Chapter6: Organic Chemistry
Section: Chapter Questions
Problem 69E: Explain why the formula CH3CH2CH3 cannot mean: CHHHCHHCHHH
Related questions
Question
3.93 When combined, aqueous solutions of sulfuric acid and
potassium hydroxide react to form water and aqueous
potassium sulfate according to the following equation
(unbalanced): Determine what mass of water is produced when a
beaker containing 100.0 g H2SO4 dissolved in 250 mL
water is added to a larger beaker containing 100.0 g
KOH dissolved in 225 mL water. Determine the mass
amounts of each substance (other than water) present
in the large beaker when the reaction is complete.
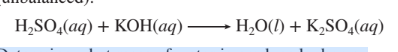
Transcribed Image Text:H.SO,(aq) + KOН ад) — Н,О() + K,SO,(aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning