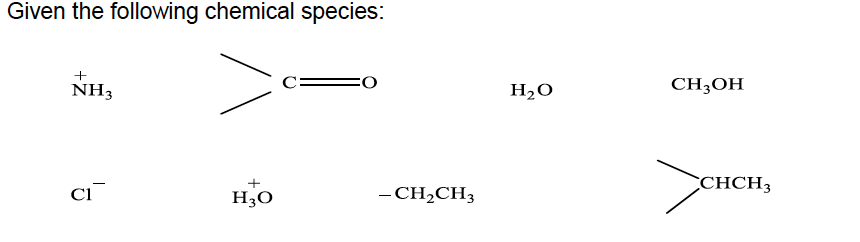
Given the following chemical species: NH3 H2O CH;OH CHCH3 H30 - CH2CH3
Q: нно HHO Draw a diagram to show how the monomers to the right could be joined to form a polymer and…
A:
Q: Pyridine (right) is an essential portion of many biologically active compounds, such as nicotine and…
A: Pyridine is an organic heterocyclic, chemical C5H5N compound. It is similar to that of benzene…
Q: functional groups in palmitic acid, a saturated fatty acid and oleic acid, an unsaturated fatty acid
A: Biological lipids are a chemically diverse group of organic compounds which are insoluble or…
Q: Part A What functional group pairing allows one amino acid to bond (a) H R ANSWER: N-CIC OH O H (b)…
A: An amine is often a group with a lone pair on the nitrogen atom. Amines structurally mimic ammonia,…
Q: Functional Group Name Structural Diagram (draw all bonds) Found where in the body??? Hydroxyl H -N H…
A: Functional groups can be described as the groups of molecules that get linked to organic molecules…
Q: ponds. Given a molecular formula (such as CH4) identify the molecule Given an organic molecule…
A: Hi! Since you posted many questions, we will be answering the first for you. Please post the other…
Q: 4. Draw the bond-line dash-wedge structure and the Fischer projection of the following compounds.…
A: The wedge and dash projection is a means of representing a molecule with three types of lines. The…
Q: Amino acids can be prepared from a-halo carboxylic acids [RCH(X)COOH] by reaction with excess NH3.…
A: Amino acids are the organic acids that contains an amino group, carboxyl group, hydrogen atom, and…
Q: Calculate the charge and draw the structure of the predominant ion for: (i) Lysine at pH 7.0…
A: The amino acids are the monomer unit of the peptide chain, which exhibits different charges at…
Q: The C-C single bond in C3H6 (cyclopropane) (use C-H bond enthalpy and bond energy calculated for…
A: Bond enthalpy for c-c in cyclopropane is 277KJ/mole.
Q: Draw the structure of the parent lipid.
A: Lipids can be defined as organic substance that are moderately insoluble in water but soluble in…
Q: "CH,OH OH HO A. Draw the 2 resulting structures that would occur upon initial hydrolysis of the…
A: Glycosidic bond: The bond that joins carbohydrate residue with other group is called Glycosidic…
Q: 25- General formula of alkyne compounds are. O A A - CnH2n+2 O B - CnH2n-2 O C - CnH2n
A: Alkynes are hydrocarbons that have triple bonds between carbon atoms.
Q: a) Sort compounds 1a to 1c in order of increasing melting point and briefly justify your ordering.…
A: Phospholipids are a group of lipids that constitutes of two fatty acids, a glycerol unit and a…
Q: Recalling that a beaker of water is three dimensional what is the three dimensional shape of the…
A: They are hydrophobic, and are located in the lipid bilayer of the hydrophobic tails. Cholesterol…
Q: Functional Groups: MATCHING 1. sulfhydryl 2. carboxyl 3. carbonyl 4. phosphate 5. amino а.- ОН b. -…
A: 1. Sulfhydryl (-SH): It is present in amino acids methionine and cysteine. So, it is present in…
Q: Accepting. Which of the following compounds readily accepts amino groups from amino acids? a.…
A: The amino group of one amino acid is transferred to keto acid to form amino acid and keto acid in…
Q: using the expanded structure of Levothyroxine. Determine the molecular geometry of each central…
A: To determine the geometry of the carbon atoms in Levothyroxine , we would draw the structure and see…
Q: HQ CH HC HQ NH OH Но Consider the following 2 molecules. Which one of these are polar and which one…
A: The molecular structure determine the polarity of the molecule and the presence of polar groups are…
Q: Instructions: Differences between Cis-fatty acid VS. Trans fatty acids Use white bond…
A: Unsaturated fatty acids are in configuration that is called cis, but partially hydrogenation can…
Q: Define the alternative chemical structure of deoxyribonucleic acid which could be copienfaithfully.
A: The Sanger chain termination sequencing method was invented by Sanger to identify the sequence of…
Q: the major components of complex biomolecules. For each of the two important biomolecules below…
A: GTP is an energy-rich molecule, just like ATP. Usually when it is hydrolyzed, the free energy of…
Q: aromatic ring он ÇH, CH, an acidic amino acid NH contains sulfhydryl group H,N NH reactions with…
A: Amino acids are categorized into different groups based on their side chain such as polar,…
Q: Proteins are quite stable. The lifetime of a peptide bond in aqueous solution is nearly 1000 years.…
A: Proteins are major biomolecules found in the body. They consist of peptide bonds. These peptide…
Q: The chemical structures of the two opposing ends in any polypeptic because: O a. the structures of…
A: A polypeptide is a string of covalently bonded amino acids that has not been folded into a…
Q: Exercise A: Amino Acid Functional Groups Figure 1 below shows one of the 20 amino acids that make up…
A: Amino acids are organic molecules that are made up of a side chain (R-group), an amino group and an…
Q: Functional Group Name Structural Diagram (draw all bonds) Found where in the body??? 7. 8.…
A: Introduction:- Alcohol is dispersed throughout the body's fluids, exposing most tissues, including…
Q: Select ALL the statements that accurately describe the differences between a hemiacetal and a…
A: Acetal and hemiacetal are both functional groups but vary slightly in thier chemical structures. The…
Q: Draw two different possible hydrogen-bonding interactions between two molecules of formamide…
A: Hydrogen bonding interactions are the weaker bonds, which are present between two electronegative…
Q: Φ and ψ in the Ramachandran plot (below) are: a) Rotational angles around the bond between the…
A:
Q: Disulfide bonds help to stabilize the three-dimensional structure of proteins. What amino acids are…
A: Introduction Proteins are formed by 20 standard amino acids with limited amino acids availability…
Q: te the structure for a generic triacylglyceride at pH 7.4.
A: The simplest lipids which are derived from fatty acids are the triacylglycerols which are also known…
Q: Check all that applies to the structure of fatty acid. H2C-O C1H35 CH2OH HỌ -C,7H35 + 3NAOH →…
A: Triacylglycerols are the esters of glycerol with the fatty acids. The hydrolysis of triacylglycerols…
Q: More ratios. Through the use of nuclear magnetic resonance spectroscopy, it is possible to determine…
A: From Henderson Hassel batch Equation pH = pKa +log [A*]/ [HA]
Q: Can i get help with this problem step by step?
A: A functional group is a group of atoms that determine the chemical properties of a compound.…
Q: 1.Histidine has three ionizable functional groups. Write the equilibrium equations for its three…
A: The equilibrium between the weak acid and its conjugate base allows the solution to resist changes…
Q: Complete Structural Formula Molecular formula (a) C-C=c (b) C-C-C-č-OH c-c- -c-c-NH,
A: Organic compounds are those that consists mostly of carbon, hydrogen and oxygen. Other atoms…
Q: Differences between Cis-fatty acid VS. Trans fatty acids
A: ANS: Unsaturated fatty acids have one or more double bonds. Each double bond may be in a cis or…
Q: The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by…
A: At pH=1 (pH<7), there is an excess of H+ ions in the aqueous solutions. So, the COO-n group…
Q: Functional Groups: MATCHING 1. sulfhydryl 2. carboxyl 3. carbonyl 4. phosphate 5. amino а.- ОН b. -…
A: Macromolecular:- A molecule containing a very large number of atoms, such as a protein, nucleic acid…
Q: Polymers of macromolecules are disassembled to monomers by: O a. Van der Waals interaction O b.…
A: A polymer is a large molecule that is made from many small repeating chemicals called monomers.
Q: || Biological Molecules-Modelling Activity AC1.1-ustrate or construct molecular models of simple…
A: Glucose is the monomer of carbohydrates. Glycine is the simplest amino acid. Glycerol is the part of…
Q: It's about chemical bonding I am a bit confuse of how to figure out what is a anion or a cation
A: A chemical bond denotes the attraction between atoms, molecules, or ions, which leads to the…
Q: biologically derived hydroxyapatite? b) How do you define porous and dense hydroxyapatite? Ple…
A: Hydroxyapatite is a hexagonal crystal type of the mineral calcium apatite, which contains calcium,…
Q: cis, cis-9,12-OCTAdecadiENoic acid Enumerate the number of carbons and unsaturations (double bonds)…
A: Linoleic acid is also known as Ocatadecadienoic acid is a polyunsaturated essential fatty acid that…
Q: Catalytic hydrogenation, used in the food industry, converts double bonds in the fatty acids of the…
A: Carboxylic acid with hydrocarbon side chain are fatty acids. Fatty acids are mostly esterified but…
Q: I. Write a balanced chemical reaction, complete with chemical structures, to show the hydrolysis of…
A: Introduction Iso-C or 2-aminouracil is an isomer of Cytosine which upon hydrolysis yields Uracil…
Q: Part G - Disulfide bonds Based on the sequence alignment, is it possible that one or more disulfide…
A: The right answer is.. Yes, the conserved cysteine and methionine residues will form two disulfide…
Q: Hair is made of protein. The protein is a structural protein called α-keratin, which contains many…
A: The nucleotide-based genetic information is converted to the amino acids that will create the…
dentify (showing clearly the basis for your answer in each case) which one(s) can serve as, (a) H-bond donor b) H-bond acceptor
Step by step
Solved in 2 steps

- What coefficients must be placed in the following blanks sothat all atoms are accounted for in the products?C6H12O6 S _________ C2H6O + _________ CO2(A) 2; 1(B) 3; 1(C) 1; 3(D) 2; 2What is the empirical formula for C3H6O3? C3H6O3 C6H12O6 CH2O None of theseWhich of the following compounds would be most soluble in carbon tetrachloride, CCl4? a. NH3 d. CH3OHb. H2O e. C6H6c. HCl
- What coefficients must be placed in the following blanks so that all atoms are accounted for in the products?C6H12O6 → ________ C2H6O + ________ CO2 Group of answer choices A. 2; 1 B. 3; 1 C. 1; 3 D. 2; 2Write a sample chemical reaction of chymotrypsin in a complete balanced equation label the following properly: substrate/s, cosubstrate/s, and cofactor/s Show all the changes in the reacting components small molecules should be in their skeletal form large molecules could be shown as hybrid structures - reacting/interacting groups in skeletal form, the rest of the molecule as abbreviations/blocks/shapeswhat is the conjugate acid for the following reaction? HC2HO4 + H2O <--> H3O+ + C2HO4- A)C2HO4- B) HC2HO4 C)H2O D) H3O+
- Consider a transition of X → y. assume that the only difference between X and y is the presence of three hydrogen bonds in y that are absent in X. What is the ratio of X to y when the reaction is in equilibrium?Consider the intermolecular forces present in a pure sample of each of the following compounds: CH₃CH₂OH and CH₃COCH₃. Identify the intermolecular forces that these compounds have in common.In the Equation: HCO3- + HCl --------> Conjugate Acid + Conjugate Base a ) Which reactant is the Acid? b) Which reactant is the Base? c) What Conjugate Acid will form in this reaction? d) What Conjugate Base will form in this reaction?
- Examine the chemical equation below, and label each reactant and product. (which is a reactant and product) H2O + CO2 ⇋ H2CO3 ⇋ HCO3- + H+ If you are blowing CO2 from your lung into a water solution, will it make the solution more acidic or basic?Arrange the following compounds in the increasing order of their boiling points :CH3CHO, CH3CH2OH, CH3OCH3, CH3COOH.What is the pH of a glycine solution in which the alpha-carboxyl group is one third dissociated? Given pka1=2.3, pka2=9.6

