Q: Calculate the mass of excess reagent remaining at the end of the reaction in which 180.0 g of SO2 ar...
A:
Q: You acquire two 1H NMR spectra on the same protein, one on a 500 MHz instrument and the other ...
A: We aquire two 1H NMR spectra on the same protein. First instrument = 500MHz Two peaks are apart by 8...
Q: ", heat HO
A: Given : We have to write the reaction .
Q: Using a chemical equation to find moles of product from moles ... Wine goes bad soon after opening b...
A:
Q: 03) Below are presented two 1H NMR spectra. Among the compounds pentan-3-ol, pentan2-ol, pent-3-amin...
A: HNMr tells us about the spiltting pattern of hydrogens in the compound.
Q: 2. Name the following hydrocarbon. Predict the hybridization (sp³, sp2, o sp) in the asteroid carbon...
A:
Q: Which of the following violates the octet rule? O CO2 O AICI4 O Co,2 SF6
A: Given : We have to tell which of the following is not following octet rule.
Q: oxygen, O2, via the following process: 2O3(g) → 3O2(g) The rate law for the decomposition of ozo...
A:
Q: The half-life of iron-59 is 44.5 days. How much of a 3.00-mg sample will remain after 222.5 days? a ...
A: The half-life of iron-59, t1/2 = 44.5 days Time elapsed, T = 222.5 days Initial mass of the sample, ...
Q: Which of the following substances contain a rigid arrangement of their constituent molecules? Sugar ...
A: arrangement of molecule in compound decide it's rigidity if molecule are close to each other then co...
Q: student mixed 50.0 mL of water containing 0.010 moles of HCI (weighing 50.0 g) with 60.0 mL of water...
A: Initial temperature = 25°C Final temperature = 26.20°C Change in temperature = 26.20 - 25 = 1.20°C
Q: You have a square planar PH4+ molecule: i) show your working to obtain the reducible representation?...
A:
Q: Explain the difference between powdered cocaine [hydrochloride] and crack cocaine in terms of method...
A: Explanation- 1.powdered cocaine - 1.The powered cocaine is also known as the hydrochloride cocaine. ...
Q: Consider the following reversible reaction at equilibrium. N2(g) + 3H2(g) = 2NH3(g) AH° = +9.22 x104...
A: We will apply Le-Chateliers principle to solve the above question.
Q: Give the number of lone pairs around the central atom and the geometry of NO3. O 0; trigonal planar ...
A:
Q: 3.67 X 2.3= 4.2 X 5= 6.3 X 2.4= 4 X 1.2= 12.5 X 4.7=
A:
Q: grams
A:
Q: 47.6 mL aliquot from a 0.5 L solution that contains 0.45g of MnSO4 (MW is 151 g/mol) required 41.9 m...
A:
Q: Arrange the following in order of increasing boiling point? Liquid A (vapor pressure at 25°C) = 23.8...
A: The intensity of the intermolecular forces determines the vapor pressure and boiling point. The stro...
Q: 1. Given the experimental information in the following table, answer the questions given. A → produc...
A: The kinetics data given is,
Q: H3C. CH3 CH3 CHa CH3 CH3 H CH CH CH 4. H. CH3 HO HO H3C CH HC CH, 8, On
A: The compound 7 and 8 are intermediate of lanosterol. The structure 7 and 8 may be different in any r...
Q: Discuss whether H-H bond should have IR activity in spectrum. If, what is the approximate wavelength...
A: We have to discuss whether the H-H bond should have IR (infrared) active or not. Hydrogen (H-H) is a...
Q: Follow proper technique when using a pipet: False O Using the pipettor, you should turn the wheel on...
A:
Q: Ammonium phosphate ((NH,), PO4 is an important ingredient in many fertilizers. It can be made by rea...
A: To calculate the mass of ammonium phosphate produced , we would first write a balanced chemical reac...
Q: Which point represents solid-liquid-gas equilibria? 24.. 1 atm Phase I. Phasc ... P. ... ... Phase I...
A: Phase diagram: The phase diagram represents the different phases of the substance and the pressure a...
Q: 1. Develop a detailed separation scheme for the separation and determination of the percent composit...
A: A mixture is a combination of two or more individual substances that may have uniform composition or...
Q: 2) What is true about the relationship of Kp and Kc for the reaction: 2 CH4(g) + 3 O2(g)=2 CO(g) + 4...
A:
Q: What is the purpose of the wash with water using separatory finnel? Bicarbonate?
A: The aqueous solution of the sodium bicarbonate having formula NaHCO3 and sodium carbonate having for...
Q: Which of the following violates the octet rule? SF6 CO2 O AICI4 O Co3?
A: Concept is based on octet rule:
Q: What is the pH of a 500 mL buffer containing 0.200 moles NH3 and 0.350 moles NH4CI? Kb = 1.8 x 10-5 ...
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The buffe...
Q: On analysis, an equilibrium mixture for the reaction 2H2S(g) 2H2(g) + S2(g) was found to contain 1.0...
A: The equilibrium reaction given is, => 2 H2S (g) ⇔ 2 H2 (g) + S2 (g) Kc = 3...
Q: 6) Draw and build 3 isomers for the for Label one pair as constit b. Label another pair as co a. C. ...
A: The compound with identical molecular formula but with a distinct structural formula is known as a c...
Q: A reaction is prepared by adding 1.37 mol each of H2, 12, and HI gases to a 1.0 L container. They ar...
A: Given that : Since you have asked multiple question, we will solve the first question for you. If yo...
Q: At 110°C, which compound has a vapor pressure of 80 kilopascals? (1) ethanoic acid (2) ethanol (3) p...
A: Since you have posted multiple questions, we are entitled to answer the first only. 1) To identify: ...
Q: Molecular oxygen, O2, has a collision diameter of 3.57 x 10-10 m. Calculate the mean free path, 2, f...
A:
Q: Draw the Lewis structures for the following compounds, identify its molecular geometry, and redraw t...
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simpl...
Q: For your research project, you need to use isotopic labels. For this you buy chlorine gas (Cl2) that...
A: The basic constituent of a substance is known as an atom that has a nucleus consisting of protons an...
Q: Greenhouse gases absorb (and trap) outgoing infrared radiation (heat) from Earth and contribute to g...
A: Given : We have to tell which of the following is a greenhouse gas.
Q: Which compound has the greatest ionic character? O RbF O LiF O NaF O CSF
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: Compound A Compound B Compound C CI Step 1 Step 2 Circle one: Circle one: oxidation oxidation reduct...
A: The oxidation number represents the loss or gain of electrons to the respective elements of the comp...
Q: Twenty-five mL of 0.100 M lactic acid (K, = 1.4 x 10) is titrated with 0.097 M KOH. %3! a) What is t...
A:
Q: Draw the structure of both diastereomers. If the structure cannot form diastereomers, explain why. С...
A: The solution is given below -
Q: Given these molecular weights of only nonpolar and non-ionic molecules consisting of only hydrogen a...
A: Melting point is directly proportional to number of the carbon atoms. Longer the carbon chain higher...
Q: 11. Draw the Lewis structure and calculate the formal charge for oxygen atoms in an HCO3 ion? Show y...
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simpl...
Q: Determine the concentration of the stock solution in molarity from which 50.0 mL was taken in order ...
A:
Q: 9. Show the mechanism of the reaction of benzene with nitric acid and sulfuric acid to yield nitrobe...
A: Organic reactions are those in which organic compounds react with each other to form organic product...
Q: Your answer is partially correct. The following data are obtained for the decomposition of N205 at 4...
A:
Q: H,0 H,0
A:
Q: 1. For the reaction below, determine the equilibrium concentration of all species. Kc=1x1020, [A]ini...
A: Given, Kc = 1x1020 [A]initial = 0.4 [B]initial = 0.6
Q: wavelength
A:
Step by step
Solved in 2 steps with 2 images

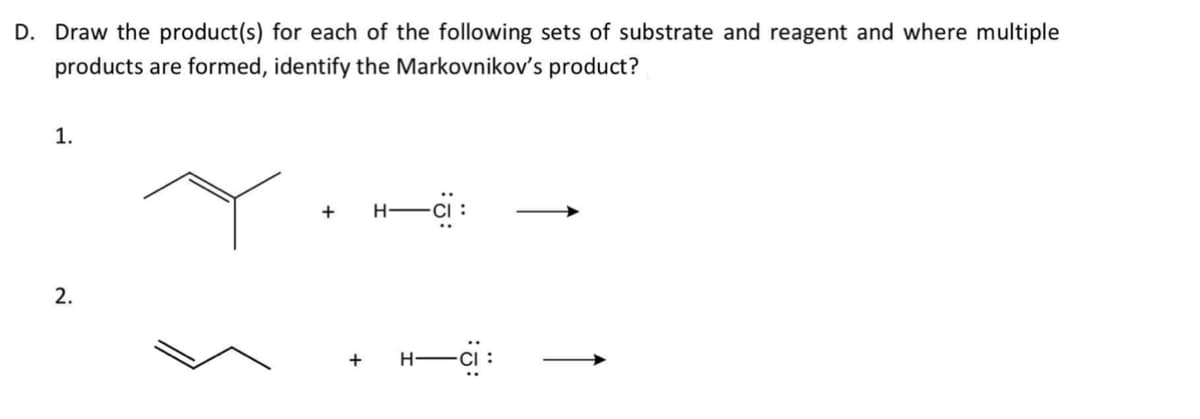
- A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of Η2 is absorbed. On hydrogenation overa palladium catalyst, 3 equivalents of Η2 are absorbed. (a) How many degrees of unsaturation are present in the unknown structure? (b) How many triple bonds are Present? (c) How many double bonds are present? (d) How many rings are present? (e) Draw a structure that fits the data.Draw the reaction mechanism that shows BH3 adding to the alkene. Why is the product anti-Markovnikov?Which product is more polar the intermediate or the final product? What will happen with the product spots if we use only hexdane? What will happen with the product spots if we use acetone?
- 44. Sterically hindered bases produce what is the major product? Zaitsev product Hoffman product Markovnikov product Anti-Markovnikov productWhich factor will promote an Sn1 reaction?1. Strong base 2. nonpolar aprotic solvent 3. 1 degree substrate 4. good leaving groupDrawing the Oxidative Cleavage Products from an Alkene Draw the products when each alkene is treated with O3 followed by CH3SCH3.
- (36) What pairs of Starting Materials result in the given Robinson Annulation product?What is the reagent R1 in this reaction sequence?45. A small, unhindered base:; bulky, hindered base: Hoffman product; Zaitsev product Zaitsev product; Zaitsev product Hoffman product; Hoffman product Zaitsev product; Hoffman product



