Given the following information on reduction potentials, calculate the standard free energy in kJ/mol based on your understanding of electron transfer through the pyruvate dehydrogenase complex. Round to nearest whole number. Lipoamide + 2H+ + 2e- dihydrolipoamide Ae° = -0.29 FAD + 2H+ + 2e- FADH, Aɛ° = -0.01
Q: 2. Which of the following statements about bond breaking is not true? A) Homolysis and heterolysis r...
A: The statement about bond breaking is not true = ? A) homolysis and heterolysis require energy. B) I...
Q: The gas constant R has the value 287 J/Kg * K for dry air. Using the ideal gas law (P = PdryRT): a.)...
A:
Q: Compute the empherical formula of the following: a. 19.1% Sn and 80.9% I b. 25.9% Fe and 74.1% Br
A: Given, a). 19.1% Sn and 80.9% I b). 25.9% Fe and 74.1% Br Determine the empirical formula of the co...
Q: 1. a) Determine the bond order of emphasized atoms in the following molecules and then ran! them in ...
A: Since part (b) of the question is incomplete, we will solve part (a) for you. To get the remaining s...
Q: equatorial bonds axial bonds Practice problems: Br
A: Both structures of Cyclohexane are given in equitorial and axial positions.
Q: 8. Propose a structure for the compound having the mass spectrum shown below. Identify the major fra...
A:
Q: I’m the hypochlorite oxidation of cyclohexanol to cyclohexanone ,what purpose does the acetic acid s...
A:
Q: Compute the molecular formula of the following a. 19.1% Sn and 80.9% I b. 25.9% Fe 74.1 Br
A: Given, a. 19.1% Sn and 80.9% I b. 25.9% Fe 74.1 Br Compute the molecular formula of the following =...
Q: Determine the final concentration in mol dm–3 of K+ and SO42– when the following three solutions in ...
A: Their are three volumetric flask of volume 500cm3 contain 0.20 mol/dm3 KAl(SO4)2 and second flask of...
Q: (c) Predict the chemical tests you would use to distinguish between the isomers in (a). (d). When th...
A: Isomers Exhibited by The Co(en)3 is optical Isomers and there exist two form dextro and laevo .
Q: How many grams of NaOH must be added to 1.00 L of water to make a 15.0% NaOH solution?
A: Given data: The volume of solution = 1 L %wV = 15.0 %
Q: 5. How could the following pair of isomeric compounds be differentiated by mass spectrometry? Br H3C...
A: Introduction : Mass Spectroscopy is a analytical technique which is used to determine the molecular...
Q: Which reaction is slowest? inin reaction coordinate reaction coordinate reaction coordinate A B А) A...
A:
Q: 21. What is the order of a reaction where each successive half life gets longer and is dependent upo...
A: The half-life of second-order of a reaction: t1/2 = 1K[A]∘ In the second-order reaction, the ha...
Q: Two ideal gas systems undergo reversible expansion under different conditions starting from the same...
A:
Q: relationship betwe
A: Enantiomers are mirror images that are non super imposible on each other. Diastereomers are non mirr...
Q: Calculate the % composition of CuSO4.5H20 (Atomic masses:Cu = 63.55 amu; S = 32.06 amu; O = 16.00 am...
A:
Q: The pH of a 0.350 M aqueous acid solution measured 5.79. What is the acid ionization for this acid? ...
A:
Q: At constant Tand n, the pressure of a gas can be doubled by: decreasing volume to half O doubling vo...
A: At constant T and n ,the pressure of a gas can be doubled by given answer as below.
Q: What are the charges of germanium and antimony in Gd3Sb4
A: Metals take positive change after losing the electrons whereas non-metals take negative charge after...
Q: 9) drug shown below: Select the correct number of chirality centers in heroin, the illicit A) 4 B) 5...
A:
Q: What is the pH after the addition of 1.0 mL of 0.10 M HCI to 20 mL of the solution above?
A:
Q: A beaker contains water up to the 130 mL mark. We introduce an object 38 g of dough and we observe t...
