HO ト=人 a) 1. Hz cgoi indlar catalyst OH Os ou, BuoH /H20 2. C+1-) b) 1. Osou, BUOH/HzO 2. Hz cgo, Lindlar catalyt c) I. Os Ou, BUOH /HzO Na css, NH3(8) -78°c 2. 1. Na c3), NH3ce,-78°c Os ou, BuOH /HzO 2.
HO ト=人 a) 1. Hz cgoi indlar catalyst OH Os ou, BuoH /H20 2. C+1-) b) 1. Osou, BUOH/HzO 2. Hz cgo, Lindlar catalyt c) I. Os Ou, BUOH /HzO Na css, NH3(8) -78°c 2. 1. Na c3), NH3ce,-78°c Os ou, BuOH /HzO 2.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.9QAP
Related questions
Question
I’m having hard time finding the best reagent for this problem can you please help?
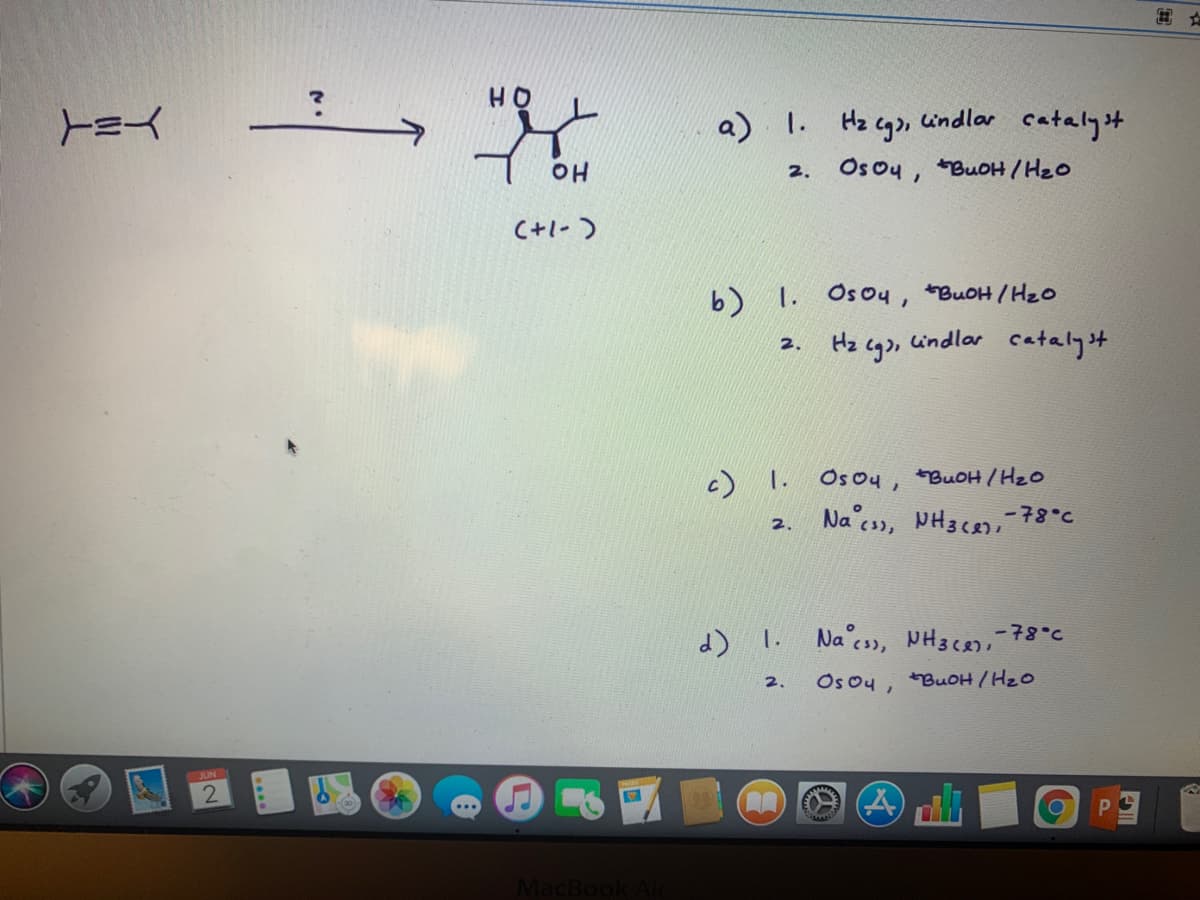
Transcribed Image Text:HO
a) I. Hz cgo,
undlar
catalyst
Os ou, BuOH/ H2o
OH
2.
C+1-)
b) 1. Osou, BUOH/HzO
2. Hz cg)n
indlar cataly't
c) 1. OsOy, BuOH /Hzo
Na cs, PH3c8
-78°c
2.
Na c3), NH3C8,-78°c
Os ou, BUOH /HzO
2.
MacBoo
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

