Q: Using the necessary inorganic reagents, propose the synthesis for 2,5-diphenylfuran from benzene and...
A: Here we to synthesize 2,5-diphenylfuran from starting material benzene and succinic acid.
Q: Draw Lewis electron dot diagram of compound CO2 to determine the molecular polarity by drawing the p...
A:
Q: A plot of Gibbs Energy against the extent of reaction has been constructed. Which point in the graph...
A: The plot of Gibbs free energy of reaction versus extent of reaction has minima at equilibrium point ...
Q: Take some water with a straw and put a few drops on plastic sheet. Put a small amount of one of the ...
A: Shape of the drop will change Or distort when we add insoluble solid like pepper and talcum powder b...
Q: The cell notation for the Galvanic cell is The Standard Reduction Potential are as follows: E°ca2*/c...
A: The cell given is Cd(s) | Cd2+ || I2 (s) | I-.
Q: Determine the concentration of A in 2A -> product second order reaction after 45 seconds. Use the gi...
A: For a second order reaction, Time, t = 45 seconds Initial concentration of A, Ao = 6.714 M Rate cons...
Q: In an SN2 reaction, which of the following nucleophiles will produce an ether? O "C=CH HO. "O-C-CH3 ...
A:
Q: Draw the Lowest Unoccupied and Highest Occupied orbitals for this. a) Lowest Unoccupied b) Highest O...
A: lowest unoccupied orbitals means the lowest energy level where electron density = 0 Highest occupied...
Q: -1 3) The IR absorption at 1630 cm belongs to (a) CH;-C=N )CH>-CH=CHICH, (c) CH2=CH-CH=CH-CH2 (d) H-...
A: The correct option are: Note: as per company policy we are supposed to answer first three questions ...
Q: HBr a)
A: protonation of etheral oxygen loss of a group as alcohol leading to the formation of carbocation at...
Q: Which of the following is the correct IUPAC name for the MAJOR substrate product formed by the dehyd...
A:
Q: Show how you could prepare 4-bromopyridine from 4-aminopyridine. Write the synthesis.
A: The conversion of 4-amino pyridine to 4-bromipyridine is needed to be shown. Since you have asked...
Q: Classify the conditions given as indicating that a reaction is at equilibrium, is not at equilibrium...
A:
Q: Hydrogen sulfide and oxygen react to form sultur dioxide and water, Ilike this! 2H,S(g)+30,(g) 2s0, ...
A:
Q: he molar conductivity at infinite dilution of hydrobromic acid, sodium acetate, & sodium bromide at ...
A: Given : We have to calculate the molar conductivity of acetic acid at infinite dilution.
Q: In a body-centered cubic unit cell, which part(s) of the 3D unit cell do the atoms occupy? corner ed...
A: A body-centered cubic unit cell consists of atoms arranged in a cube where each corner of the cube ...
Q: Estimate the net force exerted on your eardrum due to the water above when you are swimming at the b...
A:
Q: step(s). The SN2 reaction of chloroethane with NaC=CH has 3. O 1
A: Bimolecular nucleophilic substitution reaction (SN2): In this reaction the addition of the nucleophi...
Q: Acetylene and oxygen react to form carbon dioxide and water, like this: 2C,H,(g)+50,(g) → 4CO,(g)+2H...
A:
Q: explain the results of this experiment Table 1 Absorption Spectrum of Cobalt Chloride Using a Sp...
A: Given Wavelength Absorbance 400 410 420 430 440 450 460 370 480 490 500 510 520 ...
Q: Nitrogen dioxide decomposes according to the reaction: 2 NO2(g) 2 NO(g) + O2(g); Kp = 4.48 x 10-13 ...
A: Please find your solution below : Chemical equilibrium is a state in which there is no change in amo...
Q: 24) Identify the type of Organic compound shown 애
A: In the given structure carbon atom is bonded to oxygen atom by a double bond and a hydroxyl group (-...
Q: The cell notation for the Galvanic cell is Cd(syCd2+/l/½(syI" The Standard Reduction Potential are a...
A: Given cell notation is : Cd (s) | Cd2+ (aq) || I2 (s) | I- (aq) E0Cd2+|Cd = -0.403 V (anode) E0I2|...