A:
Q: nite the eacion for the acidic and busic Salts mchen It is dissolived M waier. Be sure 1o write The ...
A: These reactions are the salt of Hydrolysis.
Q: What is the molarity of a solution that contains 132 mg of HCl in 450 L of water?
A: Given :- Mass of HCl = 132 mg Volume of water = 450 L To calculate :- Molarity of solution
Q: What happens to the stereochemistry during and SN2 reaction? Why? Provide a reaction to illustrate t...
A:
Q: 1. Draw all possible stereoisomers of the structures below. Indicate the isomeric relationships betw...
A: The answer is as follows:
Q: 1 Balance the following equations: a) C(s) bFe(s) + c) P(s) d) C,H,(g)+ --> CO(g) 2 FeCl,(s) P,O,(s)...
A: Given reactions are : a). C (s) + O2 (g) -------> CO (g) b). Fe (s) + Cl2 (g) ---------> FeCl...
Q: °C and The melting point of a solid is 39.0 °F. Convert it to
A:
Q: 1. The combustion of a sample of aluminium produces 0.25 mol of aluminium oxide and releases 419 kJ ...
A: The combustion reaction of aluminum is given by: Als + 34O2g → 12Al2O3s The enthalpy change in stan...
Q: Fill in the blanks: A Scotch whisky blend contains 40% alcohol by volume. How many milliliters of al...
A: Number of mole is calculated by dividing the given mass with molecular mass.
Q: Explain the order of chemical shifts of the carbon atoms in the 13C spectra of cyclohexanone (fig 22...
A:
Q: Compute the molecular formula of the following: 1. 80%C and 29% H and molecular mass of 30 amu
A: Given, Mass percentage of C = 80% Mass percentage of H = 20% Molecular mass of the compound = 30 ...
Q: WHAT IS THE RIS CONFIUURATION RELAFION SHIP? Br OH VS HO Br.
A:
Q: If the osmotic pressure of a 8.25×10-2-M aqueous solution of CaCl2 was found to be 5.41 atm at 20°C,...
A:
Q: At 298 K, the enthalpy of denaturation of hen egg white lysozyme is 217.6 kJ/mol. The change in heat...
A:
Q: Calculate the freezing point of a solution of 1.95 g of naphthalene (C10H8) in 15.50 g of benzene. T...
A: This is a depression of freezing point or lowering of freezing point problem. This lowering of the f...
Q: 1. For the reaction 2NO(g) + 02(g) 2NO,(g) rate = k[NO] [02] %3D Is the following proposed mechanism...
A: Here we have to verify whether the proposed mechanism of reaction is plausible for the given reactio...
Q: Calculate the pH for a 0.1 M solution of C6H5COOH (K, = 6.46 x 105) and the pH for a 0.2 M %3D solut...
A:
Q: * 00 0.0 When glucose (C 6H1206) reacts with molecular oxygen, Gaseous carbon dioxide and liquid wat...
A: Given, The combustion of glucose(C6H12O6) with molecular oxygen to form gaseous carbon dioxide and l...
Q: Calculate [H3O*]equilibrium in a solution that is 0.10 M in NaF and 0.20 M in HF. Ka for HF = 7.2 x ...
A: Answer: HF is a weak acid. When added in water it will dissociate partially to give hydronium ion an...
Q: Please answer questions 1 & 2 and show work. 1. Calculate the mass, in g, of chlorine in 25g of th...
A: 1) Given, Mass of chlorofluorocarbon (CFCl3) = 25 g Mass (in grams) in Chlorine in 25 gram of chl...
Q: The activation energy of a certain reaction is 45.6 kJ/mol. At 20 °C, the rate constant is 0.0130 s....
A: A) We need to find temperature T2 B) We need to find rate constant K2
Q: 0.333 g of a hydrocarbon (CxHy) was analyzed by combustion analysis and 1.009 g of CO2 (MM = 44.01 g...
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to ...
Q: a) Write down the anodic and cathodic electrochemical reactions occurring in a Zink (Zn) - Copper (C...
A:
Q: at kind of reaction doe
A: Addition reaction is defined as a reaction in which 2 reactants add together in order to form a sing...
Q: At 25°C, the degree of dissociation for the reaction: N2O4(g) 2 NO2(g) is 4.94%. If 0.0500 mol of N2...
A: Given reaction is : N2O4 (g) <---------------> 2NO2 (g) Degree of dissociation = 4.94% Moles ...
Q: An organic compound has a molar mass of 171.1 g/mol and contains 11.10 % hydrogen atoms by mass. How...
A:
Q: The ion PCI4 has a structure that would be described as? a. octahedral b. Square planar c. trigonal ...
A: Which of the following is correct
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are ...
A: We can decide on the fate of these given reactions in the question based in the solubility pattern o...

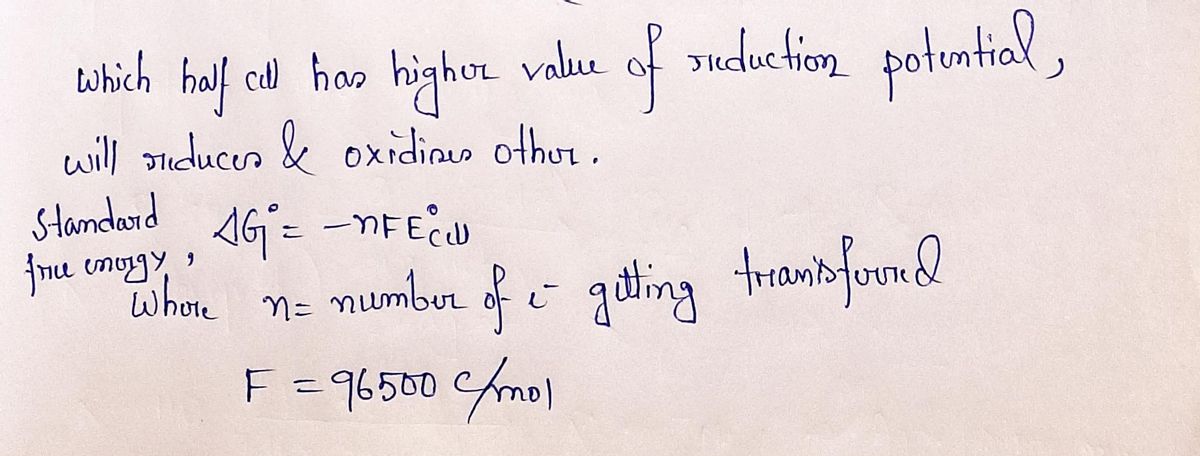
Step by step
Solved in 2 steps with 2 images

- The overall adenylate kinase reaction (2 ADP = AMP + ATP) is a combination of 3 reactions: ATP hydrolysis, pyrophosphate hydrolysis, and ATP formation. Show each step and provide the standard free energy (ΔG°′) for each and the overall reaction. If the adenylate kinase reaction is at equilibrium and intracellular [ATP] = 5 mM and [ADP] = 0.5 mM, calculate the concentration of AMP at pH 7 and 25 °C. Show all work. ATP (ADP+P) = -30.5 kj/mol; ATP (AMP+PP) = -45.6 kj/mol; PP = -19.2 kj/mol1 In relation to the redox below from electron transport chain (SuccinateDehydrogenase) Show calculations that proof how oxidation of FADH2 by ubuiqinone(Q) theroteically liberates enough enrgy to drive synthesis of ATP. △G°‘ or ATP synthesis from ADP and π is +30.5kj/mol. Explain calculation in terms of how the oxidation of FAD Hz by ubiquinone supplies sufficient energy for ATP synthesis from ADP and π.The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or Si face of the substrate? What about the addition of the H? Do the H and OH groups add from the same side of the double bond or from opposite sides?
- Assume you are studying a hydrolase that catalyzes the following reaction which has a ΔGO of + 27 kJ/mol. A-B + H2O ↔ A-H + B-O− + H+ a. What is Keq for this reaction? b. What are Keq’ and ΔGO’ for this reaction? c. Is this reaction favorable under standard biochemical conditions at pH 7? Justify your answer.You are called in as a consult for a patient who suffers from acidosis. Urine levels of both lactic acid and isocitric acid are elevated. Mitochondria isolated from the patient’s fibroblasts oxidize pyruvate very slowly whereas α-ketoglutarate and malate are oxidized normally. a. At what enzyme would you predict the patient has a defect? b. Why? What evidence do you have to choose that enzyme? c. How do you account for the increase in lactic acid?A) Assuming that ubiquinone is unavailable inside of the cell, calculate the delta G and the Keq if electrons are transferred directly from complex l to complex lll of the electron transport chain. In your answer include the net equation for this electron transfer. Constants: R = 8.31/ degree1mol, F = 96.1kJ/voltmol, T = 298K. Half reaction: E(V) Ubiquinone + 2e- + 2H+ --> Ubiquinol + H2 0.045 NAD+ + 2e- +2H+ --> NADH + H+ -0.320 1/2 O2 + 2e- + 2H+ --> H2O 0.816 Cytc (Fe3+) + + 1e- + 1H+ --> Cytc (Fe3+) 0.254 B) In a single sentence explain whether or not this electron transfer is possible
- please please help me to answer it >>> i have an exam Human xanthine oxidase catalyzes the oxidation of hypoxanthine to xanthine and can furthercatalyze the oxidation of xanthine to uric acid. For the treatment of hyperuricemia and gout,several medications are used to inhibit the activity of xanthine oxidase and reduce theproduction of uric acid. You are a biochemist and just discovered a chemical that can inhibitthe activity of the human xanthine oxidase. When analyzing its mode of inhibition, you foundthat the enzyme inhibitor complex requires 450 J.mol-1 to dissociate and that it displayskinetics somehow similar to noncompetitive inhibition. You sent your inhibitor to the ministryof health for approval as a medication for gout. Based on the data provided, are they going toauthorize it as a medication or not? Explain?Explain, using Le Châtelier’s principle, why the equilibriumconstant for the formation of NO from N2 and O2increases with increasing temperature, whereas the equilibriumconstant for the formation of NO2 from NO and O2decreases with increasing temperature.The thiamine pyrophosphate (TPP)-dependent enzyme benzoylformate decarboxylase catalyzes the decarboxylation of benzoylformate to yield benzaldehyde. Propose a mechanism based on your knowledge of TPP-dependent mechanisms. You may begin the reaction with the ylid form of TPP and you only need to show the structure of the “business end” of TPP. Show all electron flow using the arrow convention.
- The vitamin Niacin is used to form nicotinamide adenosine dinucleotide, which readily shuttles between its oxidized (NAD+) and reduced (NADH) forms. The latter serves as a cellular equivalent to NaBH4. The essential portions of the structures are shown below. Outline a mechanism for the cellular conversion of pyruvate to lactate. (Note: like NaBH4, NADH cannot reduce carboxylic acid carbonyls).Which of the reactions are spontaneous (favorable)? 1)DHAP↽−−⇀glyceraldehyde-3-phosphateΔ?=3.8 kJ/mol 2)L-malate+NAD+⟶oxaloacetate+NADH+H+Δ?=29.7 kJ/mol- 3) glutamate+NAD++H2O⟶NH+4+α-ketoglutarate+NADH+H+Δ?=3.7 kcal/ 4) C6H13O9P+ATP⟶C6H14O12P2+ADPΔ?=−14.2KJ/mol 5) −Rh(I)C2H6Δ?=−150.97 kJ/molC2H4+H2→Rh(I)C2H6ΔG=−150.97 kJ/mol 6) C4H4O5⟶C4H2O4+H2OΔ?=3.1 kJ/molwhat is the sterochemical outcome and draw the transition state responsible for steroinduction. -draw clearly

