Observations: Burette Reading (ml) Volume of KCrz07 solution (m) volume of Naso solution (mi) (Final Value - Iritial Value) Normality of KCr0, solution (N) Initial value Final Value 0.025 10 9.5 9.5 Normality of NayS,0,26
Q: GIven the following Salinity (S) and Freezing Temperature F tables we can expect for a salinity of…
A: From the table we can see clearly as the salinity is increasing freezing temperature is decreasing.
Q: .A KCI solution having N/50 normality has specific conductivity equal to 0.0102. If K+ and Cl-ions…
A: Degree of dissociation can be calculated as follows
Q: Tabulated Data: Trial I…
A: The data given is, Trial I…
Q: A KCI solution having N/50 normality has specific conductivity equal to 0.0102. If K+ and Cl -ions…
A: Ionic conductance of the salt is equal to the sum of the ionic conductances of the constituent ions.…
Q: My Question) Why this calculation is wrong? I think this calculation is same with question 1. Isn't…
A: The question is based on the concept of quantitative analysis. we have to calculate how much calcium…
Q: By experiment, the normality of H2SO4 was found to be 0.5172. If 39.65 ml of this acid neutralized…
A: 1. Given in the question = Normality of H2SO4 solution = 0.5172 Volume of acid = 39.65 mL Volume…
Q: y! how to Q What g. y zinc anc y ideal ga y What g Q What g y What g W molarit Experim…
A: Hello. Since you have posted multiple questions and not specified which question needs to be solved,…
Q: What is normality of a solution containing 90g of HCI in 2600ml of solution? (Use the given…
A: Normality is defined as number of gram equivalents of the solute dissolved per litre of solution.
Q: Reading for the unknown solution: Trial 1: 7.3 Trial 2: 6.8 Trial 3: 7.3 Reading for the…
A: Normality of AgNO3 (N1) = 0.02 Volume of water sample (V2) = 20 mL It is desired to calculate…
Q: 4. A. Identify the set-up: B. Function: organic H.O HO HSO OH snoonbe 15. A. Identify the set up: B.…
A: A. The setup shown in figure is separation by funnel. B. FUNCTION Basically this separation…
Q: How many mL of 15.1 micromolar stock solution are needed to make 50.0 mL of 4.53 micromolar…
A: Dilution :- If a stock solution of molarity M1 and volume V1 is diluted by adding water to a…
Q: Data Table 1 Enter the calculated molarity and measured conductivity values in the table. Data Table…
A: Note: Conductivity values comes from the readings of Conductometer. Molarity can be calculated as:…
Q: Calculate the equivalent weight (EW) and normality (N) for a solution of H3PO4 given the following…
A: The question is based on the concept of solutions. number of equivalent of a solute present in one…
Q: Approximate mass of NazS203·5H20 needed 1.55 Exact mass of Na2SzO3•5H20 weighed 1.57 Nominal…
A: Nominal concentration means the amount of an ingredient which is expected to be present in a…
Q: grams of glucose C12H22011 are dissolve 1360grams of water. а. 0.00 b. 0.20
A: Molality is defined as number of solute present in 1000 gm of water. Mole= MOLality×mass of…
Q: Determire He average, excluding any trails thal fall outside 0, 0005 M of thte average, O o,…
A: Outliers in a series of a values is the one which lie outside most of the other values. For example…
Q: A ceric sulfate solution that was standardized using 200 mg of arsenic trioxide (MW197.84g/n)…
A: Given: Weight of Arsenic trioxide = 200 mg Volume of solution =38.5 mL Weight of Ferrous Carbonate…
Q: Note: Kindly provide a solution pls. The answer is already provided. Thank you! 1.20 A high…
A: Answer is attached below
Q: Show your calculations below for (* and **) for Solution Solution D 3 tsp sugar 500 Solution A…
A: Percent solution is the solution expressed in the unit %. It may be percentage by weight by volume…
Q: 19 g of unknown organic sample was dissolve in 640 mL of Dicloromethane (DCM). The boiling point of…
A:
Q: ppm used for very dilute solution True False
A:
Q: DATA AND RESULTS A. Weight = 2.83 g B. Weight of water = 10 g C. Boiling point of distilled water =…
A: Solve the given question using ∆Tb formula which contains m, i, and Kb in it.
Q: Preparation and Standardization of KMNO4 solution Experimental data Complete the table below. Trial…
A:
Q: The sucrose solution contained 4.624 g of sucrose in 50 mL of water Clobserved = + 12.50° Anull = +…
A: The question is based on concept of optical rotation . we have to calculate specific rotation of the…
Q: Mass of 2 Vivarin tablets (g) 0.728 Mass of crude caffeine (g) 0.567 Mass of recrystallized caffeine…
A: Given: mass of 2 vivarian tablets = 0.728 g mass of crude caffeine = 0.567 g mass of recrystallized…
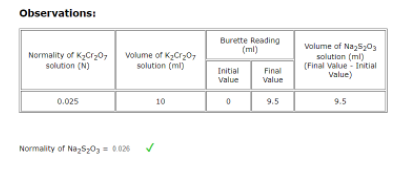
Q: Observations: Burette Reading (ml) Normality of a520 solution (N) Volume of NaSy02 soution (m)…
A: The amount of oxygen present in one liter of water in a dissolved state is called dissolved oxygen.…
Q: Part A What is the normal boring paint in "C ot methyl alcohol ta solution prepared by dissolving 27…
A: The colligative properties are those properties of solutions that depend on the number of solute…
Q: 14406e994a5937753d2#10001 Constants I F olutions ved solute? 25.0 g solute in 0.325 M NaF Express…
A:
Q: Mass Na CO3 Vi HC1,mL 0.00 0.10 Vf HC1,mL 36.75 Trial VHC) used, ml MHCI 1 0.2135 0.2025 0.2112…
A: Since you have asked a question with multiple sub-parts, as per our company guidelines we are…
Q: BaSO4 BaSO4 in (NH4)2SO4
A: This question is related to gravimetric analysis.
Q: Data Table1: Height (mL) for Stock Sugar Solutions XX Sugar Solution Concentration 0.0% Solution…
A: In the Sugar Solution Concentration Vs. Bulb Height Volume plot, we take Sugar Solution…
Q: Com X Bb Mas X Mas X Hom X K! Kahx Exan X K! Kah X Mitc X КI Kah X K Kah x K! Kah x Kah x K Kahx C…
A: Dipole moment:The charge separation of the molecule produces dipole moment. Dipole moment arises…
Q: A solution contains 6 umol Na2SO4 in 284 ml. How many ppm Nat does it contain? A.W Na=23 S=32 0=16…
A: The concentration in ppm is calculated as the ratio of grams of solute and the volume of the…
Q: What is the procedure of Labxchange Simulation Micropipetting Solutions? Or simply what are the…
A: Micropipettes are the adjustable and precision instruments used to transfer the volumes of the…
Q: Ter solution differ from an unbuffered solution
A:
Q: If the true melting point for lauric acid is 43.2 oC what is the percent error?
A:
Q: Calculate the following for each run: the initial molarities of I- and H2O2 (from the solutions…
A: This experiment is based on iodine-clock reaction in which iodide ion reacts with hydrogen peroxide…
Q: 10/11 21 85% ppun21@ole.augie edu Highest Melting Point CaCl NH,OH 1 PCI3 KI 2 Cl2 CH4 * SUBMIT…
A: Answer Order of melting point of given compounds
Q: mL of H2SO4 solution 75 (specificgravity =1.18) containing 49% H2SO4 (w/w) is diluted to 590 mL.…
A: Given information-: Volume of solution = 75 ml ( V1) Specific gravity = 1.18 H2SO4 (w/w) = 49 %…
Q: True solution is transparent. * O True O False
A: True solution is when solute is dissolved uniformly through out the solution. And is homogeneous…
Q: Part A 65 g of Na,SO, in 265 mL of Na SO, solution Give your answer to two significant figures.…
A: From given data we can easily find molarity as:
Q: Example: Calculation the weight of barium iodate Ba(IO3)2 if it dissolved in 500mL of distilled…
A: Given data volume of solution = V = 500 mL = 0.500 L molar mass of Ba(IO3)2 = 487 g/mole Solubility…
Q: Calculate the following for each run: the initial molarities of I- and H2O2 (from the solutions…
A: Total volume of the reaction mixture = 65.5 mL [KI]stock = 0.04 M [H2O2]stock = 0.04 M…
Q: Calculate the molarity of 15.4 g of sucrose (C12H22011) in 74.0 mL of solution, [Molar mass of…
A: Hello, since your question has multiple parts, we will solve the first question for you. If you want…
Q: The molarity of 90.0% (w/w) H2SO4 solution (density = 1.80 g/mL) is [Select ] . The normality of the…
A: 28) Given, mass % of H2SO4 solution = 90.0% Density of the solution = 1.80 g/mL Molarity of the…
Q: Use the following atomic masses (in g/mol): K = 39.1; Mn = 54.94; O = 16; H = 1; P = 30.97; C =…
A: Given acid-base reaction is H2SO4 + 2 NaOH --> Na2SO4 + 2 H2O At equivalents point Na…
Q: A- STANDARD PHOSPHATE SOLUTION: Standard solution 1 4 6. Conc of PO4 (ppm) O-00 O.2 0.4 0.6 0.8 1-0…
A: Calculations: Q.1: In order to find the ppm of phosphate in the unknown water sample, we have to…
Q: Calculate the new boiling point of a solution containing 5.27 g of sources (C12H22O11) and 45 g of…
A:
Q: Density of solution: Trial 1: 1.2 g/mL Trial 2: 1.2 g/mL Trial 3: 1.2 g/mL Average density = 1.2…
A:
how to solve the normality here? that is the answer but I do not know how to compute
Step by step
Solved in 2 steps

- 4. A fat sample with combination of acids contain standard hydrochloric acid for blank and sample with 8mL and 5mL respectively. The normality of the standard hydrochloric acid is 0.93N and the weight of the sample is 3 grams. Calculate the saponification value.Calculate the gravimetric factor of: FeO in Fe2O3 Bi2S3 in BaSO4 BaSO4 in (NH4)2SO4 Mn2O3 in Mn3O4 NOTE: Present complete solution and include the detailed computation of the molecular weight. Express your final answers up to FOUR decimal places.Bristol Community College Fall River, Massachusetts Experiment 6: Molar Mass of a Molecular Solid from Freezing Point-Depression Measurement Name: __________________________________ Date: _______________ Approved: ___________ DATA SHEET Mass of lauric acid (in Part II) Mass of benzoic acid (in Part II) Freezing temperature of pure lauric acid (from Part I) data from Video 2 of Part I Freezing temperature of solution (from Part II) data from Video referenced in Part II Freezing point depression, Tf ( = Tf, lauric acid – Tf, solution) Molality (m) of solution ( Eq. 1) Moles of benzoic acid ( Eq. 2) Experimental molar mass of benzoic acid (Eq. 3) Calculate the molar mass of benzoic acid, C6H5COOH. Percent error Summary Questions A student determines…
- 1. Order: isoproterenol 2 mg in 500 mL D5W to infuse at 15 mL/hrPatient weight: 20 kgHow many mcg/kg/min is the patient receiving? (Round to the 2nd decimal place/hundredth placeI would need help with these questions. The method referenced above was followed by a student and she got the following results: Caffeine Std. Conc. (ppm) Absorbance 100 1.806 50 0.899 40 0.724 30 0.545 20 0.365 10 0.183 Further, she analyzed an unknown sample and she got the following results: Sample # Absorbance 1 0.398 1. Graphically find the concentration in ppm and then calculate in mol/L of Caffeine in Sample #1? 2. Which solvent was used to extract Caffeine and why? Why is it necessary to do extraction three times? 3. Cuvettes used in this experiment were made from which material and why? 4. At what wavelength the Absorbance was measured?Density of solution:Trial 1: 1.2 g/mLTrial 2: 1.2 g/mLTrial 3: 1.2 g/mL Average density = 1.2 g/mL What is the relative average deviaion, %?
- 50 mg of granular salt is briefly stirred into a glass container of 1/5 liter volume filled with water. After briefly mixing, some salt is still seen at the bottom of the class. The visible salt is filtered from the water, weighed and determined to have a mass of 20 mg. What is the dissolved phase concentration of salt remaining in the water? Provide your answer to the nearest whole number in units of mg/l.Hello, I hope you are doing well on this fine day. For the following quetion please read carefully the question and instruction. PLEASE ANSWER QUESTION IN 15 MINTUES NOT MORE PLEASE AND THANK YOU. If you do answer the question correctly and post it in the next 15 minutes, NO NEED TO SHOW THE WORK, I JUST WOULD LIKE THE CORRECT ANSWER AS SOON AS POSSIBLE. I will write a wonderful and generous feedback/review/rating about you. You determined experimentally the density of a solution in triplicate. Considering the results what is the standard deviation of your results? Answer your question with the correct number of significant figures. The answer should be in units of g/mL Trial 1: 0.9997 g/mL Trial 2: 0.9985 g/mL Trial 3: 0.9987 g/mLYou are asked to calculate molar conductivity of a NaCl solution with a concentration of 0.03125 mol/L according to the following equation, where χ is the conductivity and is 1.41x10-3 (±0.02) ohm^-1 cm^-1 and C is the concentration of the solution. The uncertainty of the volume measurement in determining concentration is given as ±0.02 and the mole value is given with no significant error. Calculate molar conductivity in ohm^-1 cm2mol^-1 and give your result with necessary significant figures.
- Calculate the Constant Weight (in grams) of the Empty Crucibles. Show your solution. Crucible No. 1 2 3 Weighing 1 22.6035 22.0223 24.1535 Weighing 2 22.6017 22.0204 24.1533 Weighing 3 22.5994 22.0199 - Weighing 4 22.5992 - - Constant Weight, g ??? ??? ???Patients undergoing an upper gastrointestinal tract laboratory test are typically given an X-ray contrast agent that aids with the radiologic imaging of the anatomy. One such contrast agent is sodium diatrizoate, a nonvolatile water-soluble compound. A 0.378-m solution is prepared by dissolving 38.4 g sodium diatrizoate (NaDTZ) in.l.60 102 mL water at 3 1.2C (the density of water at 31.2C is 0.995 g/cm3). What is the molar mass of sodium diatrizoate? What is the vapor pressure of this solution if the vapor pressure of pure water at 31.2C is 34.1 torr?200 mg of granular salt is briefly stirred into a glass container of 1/2 liter volume filled with water. After briefly mixing, some salt is still seen at the bottom of the class. The visible salt at the bottom of the glass is filtered from the water, weighed and determined to have a mass of 50 mg. What is the dissolved phase concentration of salt remaining in the water? Provide your answer to the nearest whole number in units of mg/l.









