Q: There are three sets of sketches below, showing the same pure molecular compound (ammonia, molecular…
A: Melting point of NH3 =-77.7 oC Boiling point of NH3 = -33.3 oC To choose how ammonia might look like…
Q: 14 2. Draw a complete Bohr-Rutherford diagram showing all the parts of the atom for ¹4, N.
A: Atom is in spherical shape in which protons and neutrons are present in the nucleus and electrons…
Q: What is true about atomic orbitals? All atomic orbitals have similar energy as the electron can only…
A: Atomic orbital describes the wavelike behavior of electron in an atom. Atomic orbitals are the…
Q: Derive the electron configuration and draw the orbital notation for sulfur, 16 electrons.
A: Electronic configuration for sulfur = ? Orbital notation for sulfur = ?
Q: Which of the following compounds has the lowest dipole moment? Carbon tetrachloride Fluoroethane…
A: Two questions based on introduction to organic chemistry. First question is about dipole moment and…
Q: 1 1 a. Use the data collected by experiment to calculate a molar enthalpy of reaction for methanol.…
A: The percentage error is calculated by the ratio between the difference between the theoretical and…
Q: HOW DO WE GET THERE? Determine the molality of C3H8O3. MC3H8O3 = mol/kg
A: As we know, Depression in freezing point, ∆Tf is equal to ∆Tf = Tf° - Tf = i * Kf * m Where Tf°…
Q: 3. The decomposition of hydrogen iodide has an equilibrium constant of 1.84 x 102. What is the…
A: Rules for writing equilibrium constant expression for an equilibrium reaction are : Equilibrium…
Q: At 2°C, the vapor pressure of pure water is 23.76 mmHg and that of a certain seawater sa mmHg.…
A: To estimate the molal concentration of NaCl in the seawater sample, we can use Raoult's law, which…
Q: Sulfuric acid dissolves aluminum metal according to the reaction: 2Al (s) + 3 H2SO4 (aq) ----- Al2…
A: 2Al(s) + 3H2SO4(aq) ----- Al2(SO4)3(aq) + 3H2(g) mass of aluminium (Al) reacts = 15.2 g a) mass of…
Q: If aqueous solutions of Ba(OH)2 and HNO3 are mixed, what products are formed
A: The reaction between Ba(OH)2 and HNO3 is an Acid Base reaction. The type of reaction that occurs…
Q: The charge in the cell is raised until a reaction is achieved Given the following half equations:…
A: “Since you have asked multiple question, we will solve the first three questions for you. If you…
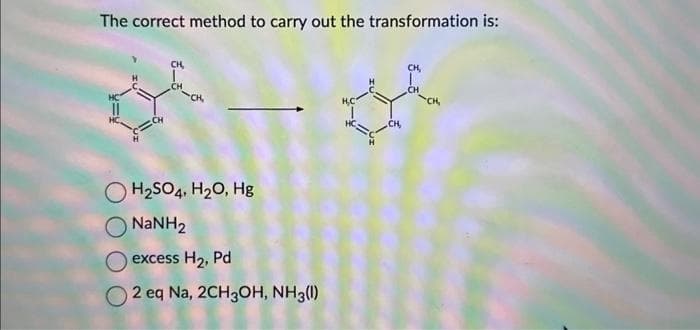
Q: The compound below is treated with N- bromosuccinimide (NBS) in the presence of light. Draw both…
A:
Q: Excess sodium is reacted with 1.00 L of chlorine gas that has a density of 1.88 g/L to form sodium…
A:
Q: estion 2 of 4 Macmillan Learning > Identify the strongest bond, the weakest bond, the longest bond,…
A: The strength of a covalent bond is measured by its bond dissociation energy, that is, the amount of…
Q: Which of the following combinations of electron pair geometry and molecular geometry are not…
A: In a molecule, the electron pair geometry refers to the spatial arrangement of all electron pairs,…
Q: Use the Nernst equation to calculate the cell potential, Ecell, for a galvanic cell consisting of…
A: Given, [Pb+2]= 0.0021 M and [Fe+2] = 0.11 M The half-cell reactions in the cell will be : At Anode…
Q: For the reaction shown here, Kc = 255 at 1000 K. CO(y) + Cl2(y) COCl2(y) Where initial [CO]=0.616…
A: The given reaction is- CO + Cl2<--> COCl2 Initial concentration of ,[CO]= 0.1500 M ,and…
Q: Add lone pairs to these Lewis structures of interhalogen compounds. BrF3 Select Draw Templates More…
A:
Q: What is the freezing point of a solution when 60 g of water (K,(water)=1.86 °C/m) is mixed with 10 g…
A:
Q: or molecule 2,6-dimethyloct-2-ene draw the expanded structure of the molecule not handwritten…
A: The structure of an organic compound is written in the following steps: Determine the number of…
Q: Determine the molarity (M) of the following solutions: 1.50 mol of NaOH in 1000. mL total volume of…
A: moles of NaOH = 1.50 mol Volume of the solution = 1000. mL = 1 L molarity of the solution = ? M
Q: What is the fundamental difference between kinetic stability and thermodynamic stability in chemical…
A: Introduction: Kinetic stability and thermodynamic stability are fundamental concepts in chemistry…
Q: Organic Chemistry HW: CANNOT BE HAND DRAWN 2,6-dimethyloct-2-ene Polarity and Solubility…
A: A question based on reactions of alkenes. Using the name of the compound, structure, polarity and…
Q: Draw the alkene that would react with the reagent given to account for the product formed. ? + H₂O…
A:
Q: 2. Sulfur and oxygen can combine to form sulfur dioxide, SO2, and sulfur trioxide, SO3. Show the…
A: A balanced chemical equation is a representation of a chemical reaction using chemical formulas and…
Q: (4) С excess O OH + НО ОН conc.H₂SO4 H₂O + Heat Х
A: When alcohol is treated with a carboxylic acid in the presence of acid a compound is formed. This…
Q: Hd 5. The following questions refer to the titration curve of a solution of an unknown weak acid. Ve…
A: The question is based on the titrations. we are titrating a weak acid with a strong base. we need…
Q: Question 6 of 10 Which of the following structures is a 20:2 (449) fatty acid? Select the correct…
A: We have to determine among the given options which one represents the 20:2 (∆4,9) fatty acid. The…
Q: 1. Perform the calculations required to answer each of the following questions. Pay attention to…
A: Mass of 1 mol = molar mass 1 mol = 6.022 x 1023 atoms/molecules/ions.
Q: Mn(s) + CuCl₂(aq) E1 S1 e MnCl₂(aq) + Cu(s) E2 Write a balanced equation for the half-reaction that…
A: In a galvanic cell a spontaneous redox reaction occur, At anode( left electrode ) - oxidation(…
Q: What is the IUPAC name of the smallest, most polar alkene that has 4 carbons cis-but-2-ene…
A: The compound we are considering has four carbon atoms, so the parent chain will be butene. The…
Q: Each pictured Lewis structure is invalid. Identify the error in each case. 01 :0- Answer Bank wrong…
A: The given Lewis structures are We have to tell why those Lewis structures are invalid.
Q: 2. For the following reactions, provide the structures for the products with appropriate…
A: alkenes undergo electrophilic addition reactions and free radical substitution reactions.
Q: compound I compound II Il Classify the pair of compounds. They are enantiomers. diastereomers.…
A: According to the given structure we have to decide the relationship between two isomers. 1. The best…
Q: If there is a table such as this one given with Ksp values, does a higher Ksp mean that the kidney…
A: The Ksp of a solution is the solubility product and will be the multiplication of concentration of…
Q: Why is the alpha carbon's hydrogen of a 1,3-dicarbonyl a good hydrogen to be deprotonated by a…
A: 1.3 di carbonyl compounds: The CH2 group present between two C=O groups is called the active…
Q: Predict the direction in which the equilibrium will lie for the following reaction C.H.COO(aq) +…
A:
Q: How many equivalents of base need to be added to fully titrate all acidic protons in…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: A student needs to analyze a mixture containing an alcohol and a ketone by silica gel TLC. After…
A:
Q: 4. A chemist adds some zinc shavings to a beaker containing a blue solution of copper chloride. The…
A: (a) The balanced chemical equation for the reaction between zinc and copper chloride can be written…
Q: Using the molecular orbital model, write electron Configurations for the following diatomic species…
A: The molecular orbital theory represents the energy state and spatial distribution of the electrons…
Q: For the species below draw additional resonance structures (where all atoms have access to an octect…
A: The bond order of bond is the measurement of energy in that bond. Higher the bond order, the higher…
Q: Question: Consider a hypothetical chemical reaction between compound A and compound B: 2A +…
A: Introduction: Chemical reactions and their kinetics play a fundamental role in understanding the…
Q: 4. Using data from Appendix IIB in the eText, calculate A,Gº and K for the reaction at 25°C:…
A: The standard Gibbs free energy change of a reaction is the difference in standard Gibbs free energy…
Q: 2. The half-life of carbon-14 is 5,730 years. Express the amount of carbon-14 remaining as a…
A: The decay of carbon-14 follows an exponential decay model. The amount of carbon-14 remaining as a…
Q: Write notes on: ) Enediol Rearrangement between D-glucose and D-fructose. 5) Difference between…
A: Introduction:Hemiacetals and acetals are important functional groups formed by the reaction of…
Q: 1) The solubility of MgF, in water at 25 °C is 0.13 g/L. Calculate the K, of MgF,
A:
Q: Which of the following aqueous mixtures would be a buffer system? O a. H₂SO4, CH3COOH O b. HNO3,…
A:
Q: why the atomic packing fraction of the simple cubic structure is either greater than or less than…
A: To explain the atomic packing fraction of the simple cubic structure is either greater than or less…
Step by step
Solved in 3 steps with 1 images

- A. OsO4 and NMO B. Br2 and H20 C. Hg(OAc)2, H2O and NaBH4, NaOH D. RCO3H E. BH3-THF and H2O2, NaOH Which reagent will complete this reaction?Rank the following groups in order of decreasing priority. a. – COOH, – H, – NH2, – OH b. – H, – CH3, – Cl, – CH2CI c. -CH2CH3, -CH3, -H, -CH(CH3)2 d. – CH = CH2, – CH3, – C ≡ CH, – H6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pd
- Cyclohexene plus 1) Hg(OAc)2, H2O; 2) NaBH4; yields __________. HO2CCH2CH2CH2CH2CO2H a cyclic diketone cyclohexyne cyclohexanol OHCCH2CH2CH2CH2CHODraw all products of the reaction of (1S,2R)-1-bromo-1,2-dimethylcyclohexane in 80%H2O/20%CH3CH2OHat room temperature..Please provide a correct aanswer Answer the following and Identify the reagent (and its concentration) that is used to confirm the presence of each given: a. CO32- b. S2- c. I-
- reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->Is the reagent for all elimination reactions conc. H2SO4? or just the reactions ones with OH?KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanone
- Classify the reaction below as an oxidation, a reduction, or neither.(CH3)2CHCH2OH → (CH3)2CHCHOA) oxidationB) reductionC) neitherChoose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidRank the following groups in order of decreasing priority. −COOH, −CH2OH, −H, −CHO



