) carbon tetrabromide
A: Organic compounds are named using specific set of rules given by IUPAC. IUPAC name contains prefix,…
Q: When (S)-1-bromo-1-phenylethane undergoes an SN2 reaction with methanethiol (CH3SH), the product of…
A: Answer : option (b) R only Explanation: Since in SN2 mechanism leaving Nucleophile and attacking…
Q: HC ECCH3 / NaNH2 H2 А 1) O3 C 2) CH3SCH3 В Lindlar's H. 1) OsO4 2) NaHSO3 (aq) D
A: The organic compounds in which two carbon atoms are bonded to each other by a double bond are called…
Q: Give the reaction conditions which could be used to bring about the transformation shown in two…
A: Oxidation and reduction are the key steps here. These steps will result in our desired product.
Q: 1. HNO3, H,SO, 2. Fe, HCI 3. NANO,, HCI 4. HBF, 5. KMN04, A CH(CHS2
A:
Q: 1. NaNH2/sivi NH3/-78°C 1. NaNH2/sivi NH3/-78°C A( C9H16) B(C19H36) H-C=C-H 2. 1-Bromo-5-metil…
A:
Q: Benzene, CeHe undergoes substitution reaction with concentrated nitric acid, HNO3 to produce…
A:
Q: Compound A (C9H12) absorbed 3 equivalents of H2 on catalytic reduction over a palladium catalyst to…
A:
Q: Give the structure of the following compound(a) iodoform
A: (a) The molecular formula of iodoform is CHI3 and it is also called as the triiodomethane, i.e. an…
Q: Give the products (if any) expected from the treatment of each of the following compounds with ozone…
A: Ozonolysis:Alkene reacts with ozone which cleaves the double bond followed by work up with dimethyl…
Q: Suggest an explanation for the observed order of SN1 reactivity of the following compounds.
A: SN1 reactions are obtained by forming the stable carbocation. These reactions proceeds through the 2…
Q: Provide Examples of dehydrohalogenation of dihalides to afford alkynes ?
A: Alkynes- Unsaturated hydrocarbon containing carbon carbon triple bond is known as alkyne. Alkynes…
Q: 2. Propose a structure of an isomer of compound ELBI which contains ester and alkene functional…
A: The compound given is,
Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. For each pair of compounds,…
A: The compounds given to be distinguished are: to be chosen from the options given: I. Halogenation:…
Q: Provide the reagents necessary to carry out the following conversion. Explain briefly what each step…
A: In given reaction reduction of epoxide to alcohol is done.
Q: Provide the appropriate reagents or product in the following examples.
A: Given ; reactant
Q: Depending on the reaction conditions, two different imines of formula C8H9N mightbe formed by the…
A:
Q: 1. Compound A, C9H12, absorbs 3 equivalents of H2 on catalytic hydrogenation over palladium catalyst…
A: The given reaction scheme is,
Q: b) Write in the products, including stereochemistry where relevant, of your compound and the…
A:
Q: Provide your answers in the Text Submission box below. Suggest reagents and reaction conditions for…
A:
Q: A certain unknown compound has the molecular formula C10H16. On catalytic hydrogenation with Pt,…
A: The degree of unsaturation in a compound can be calculated by : Degree of…
Q: Consider the following reaction schemas: (a) (b) MgBr - A+ E C8H140 H30* (c) C8H140+ 2 Na + 2NH₂ →…
A: The Grignard reagent, which acts as a source of alkyl group in organic reactions, has general…
Q: Below is a schematic representation of possible reactions that Compound X can undergo. Use the…
A: Hi, since there are multiple subparts posted. we will provide you with answers for the first three…
Q: Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is…
A: In alkylation for step 1, First the proton from the substituent is abstracted by a base. This is…
Q: Define Hemiacetal, Acetal and Ketal. ii. Give the structure of the product from the reaction of…
A: Hemiacetal is a compound which is formed when alcohol reacts with aldehyde to form a product with OR…
Q: 1. what are the differences between acyclic and cyclic forms in terms of their physical properties?
