Q: The first step in the mechanism for the reaction of an amine with a carboxylic acid to produce an…
A: The first step involved in the reaction between amine and carboxylic acid is given below
Q: Electrolysis of Cr³+ (aq) produces Cr²+ (aq). If you had 350 mL of a 0.12 M solution of Cr³+ (aq),…
A:
Q: State the Similarites between poalar and nonpolar molecules. What are the differences between an…
A: Similarites between polar and nonpolar molecule
Q: Details Trial 1 A. Preparation of Ca²+ Standard Solution Mass (g) CaCO3 Amount (mmoles) Ca = Amount…
A:
Q: data stream comprises the characters A, B, C, D and E with respective probabilities of 0.12, 0.21,…
A:
Q: 3. Write the product and the mechanism for reaction: OH H3PO4 H3C CH3
A: In this question, we will draw the structure of the product with mechanism. In this reaction…
Q: The first step in the mechanism for the reaction of an amine with a carboxylic acid to produce an…
A: Carboxylic acids are related to the class of organic compounds, from where the carbon (C) atom is…
Q: Place the following orbitals in order of increasing energy 1s,3s,4s,6s,3d,4f,3p,7s,5d,5p
A: Afbau's principle shows the increasing energy of the various orbitals.
Q: Which of the arrows points to an atom that is 30 (tertiary) and sp³ hybridized? с d a mamana www
A:
Q: Balance the following oxidation-reduction occurring in acidic solution. TO + Cl2(s) --> TO2-1 (aq) +…
A: The redox reaction occuring in acidic medium given is TO (g) + Cl2 (s) → TO2–1 (aq) + Cl–1 (aq)
Q: hond How many form from are needed to prepare 1.5 L af 9 1.8m eahpoy Solution ?
A: Molarity is a concentration term for a solution. The molarity of the given solution is defined as…
Q: Part A For the reaction at 225 °C the equilibrium contar Express your answer to two s HÅ [NH3] =…
A:
Q: The rate constant k for a certain reaction is measured at two different temperatures: temperature k…
A:
Q: Consider the following reactions. In which cases is the product formation favored by decreased…
A: In which cases (from the given reaction) is the product formation favoured by decreased pressure ?
Q: 2. Write the product and the mechanism for reaction. Write the names of starting material and…
A:
Q: The first step in the catabolism of most amino acids is the removal of the nitrogen atom by transfer…
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Given the concentration values of both reactants and products and the equilibrium constant, explain…
A:
Q: Use your graph equation to calculate the expected mass loss for your Epson salt sample, then use…
A: Mass of the sample (x) 1.03 3 1.06 3.16 Mass loss on heating (y) 0.59 0.56 0.47 1.62
Q: 5. Write at least 4 different oxidation reactions for any hexose.
A: Hexose : Monosaccharides with six carbon atoms are called hexose. The chemical formula of hexose is…
Q: For the following general reaction, rate = k[A]2 and k=1.3 x 10-2 M¹.5¹; A+B 2C Use this information…
A:
Q: 2.3 Fill in the missing starting material, reagents or products for the following reactions: (4)…
A: It is SN2 product
Q: Fire is an example of X a. exothermic; entering b. exothermic; leaving c. endothermic; entering d.…
A: Exothermic reaction and endothermic reaction: Exothermic reaction : Exothermic reactions are…
Q: White sand is made of silica (silicon dioxide, or SiO2), which has a density of 2.20 g/cm3. How many…
A:
Q: stry A 0.668-g sample of an unknown gas occupies 0.407 L at 398 K and 0.335 atm. A. Calculate and…
A:
Q: 2. Answer parts a-e using the skeletal structure of the molecule shown below. NB # a. Add all…
A: #2:(a): We have to add H atom in accordance with the octet rule. According to the octet rule, C atom…
Q: You have a 0.10 M aqueous NH3 solution. Calculate a) the number of moles b) the number of grams of…
A: H-H equation can be used to pH determination as, pOH = pKb + log [moles of conjugate acid] / [moles…
Q: Select the equilibrium constant expression for each of the following reactions: Part A 2HBr(g) H₂(g)…
A:
Q: Time felt 0:04 An aqueous of gold (III) nitrate Au(NO3)3 was electrolyzed with a current of 0.015 A…
A:
Q: 18. The two compounds below are very similar, yet they have a heat of combustion that differs by…
A:
Q: Using the equation for the trendline line, determine the values for ΔH° (kJ/mol) and ΔS° (J/mol⋅K).…
A: The thermodynamic relation between Gibbs free energy, enthalpy, entropy and temperature is,…
Q: Consider the following reaction: CH₂(g) + H₂O(g) = CO(g) + 3 H₂(g) AH = 231 kJ predict the…
A: Dear student , since you have posted multiple parts questions we are allowed to solve only first…
Q: which of the following isomers will be converted to salt a more rapidly quaternary N(CH3)₂ ~N(CH3)2…
A:
Q: Adding hydrogen to pentene leads to the formation of pentane True Error
A:
Q: 8. K/U Write the acid-dissociation constant expressions for each of the following weak acids: a.…
A:
Q: Arrange the following compounds in order of increasing R, on the TLC plate developed with in 60:40…
A: A question based on analytical separation that is to be accomplished.
Q: Chapter 2 Homework Molecular Representations H ||| 7. The lone pair on oxygen in the following…
A: IUPAC Name :
Q: Consider the following reaction: H2 (g) + I2 (g) ⇌ HI(g) If Kc = 196, what will be…
A: The equilibrium constant of a reaction is the ratio between the concentration of products to the…
Q: 17. Which CORRECTLY states the Raoult's Law? A. The total of the mole fraction of the solvent in the…
A: The solutions are given below -
Q: 55 Cs 112.9055 Fr 87.62 8.905 91.22 92.9054 905.94 36 57 72 73 74 Ba La Hf Ta W 117.34 138.0055…
A: Given options are : a. K < Fe < Li b. Rb > Na > Be c. Fe < O < P d. Sr < Se…
Q: The first step in the mechanism for the reaction of an amine with a carboxylic acid to produce an…
A: The first step in the mechanism for the reaction of an amine with a carboxylic acid to produce a an…
Q: How many sulfur atoms do you need for this reaction to produce 1 molecule of CS₂ and 2 molecules of…
A: The Chemical reaction follows the law of conservation of mass. According to the law of conservation…
Q: 1. Write the product, side products and the mechanism for reaction. Write the names of starting…
A:
Q: What is the oxidation state on the central carbon in the following compound? 0 HẠN CNH, 00 +1 +2 +3…
A: Given, Compound 1.H2N -CO-NH2 Compound 2. H3C-CH(CH3)-CH3
Q: 4. Write the product and the mechanism for reaction. Write the names of starting material and…
A: Alcohol and carboxylic acid reacts under acid medium to give ester, this reaction is often called as…
Q: Question 7 Three organometallic reagents are reacted with (R)-2-methyl-2,3-dihydro-4H-pyran-4-one to…
A:
Q: earch project i ke a comparati emical equatio
A: Inert atmosphere will be created using nitrogen or helium.
Q: Calculate the number of ATP generated from one saturated 18-carbon fatty acid. Assume that each NADH…
A: 1 NaDH produces 2.5 ATP while 1 FADH2 produces 1.5 ATP.
Q: is the number of grams of aluminum sulfate needed to prepare a 0.15 titer solution, 32 = 27 = Al)…
A: Given that - Molarity of the solution = 0.15 M Atomic mass of different elements are - Al = 27 ,…
Q: What is the oxidation state on the central carbon in the following compound? CH₁ H₂C-C-CH₂ 0-2 0-1…
A: Here, we have to find the oxidation states of the central carbon atoms of the given compounds.
Q: What is the relationship between the following molecules? FI same compound constitutional isomers…
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
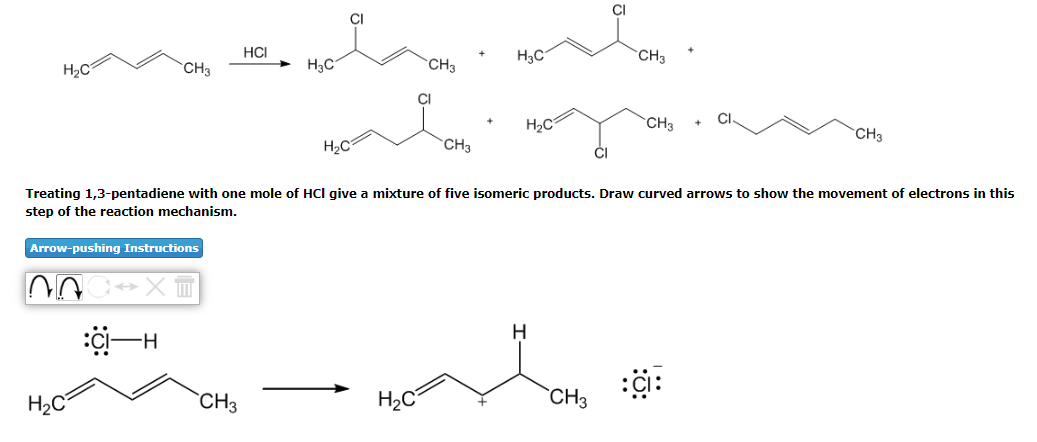
Step by step
Solved in 3 steps with 1 images

- When 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observedWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observed.(c) Use a Newman projection of the transition state to predict the major product of elimination of (2S,3R)-2-bromo-3-phenylbutaneTrue or false? 2-bromo-2-methylpropane undergoes SN1 rather than SN2 because the nucleophile experiences steric hindrance, and a stable carbocation can be formed. When hydroxide ion reacts with 1-chloropropane the main product is 1-propanol.
- Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpConsider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:Identify the reagents needed to come up with the given compounds. Pls Separate the reagents with a comma and a space. (ex. HNO3, H2SO4). For a multi-step reaction, separate the reagents with a semi-colon. (ex. HNO3, H2SO4; CH3Cl, AlCl3) There should be 9 reagents
- When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.) Propose a mechanism to account for formation of both A and Bfor each reaction give the expected substitution product and predict wether the mechanism will be first order (SN1) or second order (SN2) a) 2-chloro-2-methylbutane + CH3COOH b) isobutyl bromide + NaOMe c) 1-iodo-methylcyclohexane + CH3CH2OHWhen Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.Show why A predominates at -15 °C and B predominates at 60 °C.
- First Writedown which reaction it is? SN1, SN2, E1? Write a stepwise mechanism for the following reactions showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show all necessary lone pairs and formal charges.1. Do propose a complete substitution mechanism of the following reactions, by looking at the given conditions determine whether it is unimolecular or bimolecular and pay attention to stereochemistry; (i) (1-bromoethyl) cyclopentane reacted with ethanol and then dimethylformamide, (ii) 2-chloro-1-cyclopropylbutane reacted with propanol.Draw the four resonance structures formed during bromination of methoxybenzene, CH3OC6H5, with Br2/FeBr3 to give para-bromomethoxybenzene and then circle the most stable resonance form.

