Q: — ― FeCl(aq) FeCls (aq) FeCl (aq) FeCls (aq) FeCls (aq) FeCls (aq) FeCls (aq) FeCls (aq) FeCls (aq)…
A: A balanced reaction in which no of coefficients of each ions or atoms should be equal in reactant…
Q: Which response contains all of the following that are NOT oxidation-reduction reactions? 2 SO2(g) +…
A: Oxidation-reduction reaction: A reaction in which electron transfer takes place between the…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: 24) Curved arrows used to represent the movement of electrons during bond breaking and/or bond…
Q: 10. Predict the unknown result of the following nuclear reaction: 238 234Th+a+X C. a. neutron 11. An…
A: Since, Balanced nuclear reaction means number of proton in both side is equal and also mass number…
Q: Benzoic acid of mass 1.20 g is reacted with oxygen in a constant volume calorimeter to form H₂O(1)…
A: we have to calculate the calorimeter constant
Q: 1. Predict the major product. a. b. C. CHO 8.8.8 N NH₂ NH₂ 1. EtOH, HCI 2. NaOH, H₂O NaBH3CN, HCI H…
A: We have to give the major product of the given reactions Introduction: Imine formation reaction,…
Q: draw the structure of 2-ethyl-1-pentene
A: In 2- ethyl - 1 - pentene; 2-ethyl represent that ethyl(-C2H5 ) is present at second carbon. Word…
Q: Three samples of three different gases are listed in the table below. All the samples contain…
A: The equation which gives the relation between pressure, volume, temperature, and the number of moles…
Q: lowing reactions and their AH values. What is the AH ed target reaction? 2CA + 2B C+DB+ E A2D2E ΔΗ =…
A: Given 2C --------> A + 2B ∆H=-259.......... 1 C+ D---------> B + E ∆ H=…
Q: A mixture of oxygen and neon gases, at a total pressure of 988 mm Hg, contains 7.46 grams of oxygen…
A: 1. we have to calculate the partial pressures of the gases
Q: The most common way to obtain hy drogen today is to extract it from which is a component of natural…
A: Hydrogen is the first element of the periodic table. It does not exist as a single atom, it exists…
Q: A saline solution in a gallon jug is known to be 0.4 moles per liter. Marsha takes 20 mL from the…
A: Answer: Statistic is the value that we calculate by taking a small sample from larger size of…
Q: Draw structures for reaction sequences
A: Given that, the reaction sequence is shown below We have to complete the above reaction sequence.…
Q: 2 equivalents Li THF, -78 °C Drawing
A: Alkenyl halides on reaction with metal give alkynes. Terminal alkynes on reaction with metal give…
Q: What is the pH of a buffer solution prepared from 5.63 g of NH4NO3 and 0.10 L of 0.18 M NHs? Ka for…
A: Since you have posted multiple questions, we will provide the solution only to the first question as…
Q: Calculate the amount of moles in 6.9341g of NaHCO3
A: To calculate the amount of moles in 6.9341g of NaHCO3, we first need to know the molar mass of…
Q: Asap 1. Choose true if the equation is balance false if not 4He + 4He = Li + p 2. Choose true if…
A: We have been given three questions.In two questions, we have been asked to determine whether given…
Q: What volume in mL of 0.220 M HBr solution is required to produce 0.0145 moles of HBr?
A: Given : Molarity of HBr solution = 0.220 M Miles of HBr = 0.0145 mol
Q: Consider the electrophilic addition of Br₂ to cyclohexene. The intermediate is a hybrid of a…
A: This question belongs to organic reaction mechanism.
Q: 1. Which of the following subshell designations are invalid? Select all that apply. A. 1s B. 1p C.…
A: There are 4 different types of quantum number - 1- principal quantum number 2- azimuthal quantum…
Q: A 50.8 g sample of brass, which has a specific heat capacity of 0.375 J-g -1.0C-1 ", is put into a…
A:
Q: 3 On the plot, select the location for pka2. % of total 100 90 80 70 60 50 40 30 20 10 0 4 H₂CO3(aq)…
A:
Q: Given that ΔHDis of Cl–Cl is 242.6 kJ/mol, what is the wavelength of light required to bring about…
A: Given: ΔHDis of Cl–Cl = 242.6 kJ/mol We have to calculate the wavelength of light required to bring…
Q: Draw a structure for 4- methyl-1,2- pentadiene. • Show stereochemistry
A: In the IUPAC name 4-methyl -1, 2 pentadiene, prefix 4- methyl represents that a substituent methyl…
Q: 21.9 Suggest reagents for the transformation shown below. OMe ? OH
A:
Q: Can you redo the calculation ?
A: To redo the calculation involved in the question.
Q: A geochemist measures the concentration of salt dissolved in Lake Parsons and finds a concentration…
A: We have to calculate the percentage of Lake Parsons that has evaporated
Q: Macmillan Learning Ne(g) effuses at a rate that is ratee ratech times that of Cl₂(g) under the same…
A:
Q: In a titration, 10.543 mL of a 1.112 M weak acid solution are placed in a 125 mL Erlenmeyer flask. A…
A: Suppose the weak acid is HA. The reaction of the titration is HA (aq) + NaOH(aq) →NaA(aq) + H2O(l)
Q: Calculate the pH for each case in the titration of 50.0 mL of 0.220 M HCIO(aq) with 0.220 M KOH(aq).…
A: Given that : The volume of HClO = 50.0 mL The molarity of HClO = 0.220 M The molarity of KOH = 0.220…
Q: Would NaBh4 work as a reagent for this reaction as well?
A: Answer: In this question we have to compare the strength of two reducing agents.
