helium tank is pressurized to 28.0 atm. When connected to this tank, a balloon will inflate be tank is greater than the atmospheric pressure pushing on the outside of the balloon. Assuming the lefinitely and never burst, the pressure would eventually equalize causing the balloon to stop inflatin the balloon be when this happens? sume atmospheric pressure is 1.00 atm. Also assume ideal behavior and constant temperature. lume: L
helium tank is pressurized to 28.0 atm. When connected to this tank, a balloon will inflate be tank is greater than the atmospheric pressure pushing on the outside of the balloon. Assuming the lefinitely and never burst, the pressure would eventually equalize causing the balloon to stop inflatin the balloon be when this happens? sume atmospheric pressure is 1.00 atm. Also assume ideal behavior and constant temperature. lume: L
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter1: Gases And The Zeroth Law Of Thermodynamics
Section: Chapter Questions
Problem 1.13E: Hydrogen gas is used in weather balloon because it is less expensive than Helium. Assume that 5.57 g...
Related questions
Question
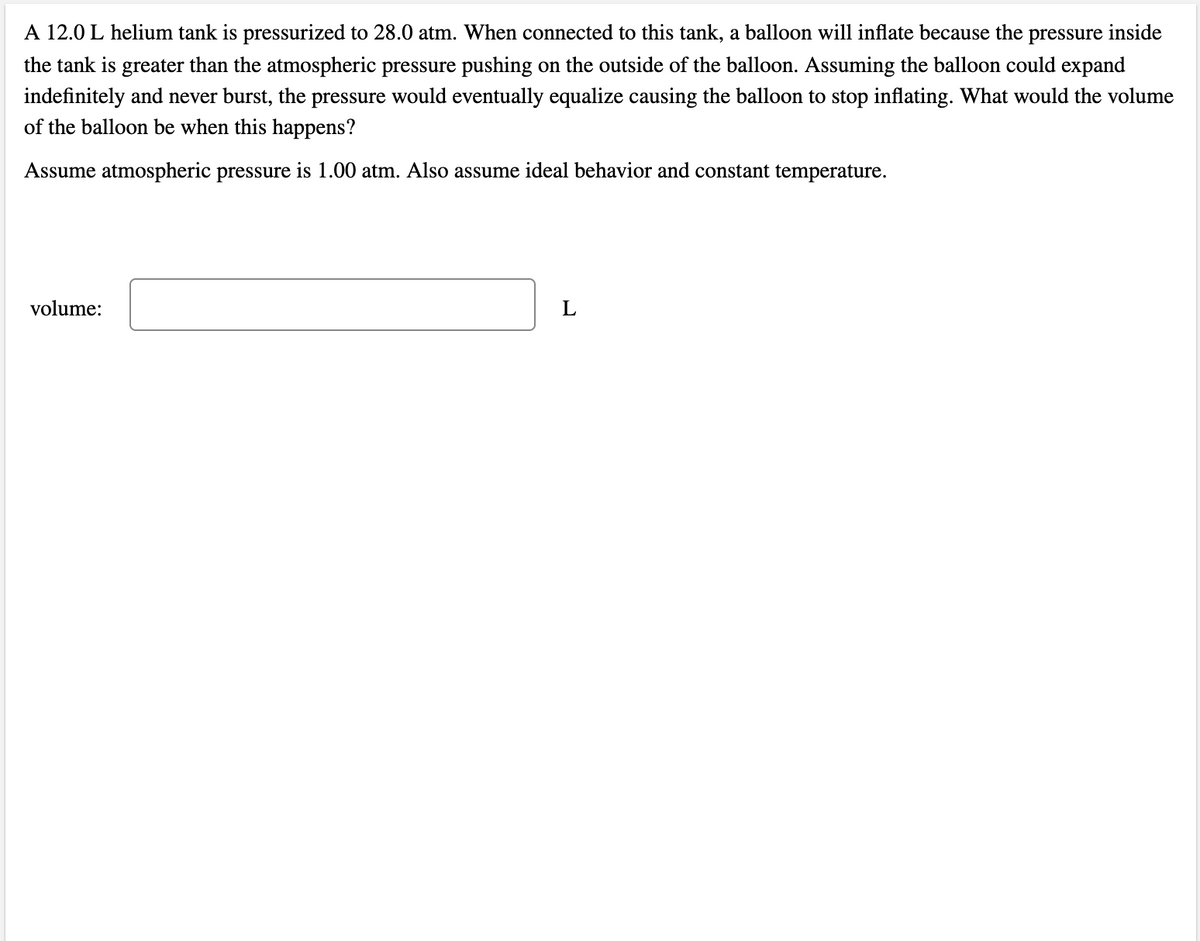
Transcribed Image Text:A 12.0 L helium tank is pressurized to 28.0 atm. When connected to this tank, a balloon will inflate because the pressure inside
the tank is greater than the atmospheric pressure pushing on the outside of the balloon. Assuming the balloon could expand
indefinitely and never burst, the pressure would eventually equalize causing the balloon to stop inflating. What would the volume
of the balloon be when this happens?
Assume atmospheric pressure is 1.00 atm. Also assume ideal behavior and constant temperature.
volume:
L
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning