Hemoglobin from different species can have different numbers of subunits. Let's consider three hemoglobin molecules: n = 1, n = 4, and n = 8, where each subunit has an oxygen P50 = 30 torr. (Recall that P5o is essentially a Kp and that oxygen partial pressure (torr) is directly proportional to concentration.) Which molecule transports the most O2 between the lungs (pO2 = 100 torr) and peripheral tissues (pO2 = 30 torr)? Please justify your answer.
Hemoglobin from different species can have different numbers of subunits. Let's consider three hemoglobin molecules: n = 1, n = 4, and n = 8, where each subunit has an oxygen P50 = 30 torr. (Recall that P5o is essentially a Kp and that oxygen partial pressure (torr) is directly proportional to concentration.) Which molecule transports the most O2 between the lungs (pO2 = 100 torr) and peripheral tissues (pO2 = 30 torr)? Please justify your answer.
Human Physiology: From Cells to Systems (MindTap Course List)
9th Edition
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Lauralee Sherwood
Chapter11: The Blood
Section: Chapter Questions
Problem 1SQE
Related questions
Question
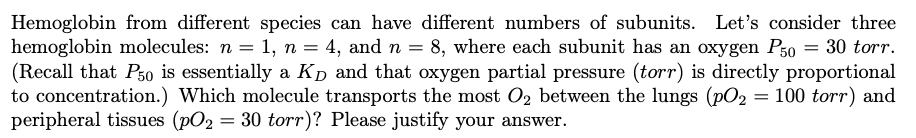
Transcribed Image Text:Hemoglobin from different species can have different numbers of subunits. Let's consider three
hemoglobin molecules: n = 1, n = 4, and n = 8, where each subunit has an oxygen P50 = 30 torr.
(Recall that Pso is essentially a Kp and that oxygen partial pressure (torr) is directly proportional
to concentration.) Which molecule transports the most O2 between the lungs (pO2 = 100 torr) and
peripheral tissues (pO2 = 30 torr)? Please justify your answer.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning