Q: What is the equilibrium that influences the colour change of the Eriochrome Black T indicator? (Why ...
A:
Q: List the following atoms in order of increasing size (atomic radius): Pb, Rn, Ba. A) Rn < Pb < Ba B)...
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Molecular formulas can be written in multiple ways. Isoprene is among the most significant contribut...
A: (a) Option 5th ==> H2CC(CH3)CHCH2 (b) It is condensed formula of isoprene, from this formula we...
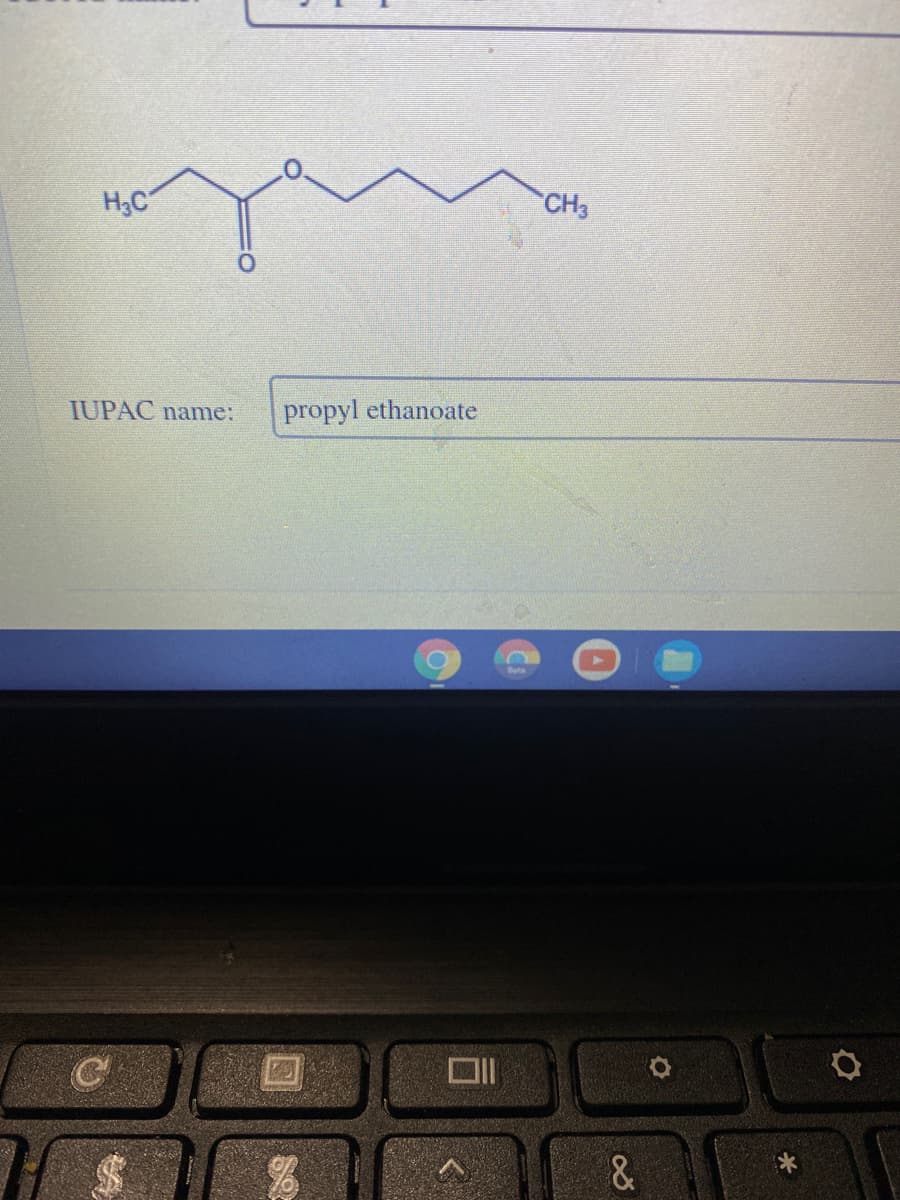
Q: Determine the number of protons and electrons in each of the following ions. Cu^2+
A: Determine the number of protons and electrons in each of the following ions. Cu2+
Q: Observations: Burette Reading (ml) Volume of KCrz07 solution (m) volume of Naso solution (mi) (Final...
A: Given: Normality of potassium dichromate solution ( K2Cr2O7) = 0.025 N Volume of potassium dichrom...
Q: 3) Suppose you add 125g of C and 125g of Cl; to an excess amount of TiO2, how much TiCla can be prod...
A: Answer :- (3) Mass (Practical yield) of TiCl4 = 123.4 g ------------------------------------------...
Q: Calculate the energy released in joules when one mole of polonium-214 decays according to the equati...
A: We have to calculate the energy released in joules.
Q: Define physical and chemical properties, provide three examples of each, discuss their reversibility...
A: Physical properties are those properties which doesn't depend on reaction with any other substance o...
Q: What direction does the net dipole moment for the molecule below point? no net dipole right out O le...
A: The molecule given is,
Q: CL.
A: CH3I => methyl iodide is polar. Thus it's dipole moment is not zero. It's dipole moment is 1.62D...
Q: Rank the substituents shown below in order of Cahn-Ingold-Prelog priorities (1 = highest priority gr...
A: a) The groups given are,
Q: Please help! And what is the direction of the molecular dipole moment in the chloroform molecule? Th...
A: NOTE: please note according to our policies , we can only answer one question at one time. you have ...
Q: (0.41 + 124.9)/1.4 × 10-2 =
A:
Q: A student found that her mixture was 13% NH¼CI, 18% NaCl and 75% SIO2. Assuming that her calculation...
A: on addition of the given percentages, the total percentage of the solution comes out to be as 106% ...
Q: 7. Combustion reactions are a notable source of carbon dioxide in the environment. Using the followi...
A:
Q: 1. What are the three steps of solution formation? Explain each step.
A: Solution is formed when solute and solvent are mixed. This process completes in 3 different steps as...
Q: .Which of the following Is expected to be a major product when 2-methylbutane reacts with bromine in...
A: Halogenation of alkanes :- Alkanes on reaction with halogens in presence of light to form alkyl hal...
Q: An equilibrium mixture contains 0.400 mol of each of the products (carbon dioxide and hydrogen gas) ...
A: The equilibrium reaction taking place is given as, Given: Moles of each product at initial equilibr...
Q: The normal boiling point of a liquid is (a) the temperature at which the vapor pressure equals 1 at...
A: First we have to know about boiling point. Boiling point of a liquid : It is defined as the tempe...
Q: Question i Consider a flask containing 25.00 ml of 0.100 M sodium benzoate (NacaH3COO). This is titr...
A:
Q: region where necking occurs
A:
Q: A What I have learned? Answer the following questions. 1. Can a physical change occur even if there ...
A: Answer 1 : Yes, If there is no chemical change that occurs but can change its physical properties....
Q: The structure of hydroxyacetone sulfate is given. a. What is the molecular formula of hydroxyacetone...
A:
Q: What direction does the net dipole mc for the molecule below point? HH H H-C H.. H. H back O out rig...
A: Cyclohexane is symmetrical molecule as shown in the given image
Q: What is the relationship between the structures below? O constitutional isomers enantiomers diastere...
A: The solution is given below -
Q: Consider a flask containing 25.00 mL of 0.100 M sodium benzoate (NaC6H5COO). This is titrated with a...
A: pH of the solution can be calculated using Henderson-Hesselbech equation.
Q: 5.47 g of potassium chlorate is reacted with 1.55 g sulfuric acid to form chloric acid and potassium...
A: Given: Mass of potassium chlorate (KClO3) = 5.47 g Mass of sulfuric acid (H2SO4) = 1.55 g Atomic mas...
Q: Question 2 The Ksp of Al2S3 is 1.99 x 10-7. Estimate the solubility of this salt in units of g. L-1....
A: The sparingly soluble salt AxBy dissolved and dissociate into ions to form xAy+ and yBx- and has fo...
Q: Sol(1) < Sol(2) < Sol(3) < Sol(4) nich one of the following statements would you conclude to be true...
A: Increase in polarizability increases the solubility of a solute. And hence more interaction develop ...
Q: Graphing practice 1. Jamie bought a new video game and decided to keep track of his scores. Jamie's ...
A: A question based on quantitative analysis, which is to be accomplished.
Q: You have a solution which is 45.6% by mass isopropyl alcohol (MM 60.11 g/mol) in water. The density ...
A: Given: Percent by mass of isopropyl alcohol = 45.6 %. And density of solution = 0.905 g/mL. Assuming...
Q: what mass (in mg) does 2.63 moles of nickle have?
A:
Q: The standard solution is prepared by dissolving a known mass of zinc pellets with conc. HCl. The sol...
A: Given information, Volume (V) = 250 mL = 0.25 L Mass of Zn (m) = 0.9809 g Molecular weight of the zi...
Q: be the thickness of the layer
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: Density Determination Challenge
A:
Q: Determine the R/S orientation of the indicated chiral center(s) in the following molecules. If there...
A:
Q: Define the following term. . A. hypotonic solution B. isotonic solution C. hypertonic solution
A: Since you have posted multiple questions with multiple sub-parts, we are entitled to answer the firs...
Q: The specific rotation, [a]p, for (-)-malic acid is -27. What is the observed rotation for a solution...
A:
Q: Lipid Molecule Laurie acid 2 oleie aeid 3. linoleie acid 4 methyl laurate 5. methyl oleate 6. ethyl ...
A: Lauric acid has both hydrocarbon and polar part Oleic Acid-1 unsaturated bond -hydrophobic and non p...
Q: Sodium catches on fire when it reacts with water. Which electron configuration represents an element...
A: Chemical properties of an element depends on the valence electrons present in its atom. Valence elec...
Q: HO, F F1 F FIB
A:
Q: Cyclic voltammetry is often used for... O determining the resistance of an electrolye solution elect...
A: question is given statements is:- Cyclic voltammetry is often used for the property
Q: Two reactions and their equilibrium constants are given. A + 2 B 2c Kj = 2.67 2 C D K2 = 0.270 Calcu...
A:
Q: A solution of hydrocyanic acid is prepared by dissolving 0.10 mol HCN in enough water to make 1.0 L ...
A: Buffer solution is one that contains a weak acid and its conjugate base. The addition of acid to buf...
Q: How much energy in kilojoules is needed to heat 5.00g of ice from -11.0 °C to 30.0 °C? The heat of f...
A: Given: Mass of ice = 5.00 g. Initial temperature of ice = -11.0 oC And final temperature of ice = 30...
Q: 6. The most abundant isotope of uranium-238 which has an isotopic mass of 258.0508 g/mol. What is th...
A: Given: Uranium-238
Q: Provide a mechanism for the following cyclization to make a spiroketal: H,SO4, H,O НО OH spiroketal
A: The reaction taking place is given as,
Q: Type of bond * Choose H-bond Electrostatic interaction
A: The question is based on the concept of chemical bonding. we have to identify the type of Bond prese...
Q: Arsenic (III) sulfide is reacted with oxygen gas to form arsenic (III) oxide and sulfur dioxide. Cal...
A:
Q: HO H R H OH HO RS v CO,H CO2H SR V
A:
Step by step
Solved in 2 steps with 1 images

- Draw the products formed when attached compound is treated withCH3CH2COCl, AlCl3Stearic acids is used in the production of detergents, soaps and cosmetics because they serves as_________________.I. emulsifierII. emollientIII. lubricantsIdentify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. two hemiacetalsb. disaccharidec. esterd. ethere. alcohol pyranosesThe product is a(n)a. beta 1-6 disaccharideb. beta 1-4 disaccharidec. alpha 1-6 disaccharided. beta 1-2 disaccharidee. alpha 1-2 disaccharidef. alcohol pyranosesg. alpha 1-4 disaccharideh. two hemiacetalsThe reaction type is:a. hydrolysisb. mutarotationc. reduction (hydrogenation)d. hemiacetal formatione. acetal formationf. oxidation (benedict's)
- Draw the organic product(s) formed when CH3CH2CH2OH is treated withfollowing reagent : TsCl, pyridineStearidonic acid (C18H28O2) is an unsaturated fatty acid obtained fromoils isolated from hemp and blackcurrant a.) What fatty acid is formed when stearidonic acid is hydrogenated withexcess H2 and a Pd catalyst?b.) What fatty acids are formed when stearidonic acid is hydrogenatedwith one equivalent of H2 and a Pd catalyst?c.) Draw the structure of a possible product formed when stearidonic acidis hydrogenated with one equivalent of H2 and a Pd catalyst, and onedouble bond is isomerized to a trans isomer.d.) How do the melting points of the following fatty acids compare:stearidonic acid; one of the products formed in part (b); the productdrawn in part (c)?Please provide clear pictures and explanation. Tysm!
- The reagent periodate (IO4-) oxidatively cleaves the carbon–carbon bondsbetween two adjacent carbons carrying hydroxyl groups. Explain howperiodate oxidation might be used to distinguish between methyl glycosidesof glucose in the pyranose and furanose forms.What is the structure of the methy ketone C6H5C3H5O?Identify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. aromaticb. alkenec. alcohold. aldehydee. ketoneThe product is a(n)a. disulfideb. etherc. alcohold. alkanee. aromaticThe reaction type is:a. dehydrationb. reduction (hydrogenation)c. hydrationd. amide synthesise. oxidationf. esterificationg. hydrolysis