Q: Aqueous sulfuric acid (H,SO,) will react with solid sodium hydroxide (NaOH) to produce aqueous sodiu...
A:
Q: How many ions of Al are present in 1 mole of Al2S3
A: Given that, 1 mole of Al2S3. We have to calculate the number of Al ions present in it. We know that ...
Q: At equilibrium, what is the value of ΔGrxn? < 1 < 0 = 1 = 0
A: Answer At equilibrium, value of ∆Grxn We know that For spontaneou...
Q: The reaction A-B is first order, and has a half-life of 375.1 s. If the concentration of A after 629...
A: t1/2 = 375.1 s t = 629.0 s [A] = 0.167 M [Ao] = (to find)
Q: Was there any physical change in the characteristics of trichloroethane when it was added to water?
A: Trichloroethane is micible in water. When it is added to water there is no any physical change.
Q: 1. The positivity rate in a certain group is 3.5%. Determine the probability that a batch will test ...
A:
Q: What is the purpose of the salt bridge in an electrochemical cell? To maintain electroneutrality To ...
A: Given : We have to tell the purpose of salt bridge in an electrochemical cell.
Q: Green plants use light from the Sun to drive photosynthesis, a chemical reaction in which liquid wat...
A: Given that, Moles of glucose= 1.5 mol Moles of water ?
Q: 3/5 Using a chemical equation to find moles of product... Wine goes bad soon after opening because t...
A:
Q: In aqueous solution initially prepared by mixing 225 g H2O (density is 1.0 g/mL), 0.3 M C, and 0.1 M...
A: We have mainly two equilibriums: A(aq)+H2O(l)↔B(aq)+C(aq) The equilibrium constant for this equilibr...
Q: The cell notation for the Galvanic cell is Cd(s)/Cd2+//I2(s)/I- The Standard Reductio...
A: Cell notation for the Galvanic cell is :- Cd(s)/Cd2+//I2(s)/I-
Q: v Draw Lewis structures for each of the following. Give the total number of valence electrons, selec...
A: Lewis dot structure of molecule can be predicted by using the octet rule where if valance electron e...
Q: 1. Which of the following match-ups about free energy is correct? A. Positive Gibb's = exergonic = n...
A:
Q: 1. Provide the major product(s) for the following reactions. Be sure to indicate stereochemistry whe...
A:
Q: To synthesize Tylenol, p-aminophenol will be acetylated using 1.4 equivalents of acetic anhydride. S...
A: Given: Mass of p-aminophenol = 2.1 g In order to synthesize, Tylenol, p-aminophenol will be acetylat...
Q: Identify the reaction type ) 2K t Brz>2KB. 9) 2 NaHCO3 > Na z COzt coz tH )Na t Hz0 > Hz + 2Na OL, H...
A: Answer Identify the types of reaction
Q: Put some water in cup. Sprinkle black pepper all over the surface. What does the pepper do? Tell you...
A: Polar molecules dissolve in polar solvents whereas non polar molecules dissolve in non polar solvent...
Q: What ending should each of the molecules below have? a) Alcohols -ol b) Aldehydes -al c) Carboxylic ...
A: What ending should be each of the molecules have ?
Q: There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of ...
A: All atoms of all elements and charges should be balanced in a balanced chemical reaction.
Q: (a) ŢLi + ? → 2He → 2;He (b) 14 C → 14 N+? (c) He ? + ¿n Al + 27 13 Не (d) 250 Cm → ?+g Sr + 4,n 38
A:
Q: n biological systems, an ACTIVE transport process is one in which an important chemical component is...
A: Since you have posted multiple questions as per guidelines we can answer only one per session . If y...
Q: A block of lead has dimensions of 4.5 cm by 5.2 cm by 6 cm. The block weighs 1587 g. From this infor...
A:
Q: At a certain temperature this reaction follows second-order kinetics with a rate constant of 1.04 M-...
A:
Q: What are the ΔHrxn and ΔSrxn values that indicate a spontaneous reaction at all temperatures? ...
A: For a spontaneous reaction : The value of ∆Hrxn and ∆Srxn = ?
Q: The distance between two atoms in a molecule is 148 pm. What is this distance in centimeters?
A: we know, 1 picometer(pm) = 10-12 meter(m) distance ( in m) = 148 pm × 10-12 m / 1 pm = 148 × 10-12...
Q: Question 18 The cell notation for the Galvanic cell is Cd(s)/Ca2*M2(s)/I" The Standard Reduction Pot...
A: This is a redox reaction. In this reaction, Cd oxidised to Cd2+ and I2 reduced to I- . Using Nernst...
Q: Complete the table. That is, fill in any missing entries in the second and third columns. Be sure ea...
A: Given : We have to fill the table.
Give the proper IUPAC name
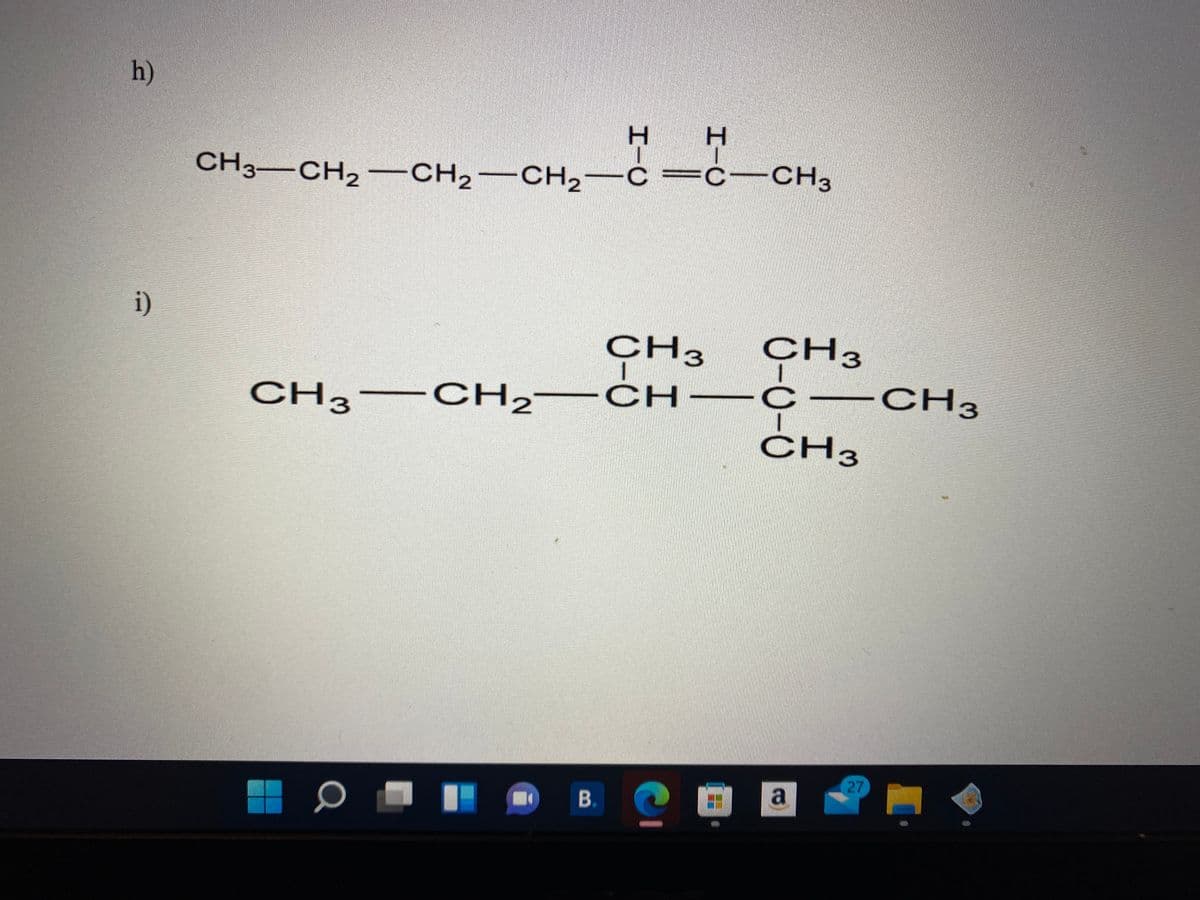
Step by step
Solved in 2 steps with 2 images