Q: 10. A particle of mass m is confined to move in one dimension on the domain 0≤x <∞, and its quantum…
A:
Q: Gold ions, Au3+(aq), can easily be made to precipitate out of a solution as gold metal. Simply bring…
A: In the reactivity or activity series of metals , gold (Au) is situated at the bottom the series i.e.…
Q: a. Using the average atomic mass, calculate the number of atoms present in 4.50 g of silver, Ag.…
A: 1 mole = 6.022 x 1023 atoms/molecules/ions Mass of 1 mole = molar mass
Q: A patient with respiratory alkalosis has a blood plasma pH of 7.50. What is the [H3O+] of the blood…
A: Given: The pH of the solution = 7.50 We have to calculate the [H3O+] for the solution.
Q: A radioactive substance has a half life of 120 years. If you have a 545 g sample of the substance,…
A:
Q: A 50.5 g sample of polystyrene is put into a calorimeter (see sketch at right) that contains 200.0 g…
A: Given: Mass of polystyrene = 50.5 g = mpTi of polystyrene = 85.4 ◦C = TpSpecific heat of polystyrene…
Q: A 100.0 ml solution of 0.25M C6H5NH2 is titrated with 0.5M HClO4. What is the pH and the volume…
A: Given that concentration of HClO4 = 0.5 M concentration of Ph-NH2 = 0.25 M Volume of Ph-NH2…
Q: To what main class of enzymes does the enzyme that catalyzes the following reaction belong? l-t…
A: In the given reaction, alcohol is formed from the corresponding aldehyde via reduction reaction.
Q: Propose a synthesis for the following transformation: 1- -one The transformation above can be…
A: Given that, a reaction scheme is shown below We have to carry out the above synthesis.
Q: What is the frequency of green light that has a wavelength of 533 nm? (c = 3.00 x 10⁸ m/s)
A: Given Wavelength = 533 nm c = 3.00 x 108 m/s
Q: Draw the condensed structural formula of ethyl methyl ether.
A: Since, In condensed structural formula, All the atom written in one line without any space left.…
Q: A solution is prepared at 25 °C that is initially 0.50M in propanoic acid (HC₂H,CO₂), a weak acid…
A:
Q: A student is studying the reaction: 2 CrO4 + 2 H30 Cr207 + 3 H2O If CrO4 is yellow and Cr207 is…
A: Solution- Given reaction: 2 CrO4 + 2 H3 ⇔ Cr2O7+ 3H2O
Q: 1.00 mol of perfect gas molecules at 27°C is expanded isothermally from an initial pressure of 3.00…
A: Given that, for an isothermal process of an ideal gas The number of moles is n = 1 mol, The…
Q: (a) How many milliliters of 0.145 M HCl are needed to neutralize completely 35.0 mL of 0.101 M…
A: Since you have asked multiple question, we will solve the first three question for you. If you want…
Q: b. Using the average atomic mass of the element, calculate the number of atoms present in 1287.3 amu…
A: We have given, Tungsten= 1287.3 amu Manganese= 219.76 amu We need to determine the number of…
Q: Consider the following system at equilibrium where AH = -111 kJ, and K = 0.159, at 723 K: N₂ (9) +…
A: The given equilibrium is N2(g) + 3H2(g) ⇔2NH3(g) According to Le Chateliar's principle, a reaction…
Q: 1. Reaction is endothermic 2. Reaction has an activation barrier of 100 kJ. The net enthalpy change…
A:
Q: What is the net ionic equation for the dissociation of hexacyanocobaltate(III) ion
A: We have find out the dissociation reaction.
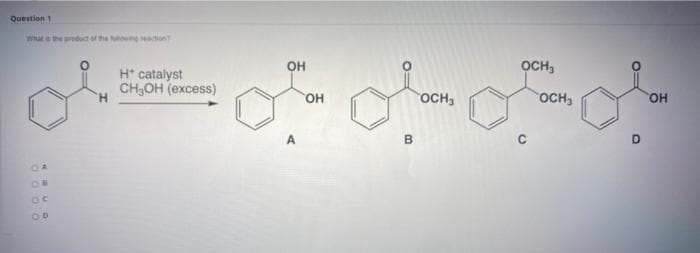
Step by step
Solved in 3 steps with 2 images

- 1. What is final product? 2. What are the 4 reactions & steps in mechanism (show product and mechanism steps)? 3. It is kinetic or thermodynamic?Give the reaction mechanism for ETHYNE and bromine solution. Draw and explain the steps of the reaction mechanism.give catalytic reaction of Cobalt naphthenate catalyst ( e.g., the reactant, product, reaction equation, phase, selectivity, promoter, catalyst support, operating temperature & pressure).
- Can you explain the mechanism please !! Its urgent !!Hello, I do not understand these questions for chemistry and I am stuck. May I get help please please?? Thank you. Draw the MAJOR organic product(s) for the following reactions. If no reaction occurs state no reaction in the box provided.The Products are from substitution and elimination reaction mechanism. explain the reaction mechanism. I got the product in substitution as CH2OCH3, i dont understand the shift in double bonds in other product. Also could you specify which ones are products from substitution and elimination...
- The Products are from substitution and elimination reaction mechanism.explain the reaction mechanism. I got the product in substitution as CH2OCH3, i dont understand the shift in double bonds in other product. Also could you specify which ones are products from substitution and elimination...Explain reason behind your option 1.All the following parameters would affect the rate and kinetics of the reaction exceptA.Solvent mediumB.Reaction temperatureC.Reaction timeD.Geometry and structure of the alkyl halideE.pHProduct and reaction mechanism?

