Q: Convert 398nm to m.
A:
Q: :0 + N N C ZH N H+ cat.
A: When ketone react with secondary amine it will form enamine
Q: 1) N-H /H® 2) 3) H20 / HO ZHN NABH3CN
A:
Q: H :0: OFF H N-H : OH NANON N-CH3 :0: HO: GUN :0: :0: OHM :0: :N-CH H₂C TAY
A:
Q: Number Log log Crooo) -3 log C1o).-a 1000 0.01 Ix 104 -4 log (o-02), -1-69 .02 300 log (300 ), 2.47…
A: pH is the measure of acidity whereas pOH is the measure of basicity. For a solution, if pH < 7…
Q: а. NH3 NaCN H HCI b. Br EtO2c co,Et `CO2Et Br HCI NO NaOEt H20 c. E1cb OP
A: As per the regulations only the first three subparts of a question could be answered.
Q: c) Ce chemPad Help XaX 1L Greek (d) Mg chemPad O Help X.X"→= Greek (e) Re chemPad O Help Greek -
A: elements are the pure form.
Q: NH₂ 1. NaNO₂, HCI 2. NH₂ -N(CH3)2 NaBH3CN [H₂SO4] FF HH NH 1. excess Mel 2. Ag2O, H₂O, heat CHCI 3…
A: CHCl3 in presence of NaOH give dichloro carbene
Q: 1. NH3 2. NaCN, HCI H 3. H*, H20, A
A:
Q: शिकाल्ब गन वेगण्णंणंद भएवपत्कोल- ०रावळीना जबवतीक व ए्रवकाट ेतपनीब्ल. ब्बकण केवोरी जटवत्वीनाह ल…
A: Balanced the following redox reaction under basic conditions ---
Q: IA) + H,0 oH oH (B) + H,0
A:
Q: Cyanidin chloride (C15H11O6Cl) contains the cyanidin ion, a pigment found in many berries. Calculate…
A: Here, we have to calculate number of moles of cyanidin chloride present in 7.2 mg.
Q: 1.0L
A: “Since you have asked multiple questions, we will solve the first question for you. If youwant any…
Q: H2C-C-0- H2 2x 105 3. H,c-c-00 -Br H,C Br 5x107 -Br
A:
Q: 8. BaCh(aq) +Na;PO4(aq) yes
A: The reactants given are, 1) BaCl2 (aq) 2) Na3PO4 (aq)
Q: H HH CH3 H. PPhs audy H. H.
A: The Wittig Reaction is reaction an aldehyde or ketone with the ylide produced from a phosphonium…
Q: 1) CI (1 equiv) cat. AICI3 2) ΗΝΟ, cat. H2SO4 CI
A: Here the reactions are the electrophilic substitution .
Q: 15N + H → 1C + + în
A:
Q: NH2 1. NANO2, H;O+ 2. H20, heat
A: The products for the given reactions has to be predicted.
Q: ÇI 3 Na SCH,CH, DMF, 0" C CHJ NaNO
A:
Q: C0(g) + H20() CO2(g) + H2(g)
A: Given data are as follows: Temperature of the system = 298 K CO moles = 1.8 mol Standard absolute…
Q: A student mixed sugar and water during a lab unt the sugar dissolved. This Is an example of a(N) O…
A: The formation of a solution can be either exothermic or endothermic process. If energy is released…
Q: (c) (i) Ph,NH (ii) Et,NH (sealed tube) CI Ph
A: We have to give the structure of the product as follows in step 2:
Q: H20, H2SO4 HgSO, OH OH OH HO OH но II IV
A: The reaction given in first image is hydration of alkyne and the reaction in second image is…
Q: O 1.0. 1.2. 7.0, 13.9 O 1.0. 3.2. 7.0, 10.9 O 1.0. 1.2. 7.0, 10.9
A: [HCl] = 0.10 N Volume = 20 mL milliequivalents of HCl = 20 mL * 0.1 n = 2.0 meq Before scaling 2.0…
Q: Convert 100.6°F to °C and K
A:
Q: 16) 3.1 x 10 kL+534x 10 kL-
A: In the given question, we have to find out 3.1×104kL + 5.34×106kL
Q: 1) ТСРВА 2) а. „MgBr H,C CH2 H;C CH3 b. H30* 3) Н2, Раc H;C CH3
A: Hydrogen in presence of Pd and C behaves as a reducing agent. mCPBA is a peroxy-carboxylic acid used…
Q: Perform the indicated conversion: 2.50 × 103 cal = _____ J
A: Unit conversion convert different units of measurements for the same quantity. Energy can be…
Q: (convert)( g HCN) → (? HCN) noth
A: 1 mol/L HCN means 1 mole of HCN is present in 1L of the solution.
Q: 28 H- H H H c-H 39 H H. H 30 (CH10)
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: 63. 65. 67. H CI 이 H CI CI H. H CI, H 사 H CI CI H H H CI 64. 66. H CI H. 이 보이 CI H H CI H CI CI H
A: Since, you have asked multiple questions, we will solve the first three question for you...if you…
Q: :0-N=0 o=N-o > 0=N-0:
A:
Q: KM, O4, KOH
A: KmnO4 (Potessium Permanganate ) is a strong oxidising agent ,so it can oxidises other molecule
Q: OH H,SO, 180°C
A: Initially H2SO4 protonates the alcoholic oxygen and forms a leaving group. Leaving group depart…
Q: Would a car be able to travel more miles on a gallon of gasoline or a gallon of gasohol?
A: From the given we need to find out among gasoline or gasohol is better for a car to travel.
Q: Determine the molecular mass of the following compounds:
A: The molecular mass of the given molecule is equal to the sum of masses of a number of elements…
Q: Convert CuCl2.6(H2O)2 to whole numbers
A: Given,CuCl2.6*(H2O)2Find and multiply the decimal number in the molecular formula with a least…
Q: জাতল এ তखर जाक! তोजल व To (s)+ oct ca9) > J03c११) +C\Cqq) हिकार रीए नवच०ण्क उच्हबेo। Blancehe…
A:
Q: OH H,SO., 100°C
A:
Q: [OH ] [H'] 1.0x103 3.6x10 5 pH РОН 6.21 4.84 5.1x10 3
A:
Q: 10. H2 (g) + C¿H4 (g) → C¿H6 (g) 11. 2C,H, (g) + 702 (8) → 4CO2 (g) + 6H2O (g)
A:
Q: molecules O CG-FID O GC-MS O LC-MS O IC-MS O IC-EC analysis
A: LC -MS is the best chromatography instrument for analysis of soluble protien molecules.
Q: CH2 II NH HCI H20 O. H H3C H H3C HO,
A:
Q: The warmest temperature ever measured in the United States is 134 °F, recorded on July 10, 1913, in…
A: In United States, the wannest temperature measured is 134°F. The conversion of 134°F into °C is as…
Q: CI 1) NaNH, / NH3 (liquid) 2) H,00 ČH3
A:
Q: n = 4 n = 3 n = 2 -0.1361 x 10-18 J -0.2420 x 10-18 J -0.5445 x 10-18 J n= 1 -2.178 x 10-18 J
A: To find the wavelength of the light emitted we have to find the energy difference between the two…
Q: Convert 1228 g/Lg/L into kg/MLkg/ML
A: A physical quantity can be expressed using two things. One is the magnitude of the quantity and…
Q: Mg(s)+N2(g)→Mg3N2(s)Mg(s)+N2(g)→Mg3N2(s) Express your answers as integers separated by commas.
A: Note: Assuming that the question is to balance the given reaction.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

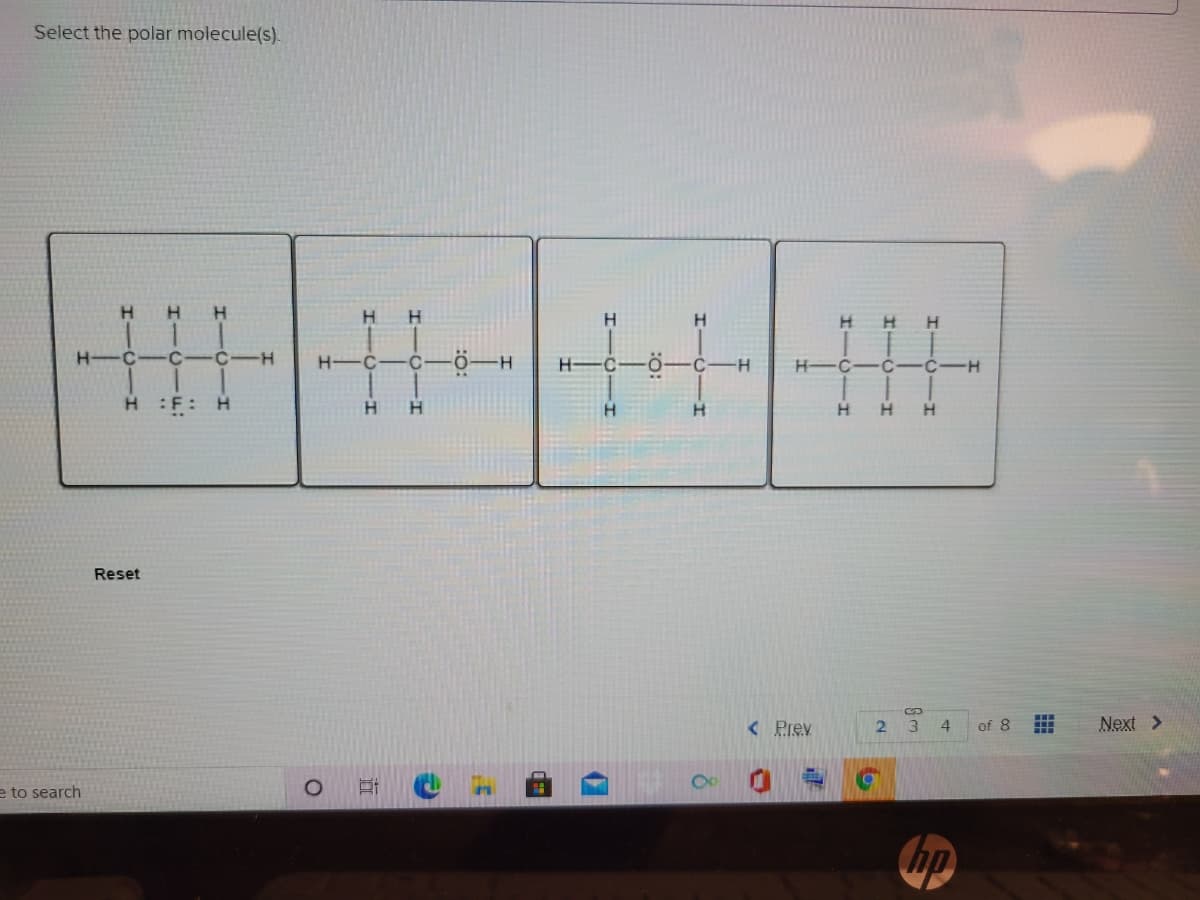
- H-p, h-bi,h-n,h-ag,h-p least polar?pls help ASAP! “which term best describes the relationship between the following two molecules?”Consider the following flat drawing of methane (CH4) . a. What is HCH bond angle implied by this drawing if you assume it is flat? b. Are the electron domains of this flat CH4 spread out as much as possible? c. Use model materials to make a model of CH4 (methane). If you assembled it correctly, thefour bonds (bonding electron domains) of your model will be 109.5° apart. d. In which representation, the drawing above or the model in your hand (circle one) are theH’s of CH4 more spread out around the central carbon? e. Confirm that your model looks like the following drawing. The wedgebond represents a bond coming out of the page, and the dash bondrepresents a bond going into the page f. You will often see methane drawn as if it were flat (like on the previous page). Why is thismisleading, and what is left to the viewer’s imagination when looking at such a drawing?
- Which ones are polar and which ones are non polar?1. Please explain how an FTIR works 2. Why it's so important and useful for organic chemistryRepresent the bonding in each molecule or ion by drawing the orbitals (hybridized and unhybridized) of each atom in the bond. Label the σ and π bonds and label each bond by the orbitals that it is made from. For instance, the C–H bond in CH4 would be made from the overlap of: C (sp3) – H (1 s). You may draw the hybridized sigma orbitals as sticks and the unhybridized p-orbitals as lobes for clairity and ease. a. HONO b. CH3 CCH c. C3 H4 d. C2 O4 2-
- - What orbital(s) are the lone pairs of electrons on the O atom placed? - What orbital is the lone pair of electrons on the N atom placed? - What type of bond(s) are formed between Ca1 – C? - What type of bond(s) are formed between C = O? - What type of bond(s) are formed between C – N? - What type of bond(s) are formed between N – Ca2? - What type of bond(s) are formed between N – H? - What is the angle between Ca1 – C = O? - What is the angle between Ca1 – C – N? - What is the angle between O = C – N? - What is the angle between C – N – Ca2?Draw the lewis structure of SO2 ( best resonance) and 2nd best resonance showing the shape and bond angles of each one, the 3D structure with polar bonds or bonds dipole of each one and the molecular polarity. Here is an example of what I want the answer to be like:pls help ASAP, thanks! “rank the following bond angles from least to greatest”
- I need to make a skeletal strcuture for this formula and i have no idea how. im very new to organic chem and i did the lewis structure but idk how to turn it into a skeletal structure. help please (CH3)3COO(CH2)2CCOCH2NHCHOHow many possible iosmers does K2[Pt(OCN)4] have? Please draw them.Can u explain it in detail what it means and how to find and draw the compound structure thank u

