Q: True or False The following is not a Hückel (4n + 2) aromatic system Cyclooctatetraene (C3H3)…
A:
Q: how to draw a trans-1,3-dimethylcyclohexane and its ring flip. (please also draw out each…
A:
Q: Directions: Name the following hydrocarbo 1. CH3 CH3 H3C-CH-CH-CH2-CH3 CH3 CH3 2. CHз — с- CH2 CH…
A: Rules for IUPAC naming :- Select the longest chain which contains carbon-carbon single bonds.…
Q: Label the sites of torsional and steric strain in attached conformation.
A: The increase in energy due to the interactions of the eclipsed groups is called torsional strain.…
Q: For the following molecule, determine the R/S configuration of each stereogenic carbon atom then…
A: To determine R/S configuration we would use the Cahn-Inglod-Prelog ( CIP ) rules. In this we assign…
Q: i. Is a methyl group at C-6 that is "down" axial or equatorial? ii. Is a methyl group that is "up"…
A: SOLUTION: Step 1: If a methyl group is substituted on C-6, it becomes methyl cyclohexane. The methyl…
Q: Identify the Newman projection that depicts the anti conformation of butane. = H. CH3 H A) I
A: Answer is : A) I
Q: соон HO. ÕH Ö CH2OH А. please put the missing groups to the Fischer on the right: 5
A:
Q: the wedges are supposed to be up, but why the both chair comformations point down? CH3 CH3 H3C CH3…
A: Two types of bonds exist in cyclohexane chair conformer. Axial and equatorial. When only a single…
Q: How many of the stractures show the identical stereoisomer? CH, CI H. CHs H Cl CH CH, 日 CI H CI H CI
A: First we will convert newmann-projection and wedge-dash molecule into Fischer projection, then we…
Q: Starting from the Newman projection below, rotate the back carbon to provide the structure in an…
A: Here, two bulky groups are -Et and -Cl. Fully eclipsed : the dihedral angle is 0°. This is least…
Q: N. H. Image I- :Z||
A: Here we have to determine the chiral and achiral molecules from the given amine molecules .
Q: draw most stable chair confirmation- add axial and equatorial to each- and provide ring flip
A: Different conformations of cyclohexane are formed by the rotation around the carbon-carbon single…
Q: Draw a structural formula of the SS configuration of the compound shown below. S CH3 CH2CH3 ÓH
A: Interpretation: A structural formula of the SS configuration of the given compound is to be drawn.…
Q: See attachment
A: Any of the spatial arrangements where the atoms in a molecule can adopt and freely transform…
Q: structure than the one given. a) H,C-0-c CH CH3 b) C-C C-CH3 H.
A:
Q: 2) Draw a hexane chain that has 2 carbon substituents and 1 halogen. Translate this drawing to the…
A: We need a six carbon chain out of which 2 carbon substituent and one halogen. I take 4- chloro-2,3-…
Q: Which of the following are aromatic?
A: For a compound to be aromatic, it should have the following characteristics. It should be a planar…
Q: Can you help me draw the enantiomer saw-horse conformation of this Newman projection?
A: Projection formula is a formal two-dimensional representation of a three-dimensional molecular…
Q: Draw curved arrows to show the movement of the electrons that result in formation of the given…
A: In the given reaction the alcoholic group in ethanol abstracts the proton from the hydronium ion and…
Q: Convert the attached representation to a Newman projection around the indicated bond.
A: Newman Projection illustrates the specific dihedral angle between proximal and distal atoms. It…
Q: Draw Newman Projections for the best and worst conformations of the structure shown, relative to the…
A: The best conformation is the one in which the steric repulsions are the minimum while the worst…
Q: e newman projection. (2) Rotate the carbon #1 to 60 degree for 6 s until a 360 degree rotation was…
A:
Q: Which of (a)-(d) shows the same compound as the following? CH3 I HIC CI CH₂CH3
A:
Q: (a) Draw C in its more stable chair conformation. (b) Convert D to a hexagon with substituents on…
A: Chair conformer: chair conformer is a stable conformer for cyclohexane compound. In this chair…
Q: 1. Circle the chiral carbon(s) in the following molecule.
A: Total 3 chiral carbons
Q: how to get from A to C1 and A to C
A: We have to get from: A to C1 : undersaturated to super-saturated solution with higher concentration…
Q: CH3 CH3 H Br H - Br NH2 СООН А В CH3 CH3 it Br H Br Br H -Br CHĄCH3 CH,CH3 D
A: R-S configuration(Absolute configuration): 1) If the 1 atom is different then given priority on the…
Q: 1. Which of the following is a chiral molecule? Encircle the substituents that make it chiral. H…
A: A. This is substituted allene. But in one side two H are present. That's why it is achiral. B. It…
Q: eck the box under each compound that exists as a pair of cis/trans isomers. If none of them do,…
A:
Q: H3C CH3 HI l H3C CH3
A:
Q: СООН H HO- H- он CH3
A: Flying wedge representation is the 3D representation. Here groups are present in above, below and in…
Q: ) Sighting down the C3-C4 bond, draw the gauche (60 degrees) and anti (180 degrees) Newman…
A:
Q: D. Rank (increasing) the substituents in each of the following sets: (a) -H, -OH, -OCH3, -CH; (b)…
A: A substituent with a higher atomic number takes precedence over a substituent with a lower atomic…
Q: Draw a hexane chain that has 2 carbon substituents and 1 halogen. Translate this drawing to the most…
A:
Q: (2 3 C. (a) Construct four models of the compound CH,CHCICHBRCH3. Take two models and hold them with…
A: (a) The Fischer projections formulas are given below for the first two models. It is clear from…
Q: Conformer E is a (cis/trans) isomer. A conformational ring flip of conformer E will yield conformer…
A:
Q: ght along the C2-C3 blond of 2-methylbutane: a)draw the newman projection of the most stable…
A:
Q: Which of (a)-(d) shows the same compound as the following? CH3 HC CI `CH2CH3 Oa. CI H CH `CH2CH3 b.…
A: We have to find out which one of the given compounds represents the given compound.
Q: C-H H. H H
A: The groups present in the upside is given as wedge bonds and downside group is given as dash bonds.…
Q: Build a Newman projection of the given compound viewed from C-2 to C-3 by making use of the given…
A:
Q: Part 1 out of 2 Build a Newman projection of the given compound viewed from C-2 to C-3 by making use…
A: First change into Fischer projection and into saw-horse projection now if change saw horse into…
Q: Name the following:
A: The given compound is heterocyclic compound, which has four membered ring with nitrogen and this…
Q: Answer this ques Draw the Newman projection of the below compound down the C2-C3 bond. Br Ill CH3 TI
A: We easily convert sawhorse projection to Newman projection. In Newman's projection front carbon is…
Q: How do you create a Newman projection that looks down both the c3-c4 and the c1-c6 bonds at the same…
A: Newman projections: The new conformations of compounds can be drawn and analyzed by Newman…
Q: Which of the following compounds is/are achiral? H;C CH; CH3 CH3 H;C CH3 2 1
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please…
Q: (e) Chlorocyclohexane (f) cis-1,2-dichlorocyclopentane (g) trans-1,2-dichlorocyclopentane
A: An asymmetric carbon atom is a carbon atom with four different substituents attached to it.…
Q: Answer the following questlon for Cis- 1-chloro - 3 - nethylegclolexane cyclohexane AG yalve (…
A: The given compound cis-1-Chloro-3-methyl cyclohexane The isomer with both methyl and chlorine…
Q: Which of (a)-(d) shows the same compound as the following? CH3 H CH2CH3 CI CI HC CH 父 H CI CI CH2CH3…
A:
Use curved arrows to draw a more stable version of the following
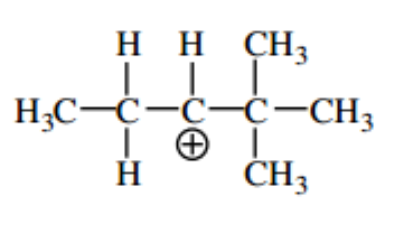
Step by step
Solved in 2 steps with 2 images

- What is the empirocal formula for each compound? a. AL2CL6 b. K2Cr2O7 c. C2H4 d.(NH2)2CNH e. CH3COOHRank the following compounds according to increasing positive character of the carbon atom. least positive C Most positive C CH3F CH3OH CH3Li CH3I CH3CH3 CH3NH2Draw the staggered and eclipsed conformations that result from rotation around the C–C bond in CH3–CH2Br.
- please tell me answer looks like for. example Ch3-CH=CTrue or False: If you perform a chair flip on cis-1,4-dichlorocyclohexane, the result is still called cis-1,4-dichlorocyclohexane.Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.



