H. H. C= H. || H. C= H. H H The diagram above shows two resonance structures for a molecule of C6H6. The phenomenon shown in the diagram best supports which of the following claims about the bonding in C6H6 ? (A) In the C6H6 molecule, all the bonds between the carbon atoms have the same length. (B) Because of variable bonding between its carbon atoms, C6H6 is a good conductor of electricity. (C) The bonds between carbon atoms in C6H6 are unstable, and the compound decomposes quickly. The C6H6 molecule contains three single bonds between carbon atoms and three double bonds between (D) carbon atoms.
H. H. C= H. || H. C= H. H H The diagram above shows two resonance structures for a molecule of C6H6. The phenomenon shown in the diagram best supports which of the following claims about the bonding in C6H6 ? (A) In the C6H6 molecule, all the bonds between the carbon atoms have the same length. (B) Because of variable bonding between its carbon atoms, C6H6 is a good conductor of electricity. (C) The bonds between carbon atoms in C6H6 are unstable, and the compound decomposes quickly. The C6H6 molecule contains three single bonds between carbon atoms and three double bonds between (D) carbon atoms.
Chapter1: Lewis Structures
Section: Chapter Questions
Problem 3EQ: 3. The skeleton of chloromethane is
__________________
The central carbon atom is bonded to...
Related questions
Question
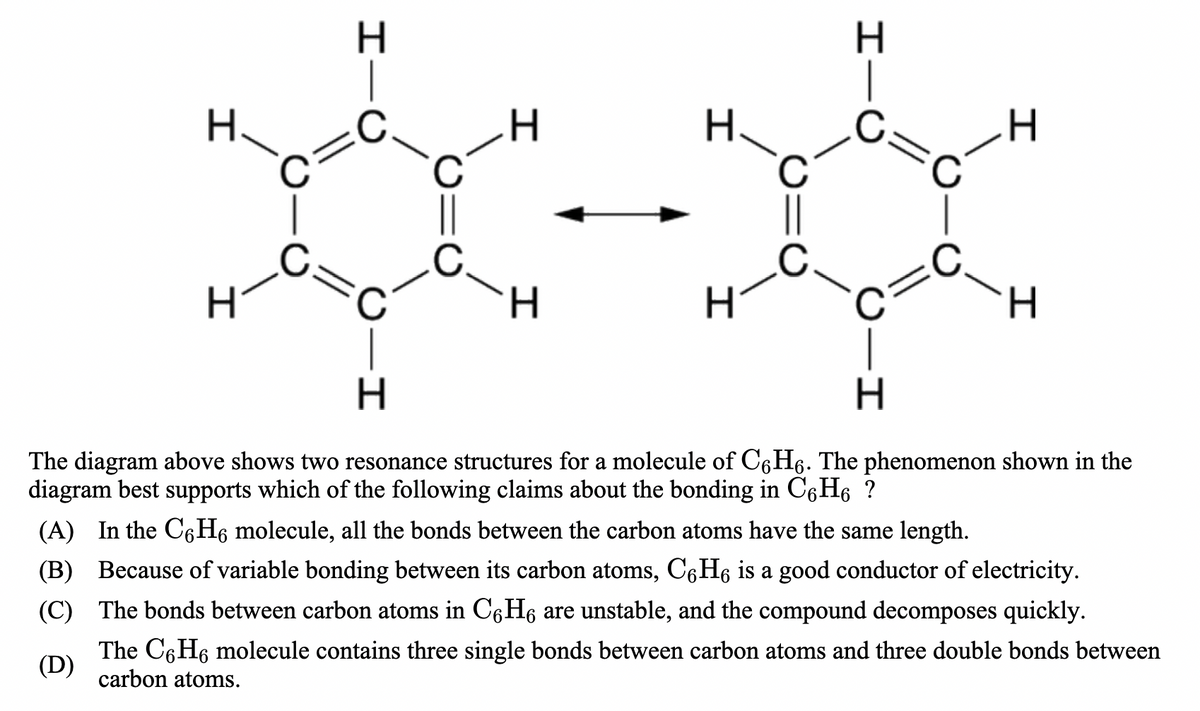
Transcribed Image Text:H.
H.
H.
H.
H.
The diagram above shows two resonance structures for a molecule of C6H6. The phenomenon shown in the
diagram best supports which of the following claims about the bonding in C6H6 ?
(A) In the C6H6 molecule, all the bonds between the carbon atoms have the same length.
(B) Because of variable bonding between its carbon atoms, C6H6 is a good conductor of electricity.
(C) The bonds between carbon atoms in C6H6 are unstable, and the compound decomposes quickly.
The C6H6 molecule contains three single bonds between carbon atoms and three double bonds between
(D)
carbon atoms.
IIU
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning