Q: The reaction PC13(g) + Cl₂(g) = PC15(g) has AH = -87.9 kJ, AS° = -170 J/K, K = 3.32 x 10 at 298 K.…
A:
Q: 3.35 Arrange the following compounds in order of decreasing acidity: (a) CH3CH=CH2, CH3CH₂CH3,…
A: Given : formula of molecules.
Q: 10:43 AM Mon Oct 17 ← Question 15 of 15 www Q Curved arrows are used to illustrate the flow of…
A:
Q: Which reaction corresponds to the standard enthalpy of formation, Δ?∘f,H2O2(l)?…
A: Standard enthalpy of formation: The standard enthalpy change for the formation of 1 mole of a…
Q: HC₂H5O₂ has a K₂ = 1.3 x 10-5. What is the pH of a 0.181M solution of HC3H502?
A:
Q: Determine the point group of each molecule. Show the different symmetry elements. 10. POCI3
A:
Q: Perform the conversions between energy units. 617 kJ= 2275 kcal= 7.76×10^6 J=
A: We can use the following conversion data to interconvert energy units:
Q: The following structure is named by selecting the correct components for the IUPAC name. The…
A: Given structure find out the iupac name
Q: A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas…
A: Given data :
Q: 1. How many moles are in 3.5 g of aspirin (C₂H8O4)?
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: a. Sodium oxide reacts with carbon dioxide to form sodium carbonate. b. Calcium metal reacts with…
A: We have to write the balanced chemical equation A) Sodium Oxide reacts with carbon dioxide to form…
Q: A solution is prepared where 12.6 g of nonvolatile solute (80.30 g/mol, nonelectrolyte) is added to…
A: A solution is prepared where 12.6 g of nonvolatile solute (80.30 g/mol, nonelectrolyte) is added to…
Q: 1. If 6.95 kJ of heat is applied to a 1012-g block of metal, the temperature of the metal increases…
A: Given, Heat supplied, Q=6.95 kJ mass of block , m= 1012 g change in temperature , ∆T= 11.4°C
Q: unanswered
A: In this question, we have to answer which quantity will increase or decrease in magnitude if…
Q: A major component of gasoline is octane (C₂H₁). When octane is burned in air, it chemically reacts…
A: Given Mass of octane = 9.50 g Mass of water = ?
Q: Write a balanced molecular equation for aluminum metal reacting with a HCI solution. Include all…
A:
Q: Is a hydrate a mixture or pure substance? Explain
A: Given : Type of compound is hydrate. To determine: whether hydrate is a mixture or pure substance.
Q: A system does 506 kJ of work and loses 271 kJ of heat to the surroundings. What is the change in…
A: Please find your solution below : According to First Law of Thermodynamics, heat can neither we…
Q: Citric acid (C6 H8 O7) named for its natural occurrence in citrus fruits, is produced in large…
A: we have to calculate the mass of citric acid produced
Q: A 100.0 mL solution containing 0.923 g of maleic acid (MW = 116.072 g/mol) is titrated with 0.306 M…
A:
Q: Calculate the initial concentration of a reactant that took 5 min to reach a final concentrationof…
A:
Q: The initial concentration of a reactant in a zero-order reactioin is 1M. The rate constant k is…
A:
Q: 3. Which is a stronger base, and why? I-Z: H N
A:
Q: 4 Book erences IW Be sure to answer all parts. Select the anti periplanar geometry for the E2…
A: For E2 elimination H and Br should be antiperiplanar to each other i.e 180° .
Q: The compressibility factor of nitrogen does not exceed 0.9 at 126K and 300 atm True or False? Show…
A:
Q: 4. Consider the following 3 step reaction mechanism: Step 1 H₂ 2H fast Step 2 Step 3 H + HCO H + CO…
A: Recall the given reaction mechanism -Step 1 H2 ↔ 2 H fastStep 2…
Q: The specific heat of a certain type of metal is 0.128 J/(g⋅∘C).What is the final temperature if 305…
A:
Q: In carrying out the first standardization in this experiment, a student used 0.5169 g of potassium…
A:
Q: PROBLEM SOLVING Show complete solutions for each of the problems. Express your final answers in 2…
A: Solution: We know that to balance redox reaction in acidic/basic medium some important steps we have…
Q: In the lab, 5.21 mL of a 11.6 M HCIO, (100.5 g/mol) solution is diluted to 625 mL. What is the…
A: Given data HClO4= 11.6 M or 5.21 ml diluted by 625 ml
Q: What information does a molecular formula provide that an empirical formula does not provide?…
A:
Q: True or False The temperature at the end of the adiabatic compression of a Diesel engine with a…
A: In the given question we have to identify the statement is true or false, we have to apply the…
Q: If you have 0.2 moles of a substance in 500 mL of water, what is the molarity? Remember to convert…
A: Given: Moles of the substance = 0.2 mol The volume of the solution = 500 mL Since 1000 mL = 1…
Q: Starting with triphenylphosphine and any desired alkyl halide, plus any desired aldehyde or ketone,…
A: Given triphenylphosphine Starting material
Q: Which of the following are favorable characteristics of an extraction solvent? Circle all that…
A: We have been given five characteristics of an extraction solvent. We have been asked to choose the…
Q: If KOH solution were used in Part B instead of Ba(OH), solution, then the chem A(OH)2 solution,…
A: Conductivity of a solution depends on the number of ions present in the solution. More the number…
Q: Calculate the standard enthalpy of formation for H2O2(l). Use the standard enthalpy of formation…
A: The given compound is H2O2(l). The standard enthalpy of formation for H2O2(l) is
Q: 2. How many grams of sulfuric acid (H₂SO4) would you measure to make 1L of a 1M solution? Show your…
A: Molarity = Moles of solute/ Volume of Solution(L) Moles of Solute = Mass of solute/Molar mass of…
Q: Determine the point group of each molecule. Show the different symmetry elements. 9. BF₂ CL
A:
Q: Task 3.2. Calculate the standard enthalpy of reaction of some thermochemical equations governed by…
A: Given : value of ∆H
Q: Ammonia NH3 chemically reacts with oxygen gas O2 to produce nitric oxide NO and water H2O .…
A:
Q: Ammonium phosphate NH43PO4 is an important ingredient in many fertilizers. It can be made by…
A: A balanced chemical equation contains an equal number of atoms of each elements on both sides of the…
Q: Fill in the missing information: symbol ст atom or ion? check all that apply neutral atom neutral…
A:
Q: 9.71 g of MgSO₄ is placed into 100.0 mL of water. The water's temperature increases by 6.70°C.…
A: Here we are required to find the Enthalpy of dissolution of MgSO4.
Q: Are we able to completely separate pigments from each other using paper chromatography?
A: Paper chromatography separation of pigments
Q: Balance the following chemical equations. Fe (s) + _Mg (s) +_________N₂ (g) → _ _KNO3 (s) →→→ KNO₂…
A: Balance chemical equation: balance chemical equation is the reaction in which number of atoms of all…
Q: On a single-component phase diagram, one must cross a coexistence line to move from the region for…
A: Water (H2O) system is an example of one component system. Water can exist in three forms (phases),…
Q: How many grams of KNO3 (101.1 g/mol) must be dissolved in 434.1 mL of water in order to create a…
A: In this question a solution of KNO3 which has a concentration of ions equal to 1.61 m. We have to…
Q: If the ground state of a reaction's product is lower than the ground state of the substrate, which…
A: Ground state of product is lower than Ground state of reactant.
Q: If all of the components have a reasonably similar size and molecular weight, which of the suspected…
A:
Draw the structure of the reactions' products, show mechanism shortly.
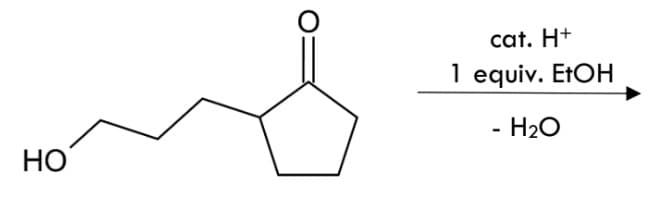
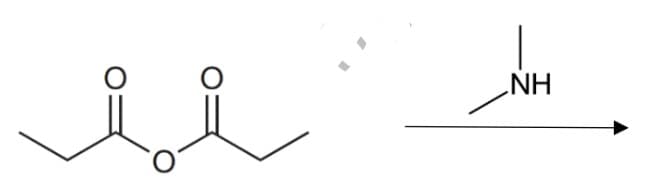
Step by step
Solved in 2 steps with 1 images