A: Note - Since you have asked multiple questions, we are bound to solve only the first question for…
Q: Please be clear in your writing, solve step by step Compound A (C9H12), when hydrogenated by…
A: We have to determine the structures of compounds A, B, and C in the following organic reaction.…
Q: Give the structure of the major organic reactant(s) that combined to give the best yvic for the…
A: From given it is Stork enamine reaction First structure is reaction of ketone with primary amine The…
Q: Esters can be prepared by the reaction of carboxylic acids with alcohols. Bearing this in mind,…
A:
Q: Below is a schematic representation of possible reactions that Compound X can undergo. Use the…
A:
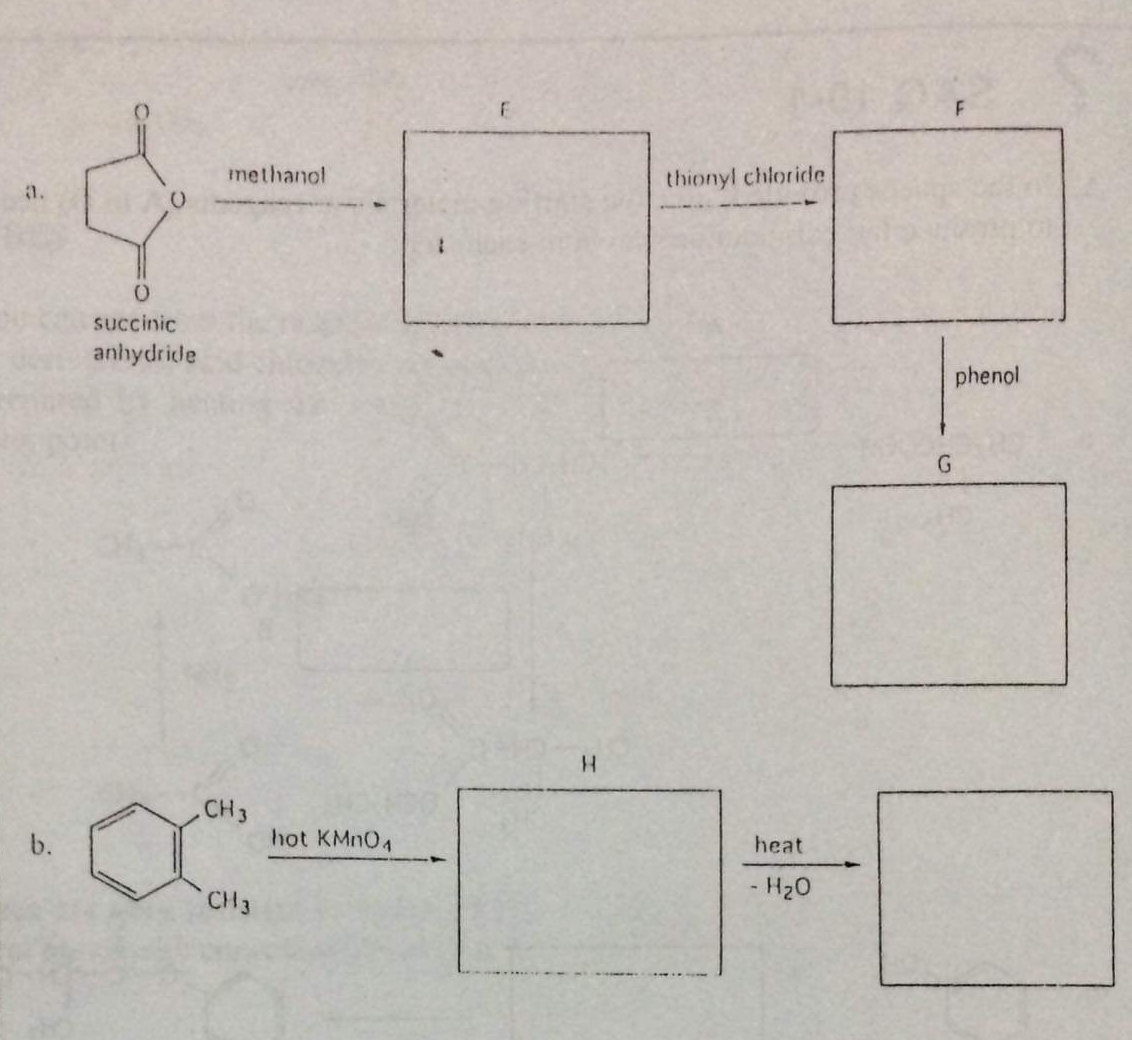
Q: What are the names of the compound A, B, C, D, E, F, G & H. Hence suggest structures for compound C…
A:
Q: between the compounds or not. (i) Compounds A and B, using UV-Vis spectroscopy with a detection…
A:
Q: rovide one alkyl halide of your choice and discuss its structure, nomenclature (systematic and…
A: Given : We have to tell one alkyl halide and its preparation as well as its application.
Q: 6. Outline the synthesis route for the product. Any appropriate reagents such as carboxylic acid or…
A: Note : The groups on benzene ring direct the electrophile to ortho, para or meta position. Step 1:…
Q: A chiral amine A having the R conguration undergoes Hofmann elimination to form an alkene B as the…
A: The ozonolysis of an alkene using reductive work up cleaves double bond to form two aldehyde or two…
Q: Define Hemiacetal, Acetal and Ketal.
A: As you have posted multiple questions, and have not mentioned the parts which you want to us to…
Q: H2Cro, A 2CH2CI2 B (i) 2Benzene AICI3
A: The first step of the reaction involves the friedal craft's alkylation reaction. In this reaction…
Step by step
Solved in 2 steps with 1 images

- The separation and purification processes given below and the method used against them are given. Which or which of these pairings are correct? I. Brewing of tea - ExtractionII. Purification of water impurities - ChromatographyIII. Separating olive pomace from olive oil while producing olive oil-DecantationIV.Petroleum fractions obtaining - Ordinary distillationV. Obtaining essential oils and using them in perfume making - Water-steam distillationA. I, II, III, IVB. I, II, III, IV, VC. I, IV, VD. I, II, III, VE. I, II, Voutline a separation scheme for isolating pure aspirin. lab video: https://www.youtube.com/watch?v=Y4NMpO1xI8U6 I- + BrO3- + 6 H+ ----- 3 I2 + Br- + 3 H2O Rate= k[I-]m[BrO3-]n[H+]p rate= (∆[I2])/(3*∆time) [I-] 0.010M [S2O3-2] 0.0010M H2O [BrO3-] 0.04M HCl 0.10M Starch Indicator (sec.) Volume, ml Volume, ml Volume, ml Volume, ml Volume, ml 1 10 10 20 10 10 1-3 drops 301.90 2 20 10 10 10 10 1-3 drops 213.28 3 10 10 10 20 10 1-3 drops 214.92 4 10 10 10 10 20 1-3 drops 86.45 Temperature of Reaction: _______20 C________ 1. Using the dilution equation (M1V1 = M2V2) calculate the concentration of the I-, BrO3- and the H+ (HCl) in the final mixture just prior to any reaction. 2. Calculate the rate for each experiment as explained in the background information. 3. Using the information above determine the rate law (m, n and p) for the reaction. Show all calculations/reasoning in your notebook. m_____________ n_________________ p…
- If I am doing an experiment on human H295R cells where I would treat the with very low concentrations of both Atrazine and DMSO. I have the cell culture media and the DMSO but I need to make a (9ppm Atrazine, DMSO) by dissolving powdered atrazine in DMSO. 1. If I have 0.04 g of Atrazine left in the bottle, how much (9ppm Atrazine, DMSO) solution can Imake? 2. How much atrazine stock willl I need to make 15.5ml of treatment solution (3ppb ATZ, cell culture media)? 3. What would be best for me to meausre the stock ATZ and what would be best for me to measure the cell culture media? 4. And then how much DMSO would I need to add to my 15.5 ml control solution?Define selective functionalisationYou finish doing an experiemnt with Benzoin. These are the results you get:Boiling start point: 137 ceciusBoiling end point: 140 ceciusVile while empty (with cap on): 15.348Vile with crude prod (with cap on: 15.748 Your starting weight: 1.00115.748 - 15.348 = 0.400 / 1.001 = 0.3996 or 39.96% YieldAnswer the following questions with this information:Initial Mass of impure sample:Mass of recrystallized benzoin:Percent reccovery of Benzoin:Melting pot range of purified benzoin:Literature melting point for benzoin:
- Ksp for BaCrO4 is 1.17x10^-10Indicate the appropriate protocol below to prepare the required solution. a) Solution 5 Preparation: Prepare: 10 mL of 23% mannitol From: solid mannitol Procedure: b) Solution 2 Preparation: Prepare: 0.5 liters of 0.01 mg/mL ascorbic acid From: solid ascorbic acid , MW = 176.1 g/mol Procedure: C) Solution 3 Preparation: Prepare: 1 liter of 150 mM Tris From: 1 Molar Tris stock solution Procedure:Please don't provide handwrittin sollution...
- Suppose you have an organic sample X that is somewhat soluble in water, even though it issomewhat more soluble in dichloromethane or ether solvents. But if you do a singleextraction, you get only 60% of your material to transfer from the water to the organic layer.How many “washes” would it take to extract over 90% of your organic material extracted fromthe water layer?A solution containing orange rind extract was analyzed using gas chromatography to measure its limonene content. The parameters used to analyze the standards and samples are listed below. Calculate the concentration of limonene in the solution in ug/mLWhich of the ff is not true about recrystallization? a. acetanalide recrystallized only at cold temp b. congo red stain is removed by using animal charcoal c. acetanalide can be dissolved in hot h20

