Consider the following reaction, A-B, the rate of reaction is 7.5 x 10-6 moldm 3s 1 The time taken for the reaction is 9.5 minutes. What is the concentration? O a. 4.275 x 10 moldm -3 O b. 427.5 x 10 3 moldm OC 42.75 x 10-3 moldm 3 Activate Windows Go to O d. 4275 x 10- moldm
Consider the following reaction, A-B, the rate of reaction is 7.5 x 10-6 moldm 3s 1 The time taken for the reaction is 9.5 minutes. What is the concentration? O a. 4.275 x 10 moldm -3 O b. 427.5 x 10 3 moldm OC 42.75 x 10-3 moldm 3 Activate Windows Go to O d. 4275 x 10- moldm
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter17: Equilibrium
Section: Chapter Questions
Problem 3QAP: How do chemists envision reactions taking place in terms of the collision model for reactions? Give...
Related questions
Question
Transcribed Image Text:5 ScAc4590506f/General?threadld-19:9597603ecb3f40b5ab24e8aee75e899c@thread.tacv28tcbx-channel
Posts Files
Class Notebook Moodle
2 more v
O Meet
versity of Technology and Applied Sciences - Ibri
Course search
Mazin Al Hinaai
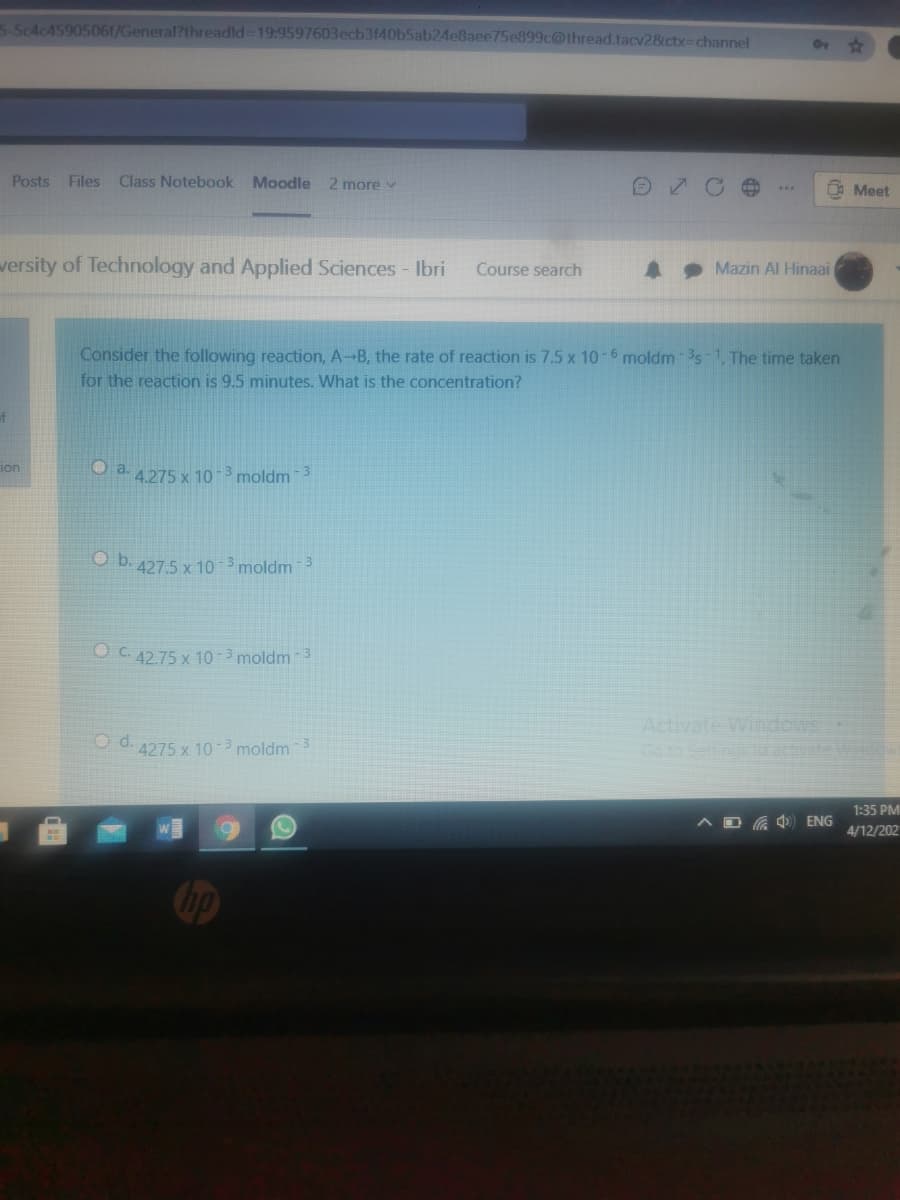
Consider the following reaction, A-B, the rate of reaction is 7.5 x 10-6 moldm 3s1. The time taken
for the reaction is 9.5 minutes. What is the concentration?
ion
O a. 4275 x 10
-3
moldm 3
O b. 427.5 x 10 3 moldm 3
OC. 42.75 x 10 -3 moldm 3
Activate Windows
Go to Settings to actvate Wdow
O d. 4275 x 10 -3 moldm
1:35 PM
A D A 4)
ENG
4/12/202
Cop
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning