Q: The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mec...
A: The given reaction is the elimination of HBr from 2-bromobutane to give alkene as the product. The e...
Q: Bond Energies (kJ/mol) Bond Bond Energy Bond Bond Energy Bond Bond Energy H–H 436 C–S 260 F–Cl...
A: In a chemical reaction, some bonds in reactants are broken (which requires energy I.e. + sign) and s...
Q: The half-life of 26?? 59 is 44.6 days. How long will it take for 95.0% of a sample of this nuclide t...
A:
Q: Consider the following chemical reaction:C(s)+H2O(g)→CO(g)+H2(g) How many liters of hydrogen gas are...
A: Given: mol of Carbon = 1.07 mol pressure = 1 atm temperature = 317 K we have to find grams of carbon...
Q: Help me please
A: Since you have posted multiple questions in a single session, we are entitled to answer first questi...
Q: The structure of the product, C, of the following sequence of reactions would be
A: The reaction of terminal alkyne with NaNH2 in ammonia causes the abstraction of the alkyne proton to...
Q: 1.80 grams of an impure mixture containing sodium carbonate required 84.0 mL of 0.125 M H2SO4(aq) fo...
A: An impure mixture containing Na2CO3 whose percentage of mixture is to be determined.
Q: Phosphorus – 29 is a positron emitter. Write an equation for this nuclear reaction and identify the ...
A: It is given that phosphorous-29 undergoes a positron emission, positron emission is also called as β...
Q: In quantum mechanics a node (nodal surface or plane) is:
A: The objective of the question is to find the correct option among the give several options for the q...
Q: An industrial process for manufacturing sulfuric acid, H2SO4, uses hydrogen sulfide, H2S, from the p...
A: Given information: Density = 2.62 g/L Pressure = 1.00 atm Temperature = 25 °C Molar heat capacity = ...
Q: The temperature of an ideal gas with a volume of 1.0 L increases from 264 K to 330 K at constant pre...
A: Initial volume, V1 =1.0 L Initial temperature, T1=264 K Final temperature, T2= 330 K The final volu...
Q: Propose a two-step synthetic strategy for the synthesis of 2-methylhexane from 5-methyl-2- hexanol.
A: it’s shown below:
Q: Give the IUPAC name for each compound (d, e, and f)
A: d) The compound given is having alcohol i.e -OH as major functional group. The longest carbon chain ...
Q: What information is required to determine how many grams of sulfur would react with a gram of arseni...
A: To find : what information is required to determine how many grams of sulfur would react with a gram...
Q: Table III-C. Type of Radioactive Decay for Some Radioisotopes Unstable Isotope Prediction Confirmed ...
A: Nuclear chemistry. Radio active disintigration.
Q: 100.0 ml of a0.500 M solution of KBr is diluted to 500.0 ml, what is the new concentration?
A: Determining new concentration.
Q: Please I need help on both questions with a good explanation for me. Thank you
A: Given, cation K+ and Anion O2- For ions, Number of electrons is equal to atomic number minus the ch...
Q: Calculate AHxn for the following reaction, using the data provided. C2H5OH(1) + 2 C2H4(g) + 5/2 O2(g...
A: The enthalpy change of a chemical reaction can be calculated by the standard heat of formation of th...
Q: Consider the reaction CO(g) + 2H2(g) CH3OH() at 25°C Calculate value of the equilibrium constant (Kp...
A:
Q: What will be the major product from the following E2 reaction? CH3 CH2-CH-ČH-CH2CH3 + CH30 Br CH3 CH...
A:
Q: The table indicates how pH indicators change color based on the pH of the solutions that you put the...
A: Given : pH range in which colour changes for bromothymol blue is 6.0 - 7.6. The colour change of bro...
Q: What is used to measure PH of protein solution
A: Protein Fouling of pH Sensing Glass Membrane In any aqueous solution, acid is present in the form of...
Q: Can I please get some help with 1a-d
A: The Diels alder is a cycloaddition reaction of diene (involves 4 pi electrons) and a dienophile (inv...
Q: Draw the major organic product(s) of the following reaction. DMF `Br + KCN • You do not have to cons...
A:
Q: Element 106 was recently named seaborgium (Sg) in honor of Glenn T. Seaborg, a pioneer in the discov...
A:
Q: For the laboratory equipment below give me the name, what it is used for, minimum measurement in mL,...
A: The apparatus is given that is used in laboratories, laboratory equipment is defined as an apparatus...
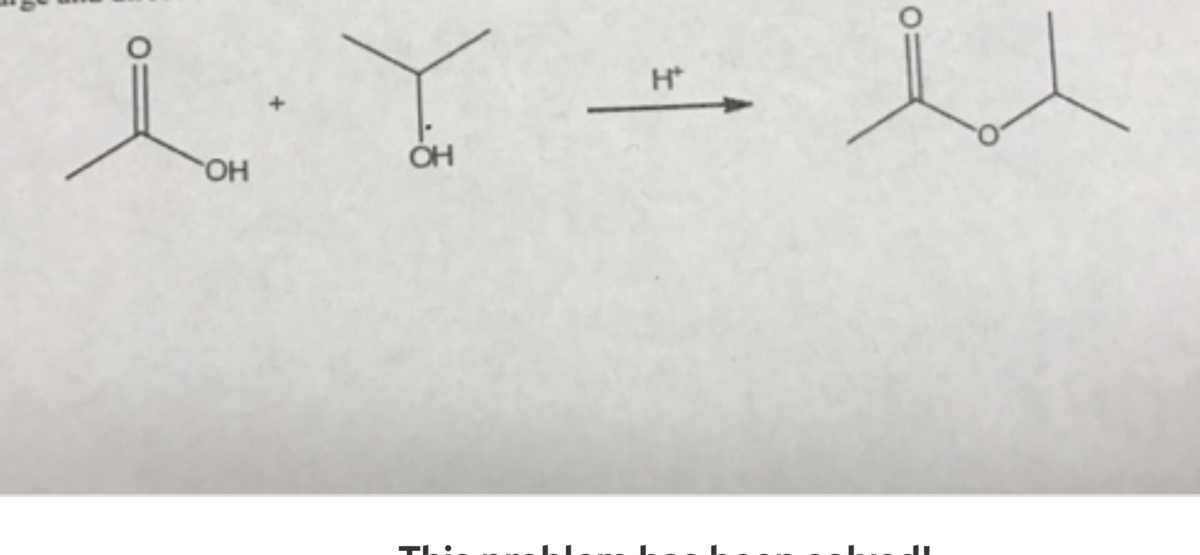
Q: Chemistry Question
A: Given substrate (reactant) is ester which undergo hydrolysis in presence of base (like NaOH) to give...
Q: The process in which complex molecules are broken into small molecules to provide energy is known as...
A: Welcome to bartleby ! Introduction : We have to tell the name of process .
Q: imes the kinetic product is the same as the thermodynamic product.) + Br2 ?
A:
Q: The relationship between the 2 molecules are: a. identical b. enantiomer c. diastereomer
A:
Q: What is the primary intermolecular force present between two SCl2 molecules? a. London or dispersion...
A: SCl2 has bent shape like water molecules. The number of valence electrons in central atom Sulphur is...
Q: You have a bottle of a colorless liquid with no label. When you vaporize a sample of liquid in a 289...
A:
Q: Homework Help
A: The reaction given is,
Q: 27. Which of the following is a biological function of proteins? a. catalysis Ob. motion Oc. transpo...
A: proteins catalyse reactions in our bodies, transport molecules like oxygen and keep us healthy as pa...
Q: How many mL of methadone is needed for a patient who was prescribed 0.015 g of methadone? The vial r...
A:
Q: There are 0.500g of He of in a closed vessel of volume 500.mL at 50.0°C, what is the pressure? O 1.0...
A:
Q: Does the lattice energy of an ionic solid compound increase or decreas as the charges of the ions in...
A: Lattice energy: Amount of energy needed to convert one mole of given ionic crystal lattice into its ...
Q: What is the relationship between the following two compounds? Constitutional isomers The same compou...
A: The molecular formula of both the compounds are, C5H12. Since it contains same molecular formula but...
Q: Are these chiral, achiral, or meso?
A: When a carbon atom is attached to four different substituents, then the carbon is called asymmetric ...
Q: Which of these compounds is expected to react the fastest in an Sn1 reaction? CH30 CH2BR O2N- CH2BR ...
A: Benzyl bromide gives SN1 reaction with the nucleophile. In the given reaction the benzyl bromide act...
Q: Barium carbonate is the source of barium compounds. It is produced in an aqueous precipitation reac...
A: The reaction between barium sulfide and sodium carbonate is considered.
Q: How many moles of CaO form when 98.60 g CaCO3 decompose?
A: The decomposition reaction of CaCO3 can be expressed as, CaCO3 ⇌ CaO + ...
Q: The unknown acid H2X can be neutralized completely by hydroxide ions according to the following equa...
A:
Q: w and name the correct orbitals involved when a Si atom and four F atoms come ether to form SİF4. Yo...
A:
Q: How much heat is required to vaporize 54.9g of acetone at its boiling point? The heat of vaporizatio...
A:
Q: Draw the following: a.)5-ethyl-3-methylnon-1-ene b.)1-butoxyhexane c.)hepta-2,4-Dione
A: Given, a.)5-ethyl-3-methylnon-1-ene b.)1-butoxyhexane c.)hepta-2,4-Dione
Q: An element X forms both a dichloride (XCl2) and a tetrachloride (XCl4). Treatment of 10.000gm of XCl...
A:
Q: structural formula for the substitution product of the reaction shown below. Br NH3 CH3OH Use the we...
A: Given reaction: Here the Br group acts as a leaving group when treated with NH3/CH3OH and NH3 acts...
Provide a complete arrow pushing mechanism for the reaction shown below
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


